Advanced Copper-Catalyzed Synthesis of 2-Substituted Thiazolo Benzimidazoles for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 2-Substituted Thiazolo Benzimidazoles for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN109467564B introduces a groundbreaking methodology for the synthesis of 2-substituted thiazolo[3,2-a]benzimidazole compounds, a class of heterocycles known for their significant pharmacological potential including anti-tumor, anti-inflammatory, and antibacterial activities. This innovation addresses long-standing challenges in heterocyclic chemistry by utilizing a direct cycloaddition strategy between terminal alkynes and 2-mercaptobenzimidazoles. By employing a copper salt catalyst system in the presence of iodine and a specific ligand, the process achieves high selectivity and yield under remarkably mild conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing of complex pharmaceutical intermediates, eliminating the need for hazardous pre-functionalized starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazolo[3,2-a]benzimidazole derivatives has been plagued by inefficient methodologies that rely on pre-functionalized unsaturated compounds. For instance, earlier reports by Chen et al. utilized 1,1-dibromoolefins, a strategy that not only suffers from poor atom economy but also generates significant bromine-containing waste, posing environmental disposal challenges. Furthermore, these traditional routes often lack regioselectivity, failing to produce specific 2-aryl substituted products efficiently. Other methods, such as those reported by Li et al., require harsh reaction conditions, specifically temperatures as high as 110°C, and excessive amounts of promoters like cuprous chloride and ICy.HCl ligands (2 equivalents each), which drastically inflates raw material costs. Additionally, approaches involving 1,2-diiodoolefins necessitate nitrogen protection and complex operational procedures, further hindering their viability for large-scale industrial application.
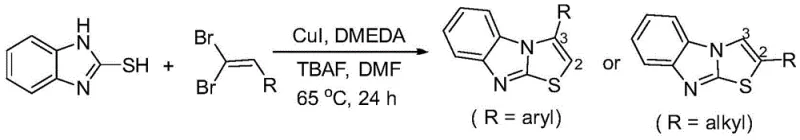
The Novel Approach
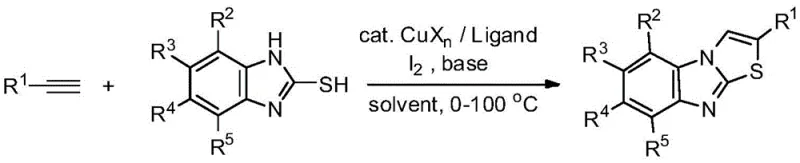
In stark contrast to these legacy methods, the novel protocol disclosed in CN109467564B utilizes simple, commercially available terminal alkynes as direct coupling partners. This eliminates the multi-step preparation of dibromo- or diiodo-olefins, thereby streamlining the supply chain and reducing the overall carbon footprint of the synthesis. The reaction proceeds efficiently at temperatures ranging from 0°C to 100°C, with an optimal window of 20°C to 60°C, significantly lowering energy consumption compared to the 110°C required by previous art. The use of a 1,10-phenanthroline derivative as a ligand, combined with elemental iodine and a base, creates a highly active catalytic system that ensures high conversion rates. This approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process by avoiding the handling of unstable poly-halogenated intermediates.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate interplay between the copper catalyst, the phenanthroline ligand, and the iodine oxidant. The mechanism likely involves the initial activation of the terminal alkyne by the copper species, forming a copper-acetylide intermediate. Simultaneously, the 2-mercaptobenzimidazole undergoes deprotonation by the base to generate a nucleophilic thiolate species. The presence of elemental iodine plays a crucial role in facilitating the oxidative cyclization, potentially generating an iodonium intermediate that activates the alkyne towards nucleophilic attack by the sulfur atom. This sequence leads to the formation of the thiazole ring fused to the benzimidazole core. The specific choice of the 1,10-phenanthroline derivative, particularly those with steric bulk like 2,9-diisopropyl-1,10-phenanthroline, stabilizes the copper center and prevents catalyst deactivation, ensuring sustained catalytic turnover throughout the reaction duration.
From an impurity control perspective, this mechanism offers distinct advantages. The high regioselectivity observed, favoring the 2-substituted product over the 3-substituted isomer, minimizes the formation of difficult-to-separate structural analogs. This is critical for pharmaceutical applications where impurity profiles must be strictly controlled to meet regulatory standards. The mild reaction conditions also suppress side reactions such as polymerization of the alkyne or decomposition of the sensitive benzimidazole ring. Consequently, the crude reaction mixture is cleaner, reducing the burden on downstream purification processes like column chromatography or recrystallization, which directly translates to higher overall recovery yields and reduced solvent waste in the final production stages.
How to Synthesize 2-Substituted Thiazolo[3,2-a]benzimidazole Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential. The patent outlines a robust procedure where terminal alkynes and 2-mercaptobenzimidazoles are combined in a molar ratio typically ranging from 1:0.2 to 1:5, with a preferred ratio of 1:1.3 for optimal balance between conversion and cost. The catalyst loading is remarkably low, with copper salts used at 0.01 to 0.5 equivalents, demonstrating the high efficiency of the system. The reaction is conducted in organic solvents such as acetonitrile, which has been identified as particularly effective, though a wide range of polar and non-polar solvents are compatible. Detailed standardized synthesis steps for specific derivatives are provided in the technical guide below, ensuring reproducibility across different laboratory and plant settings.
- Combine terminal alkyne, 2-mercaptobenzimidazole, copper salt catalyst (e.g., CuI), 1,10-phenanthroline ligand, iodine, and base in an organic solvent like acetonitrile.
- Heat the reaction mixture to a temperature between 20°C and 60°C, preferably 40°C, and stir for 1 to 24 hours, typically 17 hours.
- Upon completion, perform post-treatment via recrystallization or chromatography to isolate the high-purity 2-substituted product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary value driver is the elimination of pre-functionalized starting materials. By sourcing simple terminal alkynes instead of complex dibromoolefins or diiodoolefins, companies can leverage a broader and more competitive supplier base, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The stability and availability of terminal alkynes ensure a continuous supply flow, mitigating risks associated with the scarcity of specialized reagents. Furthermore, the simplified workflow reduces the number of unit operations required, directly lowering labor and utility costs associated with extended reaction times and complex workups.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the drastic simplification of the raw material list. Traditional methods require the separate synthesis and purification of halogenated olefins, which adds multiple steps and associated costs before the main cyclization even begins. By bypassing these precursor steps, the overall cost of goods sold (COGS) is substantially lowered. Additionally, the low catalyst loading and the ability to use inexpensive bases like potassium carbonate further contribute to margin improvement. The high atom economy means less waste is generated per kilogram of product, reducing waste disposal fees and aligning with sustainability goals without compromising financial performance.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals. Terminal alkynes and 2-mercaptobenzimidazoles are widely produced and stocked by global chemical suppliers, unlike the niche halogenated intermediates required by older methods. This ubiquity reduces lead time for high-purity pharmaceutical intermediates and minimizes the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate a variety of solvents and bases, provides flexibility in sourcing; if one solvent becomes unavailable or expensive, alternatives like toluene or ethanol can be substituted without re-validating the entire process, ensuring uninterrupted manufacturing continuity.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the mild thermal requirements and the absence of hazardous gas evolution or explosive intermediates. The reaction can be safely performed in standard stainless steel reactors without the need for specialized high-pressure or high-temperature equipment. From an environmental compliance standpoint, the reduction in halogenated waste streams simplifies effluent treatment. The process adheres to green chemistry principles by maximizing resource efficiency, making it easier for facilities to maintain their environmental permits and avoid regulatory penalties, thus securing the long-term viability of the production site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this technology for adoption. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What are the primary advantages of this copper-catalyzed method over prior art?
A: Unlike previous methods requiring pre-functionalized olefins (e.g., dibromoolefins) which generate halogen waste and have poor atom economy, this method uses readily available terminal alkynes directly. It operates under mild conditions (40°C) with high atom economy and avoids the need for expensive promoters used in older protocols.
Q: What is the substrate scope for the alkyne component?
A: The process demonstrates excellent versatility, accommodating various terminal alkynes including aryl alkynes (with electron-withdrawing or donating groups), heteroaryl alkynes (pyridyl, thienyl), and even alkyl alkynes. This broad scope allows for the synthesis of diverse drug-like scaffolds.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the reaction can be scaled up to gram-level and beyond while maintaining high efficiency and selectivity. The use of common solvents like acetonitrile and mild temperatures facilitates safer and more economical commercial scale-up compared to high-temperature alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Thiazolo[3,2-a]benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN109467564B for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle copper-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee product quality. We are committed to delivering reliable pharmaceutical intermediate supplier services that meet the exacting standards of the global market.
We invite you to collaborate with us to leverage this efficient synthesis for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom projects. Together, we can accelerate your timeline to market while optimizing your manufacturing economics.
