Scalable Synthesis of Potent 2-Indolone-Triazole Antitumor Intermediates for Global Pharma Supply Chains
The pharmaceutical landscape is continuously evolving towards more targeted and potent therapeutic agents, particularly in the realm of oncology. Patent CN111153889B, published in May 2022, introduces a significant advancement in the design of small-molecule tyrosine kinase inhibitors. This intellectual property discloses a novel class of 2-indolone-triazole antitumor compounds that exhibit strong inhibitory activity against the human VEGFR-2 enzyme. For R&D directors and procurement specialists in the global pharma sector, this represents a critical opportunity to access next-generation pharmaceutical intermediates with improved efficacy profiles. The patent outlines a robust synthetic methodology that not only achieves high yields but also simplifies the production process, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates. By integrating a 1,2,3-triazole structure into the classic 2-indolone core, the inventors have created a new chemical entity that addresses some of the limitations of earlier kinase inhibitors while maintaining a synthesis pathway that is feasible for industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional tyrosine kinase inhibitors, such as the well-known drugs Semaxanib, Sunitinib, and Toceranib, have established the 2-indolone scaffold as a privileged structure in anticancer therapy. However, these first and second-generation molecules often face challenges related to patent expiration, emerging drug resistance, and complex synthetic routes that rely on scarce or expensive reagents. 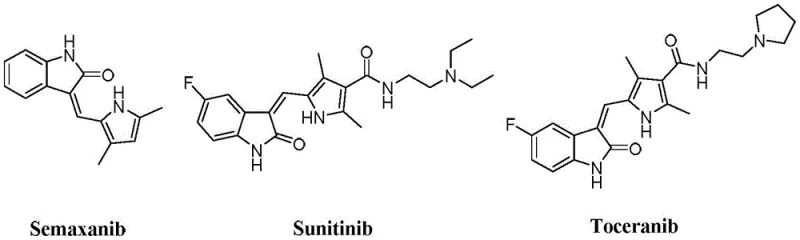 As shown in the reference structures, these molecules possess intricate side chains that can be difficult to functionalize efficiently on a multi-ton scale. Furthermore, the metabolic stability of some conventional indolones can be suboptimal, leading to rapid clearance in vivo and necessitating higher dosing frequencies. From a supply chain perspective, the reliance on specific, hard-to-source precursors for these older analogues can create bottlenecks, increasing lead times and volatility in the reliable pharmaceutical intermediate supplier market. The industry constantly seeks alternatives that offer better pharmacokinetic properties without compromising on manufacturability.
As shown in the reference structures, these molecules possess intricate side chains that can be difficult to functionalize efficiently on a multi-ton scale. Furthermore, the metabolic stability of some conventional indolones can be suboptimal, leading to rapid clearance in vivo and necessitating higher dosing frequencies. From a supply chain perspective, the reliance on specific, hard-to-source precursors for these older analogues can create bottlenecks, increasing lead times and volatility in the reliable pharmaceutical intermediate supplier market. The industry constantly seeks alternatives that offer better pharmacokinetic properties without compromising on manufacturability.
The Novel Approach
The innovation presented in CN111153889B overcomes these hurdles by strategically incorporating a 1,2,3-triazole ring into the molecular architecture. This modification is not merely cosmetic; the triazole ring serves as a stable linker that connects the indolone core to various substituted phenyl groups, allowing for fine-tuning of the electronic and steric properties of the drug. 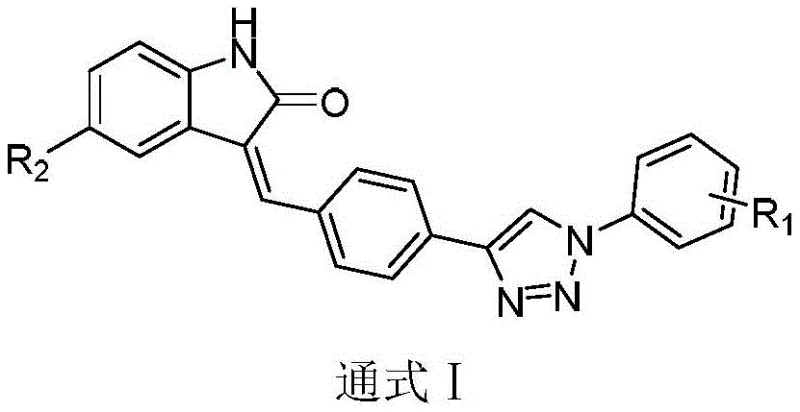 The general formula I depicted above illustrates the modularity of this approach, where R1 and R2 substituents can be varied to optimize potency against VEGFR-2. This structural flexibility enables medicinal chemists to rapidly generate libraries of analogues for SAR (Structure-Activity Relationship) studies. More importantly for production teams, the synthesis relies on convergent strategies rather than linear, step-heavy sequences. This means that key fragments can be prepared in parallel and joined together in the final stages, drastically reducing the overall cycle time and improving the throughput for high-purity pharmaceutical intermediates.
The general formula I depicted above illustrates the modularity of this approach, where R1 and R2 substituents can be varied to optimize potency against VEGFR-2. This structural flexibility enables medicinal chemists to rapidly generate libraries of analogues for SAR (Structure-Activity Relationship) studies. More importantly for production teams, the synthesis relies on convergent strategies rather than linear, step-heavy sequences. This means that key fragments can be prepared in parallel and joined together in the final stages, drastically reducing the overall cycle time and improving the throughput for high-purity pharmaceutical intermediates.
Mechanistic Insights into CuAAC Click Chemistry and Condensation
The core of this synthetic breakthrough lies in the utilization of Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC), widely known as "Click Chemistry." This reaction is renowned for its high efficiency, specificity, and tolerance to a wide range of functional groups. In the context of this patent, the formation of the 1,2,3-triazole ring is achieved by reacting a substituted azidobenzene with 4-ethynylbenzaldehyde. The mechanism involves the coordination of the alkyne to the copper(I) catalyst, which activates the triple bond towards nucleophilic attack by the azide. This results in the formation of a six-membered metallacycle intermediate that subsequently collapses to form the stable triazole ring. The beauty of this mechanism is its near-quantitative yield and the fact that it produces very few byproducts, which simplifies downstream purification significantly. For quality control teams, this translates to a cleaner impurity profile and less burden on analytical resources to identify and quantify trace contaminants.
Following the construction of the triazole-aldehyde intermediate, the final step involves a Claisen-Schmidt condensation reaction. This classic organic transformation couples the aldehyde functionality of the triazole intermediate with the active methylene group of the 2-indolone (or 5-fluoroindolin-2-one) core. Catalyzed by a mild base such as piperidine in ethanol, this reaction proceeds through an enolate intermediate that attacks the carbonyl carbon of the aldehyde. Subsequent dehydration yields the conjugated double bond system characteristic of the final product. This step is crucial as it establishes the planar geometry required for the molecule to fit into the ATP-binding pocket of the VEGFR-2 kinase. The use of ethanol as a solvent and piperidine as a catalyst ensures that the reaction conditions remain mild and environmentally benign, avoiding the need for harsh acids or toxic solvents that would complicate waste management and increase operational costs in a GMP facility.
How to Synthesize 2-Indolone-Triazole Efficiently
The synthesis protocol described in the patent is designed for operational simplicity, making it ideal for technology transfer from lab to plant. The process begins with the preparation of the azide component, followed by the click reaction to build the central scaffold, and concludes with the condensation to attach the pharmacophore. Each step has been optimized to maximize yield and minimize purification complexity. For detailed standard operating procedures and specific reaction parameters, please refer to the synthesis guide below.
- Preparation of substituted azidobenzene intermediates through diazotization of anilines followed by sodium azide substitution under controlled low-temperature conditions.
- Execution of a copper-catalyzed azide-alkyne cycloaddition (CuAAC) reaction between the azide and 4-ethynylbenzaldehyde to construct the 1,2,3-triazole core.
- Final Claisen-Schmidt condensation of the triazole-containing aldehyde with 2-indolone or 5-fluoroindolin-2-one to yield the target antitumor compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patent are profound. The shift towards this specific chemical architecture offers tangible benefits in terms of cost structure and supply security. By leveraging commodity chemicals and efficient catalytic cycles, manufacturers can achieve significant reductions in the cost of goods sold (COGS) without sacrificing quality. This aligns perfectly with the industry's ongoing drive for cost reduction in pharmaceutical intermediate manufacturing. The robustness of the synthetic route ensures that production schedules can be met consistently, mitigating the risks associated with supply disruptions that often plague more exotic or complex synthetic pathways.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive palladium or other precious metal catalysts often used in cross-coupling reactions. Instead, it utilizes inexpensive copper salts and vitamin C (ascorbic acid) as a reducing agent to maintain the active copper(I) species. This substitution dramatically lowers the raw material costs per kilogram. Furthermore, the high atom economy of the click reaction means that less waste is generated, reducing the costs associated with waste disposal and environmental compliance. The final condensation step uses ethanol, a green solvent that is cheap, recyclable, and easy to remove, further driving down processing expenses and enhancing the overall economic viability of the project.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as various substituted anilines, sodium azide, and 4-ethynylbenzaldehyde, are widely available commodity chemicals with established global supply chains. This availability reduces the risk of single-source dependency, a critical factor for reducing lead time for high-purity pharmaceutical intermediates. Because the intermediates are stable and the reaction conditions are not overly sensitive to moisture or air (especially the condensation step), the process is resilient to minor variations in plant conditions. This reliability allows for better inventory planning and ensures a steady flow of materials to downstream formulation partners, securing the continuity of the drug development pipeline.
- Scalability and Environmental Compliance: The reactions described operate under relatively mild temperatures and pressures, which simplifies the engineering requirements for scale-up. There is no need for specialized high-pressure reactors or cryogenic cooling systems beyond standard ice baths for the diazotization step. This ease of scale-up facilitates the transition from kilogram to multi-ton production scales seamlessly. Additionally, the avoidance of chlorinated solvents in the final steps and the use of aqueous workups where possible align with modern green chemistry principles. This reduces the environmental footprint of the manufacturing process, helping pharmaceutical companies meet their sustainability goals and regulatory obligations regarding solvent emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 2-indolone-triazole derivatives. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the biological target of these 2-indolone-triazole compounds?
A: These compounds are designed as potent inhibitors of the human VEGFR-2 enzyme, a critical target for blocking tumor angiogenesis and inhibiting cancer cell proliferation, particularly in lung cancer models like H460.
Q: How does the introduction of the triazole ring benefit the molecular structure?
A: The 1,2,3-triazole moiety acts as a robust bioisostere that enhances metabolic stability and improves binding affinity to the kinase domain, offering a structural advantage over traditional 2-indolone scaffolds found in earlier generation drugs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available commodity chemicals and avoids expensive transition metal catalysts, relying instead on simple copper salts and vitamin C, which significantly simplifies purification and supports cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Indolone-Triazole Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of having a partner who can translate complex patent chemistry into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering stringent purity specifications for every batch, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis for clinical trials or large-scale manufacturing for commercial launch, our infrastructure is designed to support your growth and mitigate supply chain risks effectively.
We invite you to collaborate with us to explore the full potential of this innovative antitumor scaffold. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your drug development timeline and secure a competitive advantage in the oncology market.
