Revolutionizing Benzoate Ester Production: A Metal-Free Boron Catalysis Strategy for Commercial Scale-Up
Revolutionizing Benzoate Ester Production: A Metal-Free Boron Catalysis Strategy for Commercial Scale-Up
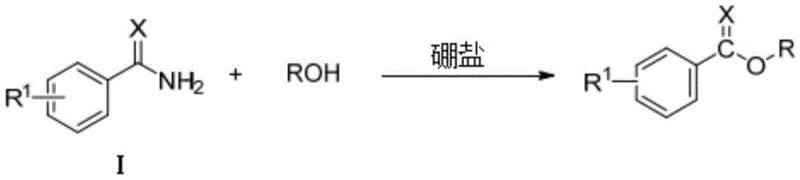
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN116143621A, which discloses a novel method for preparing benzoate compounds through boron salt-catalyzed activation of amides. This technology represents a substantial leap forward for the production of high-purity pharmaceutical intermediates, offering a robust alternative to traditional esterification routes that often rely on harsh conditions or toxic heavy metals. By leveraging the unique Lewis acidity of boron salts, specifically boron trifluoride etherate, this process enables the direct alcoholysis of benzamides under remarkably mild conditions. For R&D directors and procurement specialists alike, this innovation signals a pathway to reduce both environmental impact and production costs while maintaining exceptional product quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ester compounds, which are ubiquitous in pharmaceuticals, agrochemicals, and fragrances, has relied heavily on classical Fischer esterification or metal-catalyzed carbonylation strategies. While effective, these conventional approaches are fraught with significant industrial drawbacks that hinder scalable, green manufacturing. Traditional acid-catalyzed methods often require excessive amounts of strong mineral acids and high temperatures, leading to energy inefficiency and the generation of corrosive waste streams that demand costly neutralization and disposal protocols. Furthermore, modern transition-metal catalyzed methods, although powerful, introduce the persistent challenge of residual metal contamination. For pharmaceutical applications, removing trace amounts of catalysts like palladium, nickel, or copper to meet stringent ICH Q3D guidelines adds complex purification steps, such as scavenger resin treatment or repeated recrystallization, which inevitably erode overall yield and inflate production timelines. Additionally, many of these sensitive catalytic systems require strictly anhydrous and oxygen-free environments, necessitating expensive inert gas blanketing and specialized equipment that increases capital expenditure.
The Novel Approach
In stark contrast, the methodology outlined in patent CN116143621A introduces a paradigm-shifting approach that utilizes boron salts to activate the amide bond for nucleophilic attack by alcohols. This strategy effectively bypasses the limitations of legacy technologies by operating under air atmosphere at moderate temperatures, typically around 80°C, without the need for exotic ligands or moisture-sensitive setups. The core innovation lies in the ability of the boron catalyst to coordinate with the carbonyl oxygen of the benzamide, significantly lowering the activation energy required for the alcoholysis reaction. This allows for the use of simple alkyl alcohols not just as reactants but effectively as the reaction medium, promoting a solvent-free or near-solvent-free process that aligns perfectly with green chemistry principles. The result is a streamlined workflow that delivers target benzoate esters with exceptional yields, often exceeding 97%, while eliminating the logistical burdens associated with heavy metal handling and inert atmosphere maintenance.
Mechanistic Insights into Boron Salt-Catalyzed Amide Alcoholysis
To fully appreciate the technical superiority of this process, one must delve into the mechanistic nuances of how boron trifluoride etherate facilitates this transformation. The boron atom, possessing an empty p-orbital, acts as a potent Lewis acid that selectively coordinates with the lone pair electrons on the carbonyl oxygen of the benzamide substrate. This coordination withdraws electron density from the carbonyl carbon, rendering it highly electrophilic and thus far more susceptible to nucleophilic attack by the hydroxyl group of the alcohol. Unlike transition metal catalysts that might undergo complex oxidative addition and reductive elimination cycles prone to deactivation, the boron catalyst operates through a straightforward activation-deactivation cycle that is robust against air and moisture. This mechanistic simplicity translates directly to process reliability; there is no risk of catalyst poisoning by trace water or oxygen, which are common killers of palladium or rhodium-based systems. Consequently, the reaction proceeds smoothly to completion, minimizing the formation of side products such as hydrolysis-derived carboxylic acids or unreacted starting materials that complicate downstream purification.
Furthermore, the impurity profile generated by this boron-catalyzed route is inherently cleaner than that of metal-catalyzed alternatives. Since the catalyst is a main group element salt rather than a transition metal complex, the risk of generating organometallic byproducts or colored impurities is virtually non-existent. This is a critical advantage for the synthesis of high-purity pharmaceutical intermediates, where color and metal content are key quality attributes. The absence of heavy metals means that the crude reaction mixture can often be purified via simple distillation or basic chromatography, avoiding the need for specialized metal scavengers that add cost and waste. The patent data demonstrates this efficacy across a range of substrates, including the successful conversion of thioamides to thioesters, a transformation that is notoriously difficult with many other catalytic systems due to the strong affinity of sulfur for soft metal centers which leads to catalyst deactivation. Here, the hard Lewis acid character of boron avoids this pitfall, ensuring broad substrate tolerance.
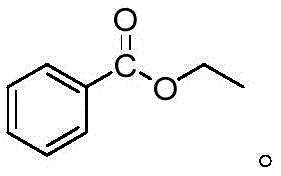
How to Synthesize Benzoate Esters Efficiently
Implementing this boron-catalyzed alcoholysis protocol in a laboratory or pilot plant setting is remarkably straightforward, requiring minimal specialized equipment beyond standard heating and stirring capabilities. The process begins with the charging of the benzamide substrate and the chosen alkyl alcohol into a reaction vessel, typically utilizing a molar excess of alcohol (e.g., 1:10 ratio) to drive the equilibrium towards the ester product. Following this, the boron trifluoride etherate catalyst is introduced, and the mixture is heated to approximately 80°C. Reaction progress can be easily monitored using thin-layer chromatography (TLC), with completion often achieved within a few hours depending on the specific steric hindrance of the alcohol used. The detailed standardized synthesis steps, including precise workup procedures and purification parameters for various derivatives like ethyl benzoate and propyl benzoate, are outlined in the technical guide below.
- Charge a reaction vessel with benzamide substrate and alkyl alcohol in a 1: 10 molar ratio under ambient air conditions.
- Add boron trifluoride etherate catalyst at a 2: 1 molar ratio relative to the benzamide substrate.
- Heat the mixture to 80°C and monitor via TLC until conversion is complete, followed by standard purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boron-catalyzed technology offers tangible strategic benefits that extend far beyond mere chemical curiosity. The primary value proposition lies in the drastic simplification of the supply chain for raw materials and the reduction of operational overheads. By eliminating the dependence on precious metal catalysts, manufacturers can insulate themselves from the volatile pricing fluctuations of commodities like palladium and platinum, which are subject to geopolitical instability and mining constraints. Moreover, the ability to run reactions in air without rigorous drying of solvents or substrates reduces the energy load on HVAC and utility systems, contributing to a lower carbon footprint and reduced utility costs. This operational resilience ensures a more stable and predictable production schedule, minimizing the risk of batch failures due to environmental excursions that frequently plague sensitive metal-catalyzed processes.
- Cost Reduction in Manufacturing: The economic implications of switching to this boron salt catalytic system are profound, primarily driven by the removal of expensive catalytic materials and the associated recovery infrastructure. Traditional methods often require dedicated units for metal recovery or the purchase of costly scavenger resins to meet regulatory limits, both of which represent significant line items in the cost of goods sold (COGS). In this new process, the catalyst is inexpensive and readily available, and the lack of heavy metals removes the need for these expensive downstream purification steps entirely. Additionally, the solvent-free nature of the reaction, where the alcohol serves as both reagent and medium, significantly reduces the volume of organic solvents required, leading to substantial savings in solvent procurement, storage, and waste disposal fees. These cumulative efficiencies result in a leaner manufacturing process that maximizes margin potential without compromising on product quality.
- Enhanced Supply Chain Reliability: From a logistics perspective, this methodology enhances supply chain robustness by utilizing commodity chemicals that are widely available from multiple global suppliers. Boron trifluoride etherate and simple alkyl alcohols are bulk chemicals with stable supply lines, unlike specialized ligands or organometallic complexes that may have single-source bottlenecks. The tolerance for air and moisture further simplifies storage and handling requirements, reducing the need for specialized containment or inert gas blanketing during transport and warehousing. This ease of handling translates to reduced lead times for raw material procurement and greater flexibility in production planning, allowing manufacturers to respond more agilely to market demand spikes. The high yields reported in the patent, often approaching quantitative levels, also mean that less starting material is required to produce a given amount of product, optimizing inventory turnover rates.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the inherent safety and simplicity of the reaction conditions. The absence of pyrophoric reagents or high-pressure hydrogenation steps lowers the safety risk profile, making it easier to obtain regulatory approvals for large-scale manufacturing facilities. Environmentally, the process aligns with increasingly strict global regulations regarding volatile organic compounds (VOCs) and heavy metal discharge. By minimizing solvent usage and eliminating toxic metal waste, manufacturers can significantly reduce their environmental liability and the costs associated with wastewater treatment and hazardous waste disposal. This green credential is increasingly becoming a prerequisite for doing business with major multinational pharmaceutical companies who are committed to sustainable supply chains, thereby opening up new market opportunities for suppliers who adopt this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-catalyzed esterification technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating its suitability for your specific production needs. Understanding these details is crucial for R&D teams assessing process feasibility and procurement teams evaluating vendor capabilities.
Q: What are the advantages of using boron salt catalysis over traditional metal catalysts for esterification?
A: Boron salt catalysis eliminates the need for expensive transition metals like palladium or nickel, thereby removing the risk of heavy metal contamination in the final API intermediate and simplifying the purification process significantly.
Q: Can this method be performed without inert gas protection?
A: Yes, the patented process operates effectively in an air environment, which drastically reduces operational complexity and nitrogen consumption compared to moisture-sensitive metal-catalyzed reactions.
Q: Is this method suitable for thioamide substrates?
A: Absolutely. The methodology demonstrates excellent substrate scope, successfully converting thioarylamides into thioesters with high efficiency, addressing a common limitation in conventional amide activation strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoate Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, green synthetic methodologies is not just a technical upgrade but a strategic imperative for modern chemical enterprises. Our team of expert process chemists has extensively evaluated the boron-catalyzed amide alcoholysis pathway described in CN116143621A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the lab are faithfully reproduced in our commercial reactors. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of benzoate ester we deliver meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific benzoate derivatives or scale-up of existing processes to improve cost-efficiency, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how adopting this boron-catalyzed method can optimize your supply chain and enhance your competitive edge in the global market.
