Scalable Industrial Synthesis of 3-Amino-2,2-Dimethylpropanamide for Aliskiren Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antihypertensive agents, particularly for renin inhibitors like Aliskiren. Patent CN100588646C introduces a transformative industrial preparation method for 3-amino-2,2-bismethylpropionamide, a pivotal structural fragment essential for the assembly of this potent drug. This innovation addresses longstanding bottlenecks in synthetic efficiency by utilizing cyanoacetamide, a commercially abundant starting material, rather than scarce or hazardous precursors. The strategic shift away from toxic reagents and complex purification protocols marks a significant advancement in green chemistry and process safety. By streamlining the synthetic sequence into manageable alkylation and reduction steps, this technology offers a viable solution for high-volume production demands. 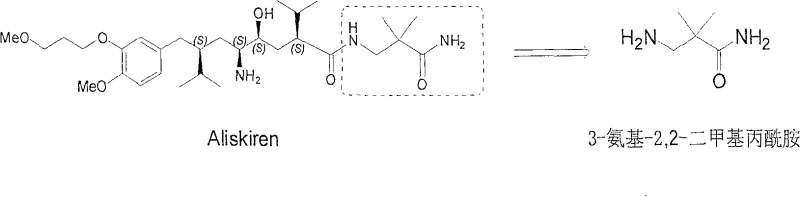
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key amine fragment has been plagued by severe safety hazards and inefficiencies that render traditional methods unsuitable for modern GMP manufacturing. One prominent literature route involves the reaction of 1-nitroisobutene with potassium cyanide, followed by reduction with iron powder. 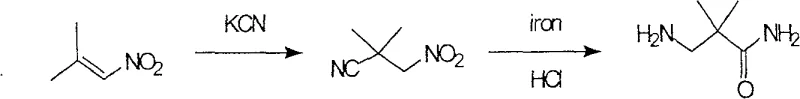 This pathway is fundamentally flawed for large-scale operations due to the extreme toxicity of cyanide salts, posing unacceptable risks to personnel and the environment. Furthermore, the reduction step generates substantial by-products, necessitating tedious column chromatography purification which results in abysmal overall yields of approximately 5 percent. Another reported approach utilizes N-hydroxysuccinimide derivatives requiring multi-step protection and oxidation sequences.
This pathway is fundamentally flawed for large-scale operations due to the extreme toxicity of cyanide salts, posing unacceptable risks to personnel and the environment. Furthermore, the reduction step generates substantial by-products, necessitating tedious column chromatography purification which results in abysmal overall yields of approximately 5 percent. Another reported approach utilizes N-hydroxysuccinimide derivatives requiring multi-step protection and oxidation sequences. 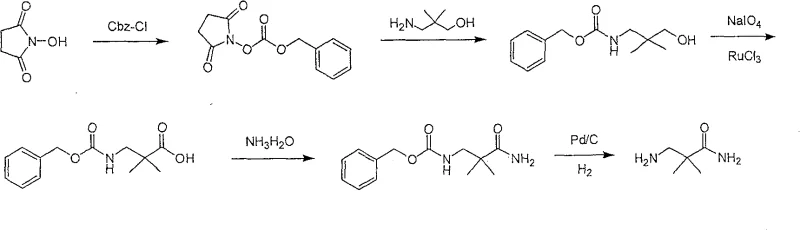 This alternative suffers from excessive route length and employs expensive ruthenium trichloride oxidants that require impractically long reaction times of up to three days, creating significant bottlenecks in production throughput.
This alternative suffers from excessive route length and employs expensive ruthenium trichloride oxidants that require impractically long reaction times of up to three days, creating significant bottlenecks in production throughput.
The Novel Approach
In stark contrast, the patented methodology revolutionizes the production landscape by adopting a concise two-step strategy starting from cyanoacetamide. The process initiates with a straightforward alkylation where sodium ethoxide facilitates the introduction of two methyl groups, effectively constructing the gem-dimethyl core. This is followed by a catalytic hydrogenation or chemical reduction step that converts the nitrile functionality directly into the primary amine. This streamlined approach eliminates the need for hazardous cyanide handling entirely and bypasses the requirement for chromatographic separation. Instead, purification is achieved through efficient solvent washing and crystallization techniques, which are inherently scalable and cost-effective. The overall yield is dramatically improved to a range of 53 to 74 percent, representing a massive leap in material efficiency compared to legacy methods.
Mechanistic Insights into Alkylation and Catalytic Hydrogenation
The success of this synthesis relies heavily on the precise control of enolate chemistry during the initial alkylation phase. Sodium ethoxide acts as a strong base to deprotonate the methylene group adjacent to the carbonyl in cyanoacetamide, generating a nucleophilic enolate species. This intermediate reacts rapidly with methylating agents such as methyl iodide or dimethyl sulfate to install the requisite methyl groups. Maintaining the reaction temperature between room temperature and 80 degrees Celsius is critical to prevent over-alkylation or decomposition of the sensitive nitrile group. The subsequent transformation involves the reduction of the nitrile to a primary amine, typically achieved via catalytic hydrogenation using Raney Nickel or Palladium on Carbon. This heterogeneous catalysis allows for clean conversion under moderate pressures of 1 to 10 atmospheres, ensuring high selectivity for the amine without reducing the amide functionality.
Impurity control is intrinsically built into this mechanism through the physical properties of the intermediates. Unlike the messy iron powder reductions of the past which create sludge and complex mixtures, the catalytic hydrogenation produces a cleaner reaction profile. The use of solvents like ethanol or methanol, potentially mixed with ammonia water, helps stabilize the intermediate and suppress side reactions. Any unreacted starting material or mono-alkylated by-products can be effectively removed during the workup phase using dichloromethane extraction and brine washing. The final product isolation via filtration after solvent removal ensures a high-purity solid suitable for downstream coupling reactions in Aliskiren synthesis, minimizing the risk of carrying over genotoxic impurities or heavy metals into the final API.
How to Synthesize 3-Amino-2,2-Dimethylpropanamide Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety in a pilot or plant setting. The process begins with the preparation of the alkylated nitrile intermediate, followed by the reduction step which can be tuned based on available equipment. Detailed standard operating procedures regarding stoichiometry, addition rates, and safety interlocks are essential for successful implementation.
- Alkylation of cyanoacetamide using sodium ethoxide and methylating agents like methyl iodide or dimethyl sulfate to form dimethylcyanoacetamide.
- Catalytic hydrogenation of the nitrile group using Raney Nickel or Pd/C under controlled pressure and temperature to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this patented route offers profound economic and logistical benefits that extend beyond simple yield improvements. The substitution of scarce, custom-synthesized starting materials with commodity chemicals like cyanoacetamide drastically reduces raw material costs and mitigates supply risk. Cyanoacetamide is widely available from multiple global suppliers, ensuring a stable and competitive sourcing landscape that protects against single-source bottlenecks. Furthermore, the elimination of column chromatography represents a massive reduction in processing time and solvent consumption, directly translating to lower manufacturing overheads and increased batch turnover rates.
- Cost Reduction in Manufacturing: The removal of expensive and toxic reagents such as potassium cyanide and ruthenium trichloride significantly lowers the bill of materials. Additionally, avoiding chromatographic purification reduces the consumption of silica gel and vast quantities of organic solvents, leading to substantial savings in waste disposal and solvent recovery costs. The simplified workup procedure also reduces labor hours and equipment occupancy time, enhancing overall plant productivity without compromising quality standards.
- Enhanced Supply Chain Reliability: By relying on robust, non-hazardous chemistry, the manufacturing process becomes less susceptible to regulatory shutdowns or safety incidents that could disrupt supply continuity. The use of standard hydrogenation equipment and common solvents means that production can be easily transferred between different manufacturing sites if necessary. This flexibility ensures that pharmaceutical partners can maintain consistent inventory levels of this critical intermediate, supporting uninterrupted API production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations like filtration and distillation that are standard in chemical engineering. The avoidance of heavy metal oxidants and cyanide waste simplifies environmental compliance and reduces the burden on wastewater treatment facilities. This aligns with modern sustainability goals and reduces the ecological footprint of the manufacturing process, making it an attractive option for companies focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer.
Q: Why is the new synthesis route preferred over the nitroisobutene method?
A: The traditional nitroisobutene route relies on highly toxic potassium cyanide and yields only about 5% product requiring complex purification. The new method uses safe, commodity cyanoacetamide with significantly higher yields and simpler workup.
Q: Does this process require column chromatography for purification?
A: No, a key advantage of this patented method is the elimination of column chromatography. The intermediates and final product can be purified through solvent washing and crystallization, which is critical for industrial scale-up.
Q: What catalysts are suitable for the reduction step?
A: The patent specifies the use of conventional hydrogenation catalysts such as Raney Nickel or Palladium on Carbon (Pd/C), allowing for flexible process optimization based on availability and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2,2-Dimethylpropanamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antihypertensive therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-amino-2,2-dimethylpropanamide meets the highest international standards. Our commitment to process excellence allows us to deliver materials that facilitate smooth downstream synthesis for our global partners.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method. We encourage potential partners to contact us for specific COA data and route feasibility assessments to ensure seamless integration into your existing supply chain.
