Advanced Low-Temperature Nitration Strategy for High-Purity o-Nitro-p-tert-butylphenol Manufacturing
Introduction to Advanced Nitration Technology
The global demand for high-performance optical brighteners and specialized polymer additives continues to drive innovation in the synthesis of key phenolic intermediates. Patent CN101696169B introduces a transformative approach to producing o-nitro-p-tert-butylphenol, a critical building block for fluorescent whitening agents like OB and MN. This technology addresses long-standing inefficiencies in traditional nitration processes by utilizing a solvent-mediated, low-temperature reaction system that replaces hazardous mixed-acid protocols. For R&D directors and process engineers, this patent represents a significant leap forward in controlling regioselectivity and thermal safety during electrophilic aromatic substitution. By shifting from corrosive mixed acids to concentrated nitric acid in organic media, the process not only enhances yield but also fundamentally alters the safety profile of large-scale manufacturing. This report analyzes the technical merits and commercial implications of adopting this streamlined synthesis route for industrial applications.
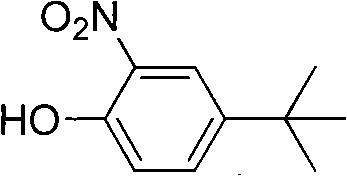
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of o-nitro-p-tert-butylphenol has been plagued by significant safety hazards and suboptimal yield profiles inherent to older nitration technologies. The first conventional method involves nitration with dilute nitric acid at room temperature, which suffers from poor selectivity due to the high susceptibility of the phenolic ring to oxidation. This lack of control results in a complex mixture of by-products, limiting the isolated yield to approximately 65% and necessitating extensive purification efforts that erode profit margins. The second traditional approach utilizes mixed acid (nitric and sulfuric acid), which, while more reactive, introduces severe safety liabilities. During the subsequent vacuum distillation required to isolate the product, the concentration of unstable polynitrated compounds increases in the still pot. This accumulation leads to heightened viscosity and localized overheating, creating a dangerous potential for explosive decomposition that poses unacceptable risks to personnel and infrastructure in a commercial plant setting.
The Novel Approach
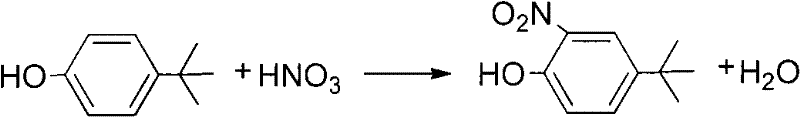
In stark contrast, the novel methodology disclosed in the patent employs concentrated nitric acid (69% mass fraction) within a controlled organic solvent environment, operating at a mild temperature range of 5°C to 20°C. This strategic shift eliminates the need for sulfuric acid, thereby removing the source of severe corrosion and the generation of vast quantities of acidic waste sludge. The presence of solvents such as ethanol, isopropanol, or acetic acid acts as a thermal buffer and reaction moderator, ensuring that the exothermic nitration proceeds smoothly without thermal runaway. By maintaining these low temperatures, the process effectively suppresses the formation of undesirable polynitrated species and oxidative degradation products. Consequently, this approach delivers a much cleaner reaction profile with yields consistently exceeding 85%, while the workup is simplified to solvent recovery and steam distillation, bypassing the hazardous high-temperature vacuum distillation steps of the past.
Mechanistic Insights into Solvent-Controlled Electrophilic Nitration
The core mechanistic advantage of this process lies in the modulation of the nitronium ion activity through solvent interaction and temperature control. In traditional mixed acid systems, the concentration of the active electrophile, the nitronium ion (NO2+), is extremely high, often leading to rapid, uncontrolled multiple substitutions on the activated phenolic ring. However, in the patented solvent system, the organic medium likely forms hydrogen bonds or solvation shells around the nitric acid species, effectively tempering the reactivity of the electrophile. This moderation allows for a more selective attack at the ortho-position relative to the hydroxyl group, favored by electronic activation, while the bulky tert-butyl group at the para-position sterically hinders substitution at that site. The low temperature regime (5-20°C) further kinetically favors the mono-nitration product by raising the activation energy barrier for secondary nitration events, ensuring that the reaction stops predominantly at the desired o-nitro stage.
Furthermore, the choice of solvent plays a pivotal role in impurity control and phase behavior during the reaction. Solvents like acetic acid or alcohols not only dissolve the starting p-tert-butylphenol effectively but also help dissipate the heat of reaction, preventing local hot spots that could trigger radical oxidation pathways. This homogeneous reaction environment ensures that the concentration of reactants remains uniform throughout the vessel, minimizing the formation of tarry residues often seen in heterogeneous mixed-acid nitrations. The result is a reaction mixture where the target o-nitro-p-tert-butylphenol is the dominant species, simplifying downstream isolation. The subsequent steam distillation step leverages the volatility differences between the product and non-volatile oxidative by-products, yielding a final material with purity specifications reaching 98% without the need for complex chromatographic separations or recrystallizations.
How to Synthesize o-Nitro-p-tert-butylphenol Efficiently
The implementation of this synthesis route requires precise control over addition rates and thermal management to maximize the benefits of the solvent system. The process begins by dissolving the p-tert-butylphenol starting material in a selected solvent, such as ethanol or acetic acid, ensuring a homogeneous solution before the introduction of the nitrating agent. Once the solution is cooled to the target range of 5°C to 20°C, concentrated nitric acid is added dropwise at a controlled rate to manage the exotherm. Following the completion of the addition, the reaction mixture is stirred for a defined period, typically 3 to 4 hours, to ensure full conversion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by experimental data, are outlined below.
- Dissolve p-tert-butylphenol in a suitable organic solvent such as ethanol, isopropanol, or acetic acid within a reaction vessel equipped with cooling capabilities.
- Maintain the reaction temperature between 5°C and 20°C while slowly adding concentrated nitric acid (69% mass fraction) dropwise to control the exothermic nitration reaction.
- After the reaction completes in 3-4 hours, remove the solvent via reduced pressure distillation and purify the crude product through steam distillation to isolate the target phenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of sulfuric acid from the process matrix drastically reduces the burden on wastewater treatment facilities, as there is no longer a need to neutralize large volumes of spent sulfuric acid or dispose of acid sludge. This reduction in hazardous waste generation translates directly into lower environmental compliance costs and reduced liability exposure for the manufacturing site. Additionally, the avoidance of high-risk vacuum distillation steps involving unstable polynitrates enhances overall plant safety, potentially lowering insurance premiums and minimizing the risk of production stoppages due to safety incidents. The robustness of the process also implies a more predictable production schedule, as the reaction is less sensitive to minor fluctuations in operating conditions compared to the volatile mixed-acid alternatives.
- Cost Reduction in Manufacturing: The transition to a single-acid system using concentrated nitric acid significantly streamlines the raw material supply chain by removing the requirement for purchasing and handling corrosive sulfuric acid. This simplification reduces storage infrastructure costs and minimizes the capital expenditure required for acid-resistant reactors and piping. Furthermore, the higher conversion efficiency means that less starting material is wasted on by-products, optimizing the atom economy of the process. The ability to recover and recycle the organic solvents used in the reaction further contributes to long-term operational cost savings, creating a more sustainable and economically viable manufacturing model for high-volume production.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents like ethanol and acetic acid ensures that the supply chain for raw materials remains resilient and less prone to geopolitical or logistical disruptions often associated with specialized reagents. The simplified workup procedure, which relies on standard distillation techniques rather than complex purification trains, allows for faster batch turnover times. This increased throughput capacity enables suppliers to respond more agilely to market demand spikes, ensuring consistent availability of this critical intermediate for downstream customers in the optical brightener and polymer industries.
- Scalability and Environmental Compliance: From a scale-up perspective, the low-temperature nature of this reaction makes it highly amenable to continuous flow processing or large-batch reactor operations without the fear of thermal runaway. The absence of heavy metal catalysts or toxic reagents aligns perfectly with modern green chemistry principles and increasingly stringent global environmental regulations. This compliance advantage facilitates easier permitting for new production lines and ensures long-term operational continuity in regions with strict environmental oversight, securing the supply chain against regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is the solvent-controlled nitration method safer than traditional mixed acid nitration?
A: Traditional mixed acid nitration often leads to the accumulation of polynitrated by-products which increase viscosity and pose severe explosion risks during vacuum distillation. The patented solvent-controlled method operates at low temperatures (5-20°C) and avoids sulfuric acid, significantly reducing thermal runaway risks and equipment corrosion.
Q: What represents the primary cost advantage of this synthesis route for procurement teams?
A: The primary cost advantage lies in the elimination of mixed acid waste treatment and the simplification of the purification process. By avoiding sulfuric acid and achieving higher conversion rates with concentrated nitric acid alone, manufacturers save substantially on raw material consumption and environmental compliance costs associated with acidic wastewater neutralization.
Q: How does this method impact the purity profile required for optical brightener synthesis?
A: This method achieves product purity levels exceeding 98% directly after steam distillation. The controlled low-temperature environment minimizes oxidative side reactions common in room-temperature dilute acid methods, ensuring a cleaner impurity profile that is critical for downstream fluorescence applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Nitro-p-tert-butylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering o-nitro-p-tert-butylphenol with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for optical brightener synthesis. Our facility is equipped to handle the specific solvent recovery and low-temperature processing requirements of this advanced nitration method, guaranteeing a consistent and reliable supply.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your supply chain.
