Advanced Manufacturing of Optically Pure Tetrahydroprotoberberine Intermediates for Neurological Drug Development
The pharmaceutical landscape for neurological therapeutics is constantly evolving, driven by the need for high-purity active ingredients with specific stereochemical configurations. Patent CN102399166A introduces a groundbreaking methodology for the preparation of optically isomerized stepheridine and its derivatives, addressing critical bottlenecks in the supply of tetrahydroprotoberberine (THPBs) compounds. These compounds, known for their potent dopamine receptor antagonistic activity, are pivotal in treating conditions such as schizophrenia and migraines. However, historical reliance on natural extraction has resulted in supply chain fragility due to limited plant resources, while previous synthetic routes struggled with racemic mixtures and complex purification. This technical insight analyzes the novel synthetic pathway disclosed in the patent, highlighting its potential to revolutionize the manufacturing of high-purity pharmaceutical intermediates for global drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroprotoberberine scaffolds has been plagued by significant regioselectivity and stereochemical challenges. Traditional methods utilizing the Mannich reaction often suffer from poor positional control, resulting in 40% to 60% of the product being the undesired 11-position substituted isomer rather than the target structure. Furthermore, these classical routes are chemically restricted; they are generally only applicable when the 9-position is a hydroxyl group, failing completely when a methoxy or other alkoxy group is required at this position. Alternative approaches involving chiral metal catalysts for asymmetric synthesis have been reported, but these are economically prohibitive for large-scale manufacturing due to the exorbitant cost of the catalysts and the苛刻 (harsh) reaction conditions required. Additionally, such methods are typically confined to milligram-scale laboratory preparations, lacking the robustness needed for commercial kilogram-level production, and often involve lengthy synthetic sequences with cumulative low yields.
The Novel Approach
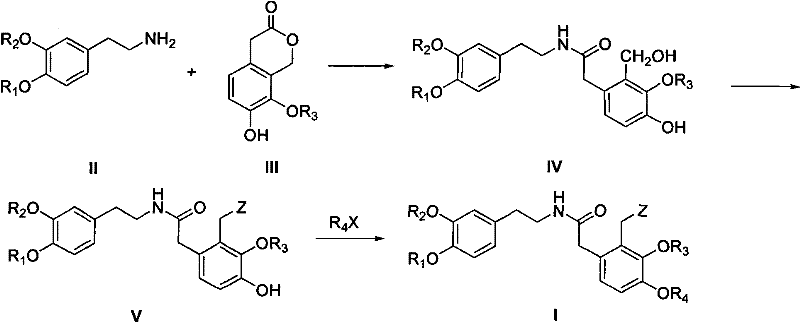
The methodology outlined in patent CN102399166A circumvents these historical impediments by employing a unique intermediate strategy centered on general Formula (I). This approach utilizes a Bischler-Napieralski cyclization followed by a resolution strategy rather than direct asymmetric synthesis, which drastically simplifies the operational complexity. By constructing the core skeleton through a protected amide intermediate, the process eliminates the regioselectivity issues associated with the Mannich reaction, ensuring that no 11-position substituted by-products are generated. This results in a much cleaner reaction profile and significantly higher crude purity. Moreover, the route is versatile enough to accommodate 9-alkoxy substitutions, expanding the chemical space available for derivative development. The shift from expensive chiral catalysis to a resolution-based purification using common chiral acids represents a paradigm shift in cost-efficiency and scalability for this class of compounds.
Mechanistic Insights into Bischler-Napieralski Cyclization and Chiral Resolution
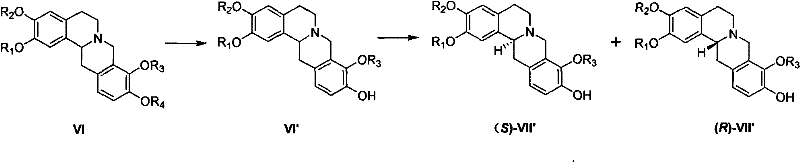
The core of this synthetic innovation lies in the precise execution of the ring-closing steps and the subsequent separation of enantiomers. The process begins with the formation of the key intermediate (Formula I) through aminolysis and halogenation, setting the stage for cyclization. The Bischler-Napieralski reaction is then employed using reagents such as phosphorus oxychloride (POCl3) or phosphorus oxybromide (POBr3) in solvents like acetonitrile or toluene. This step generates an imine or iminium salt intermediate (Formula X or XI), which is subsequently reduced using metal borohydrides such as sodium borohydride (NaBH4) or potassium borohydride (KBH4). The reduction is carefully controlled, often at temperatures ranging from -20°C to reflux, to ensure the formation of the tetrahydroprotoberberine skeleton (Formula VI) without over-reduction or side reactions. Following the ring closure, which can be facilitated by bases like sodium hydroxide or potassium carbonate, the resulting racemic mixture undergoes optical resolution.
The resolution mechanism exploits the solubility differences between diastereomeric salts formed with chiral resolving agents. By reacting the racemic base (Formula VI) with acidic resolving agents such as L-tartaric acid, D-dibenzoyltartaric acid, or L-mandelic acid in solvents like methanol or ethanol, distinct crystalline salts are formed. These salts, having different physical properties, can be separated via filtration. The patent data indicates that through iterative recrystallization, the optical purity can be elevated from initial values around 90% to exceed 98%. This is a critical quality attribute for pharmaceutical intermediates, as the biological activity of THPBs is highly stereospecific, with the levorotatory isomer (l-SPD) typically exhibiting the desired therapeutic effects while the dextrorotatory form may be inactive or possess different pharmacokinetics. The ability to access both (S) and (R) enantiomers efficiently allows for comprehensive biological screening and the production of single-enantiomer drugs.
How to Synthesize Optically Pure Stepheridine Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression from commercially available starting materials to the final chiral product. The process integrates standard unit operations such as reflux, crystallization, and filtration, making it highly adaptable to existing chemical manufacturing infrastructure. The detailed标准化 (standardized) synthetic steps involve the initial coupling of substituted phenethylamines with chroman-3-ones, followed by functional group manipulation to enable cyclization. The critical resolution step requires careful control of solvent composition and temperature to maximize the yield of the desired enantiomer while maintaining high optical purity. For a complete breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this high-yielding process, please refer to the technical guide below.
- Prepare the key amide intermediate (Formula I) via aminolysis of substituted phenethylamine and chroman-3-one, followed by halogenation and hydroxyl protection.
- Execute Bischler-Napieralski cyclization using phosphorus oxychloride, followed by reduction with borohydrides and base-catalyzed ring closure to form the tetrahydroprotoberberine skeleton.
- Perform optical resolution using chiral acids like tartaric acid derivatives to separate diastereomeric salts, yielding optically pure (S)- and (R)-enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of rare and expensive chiral metal catalysts removes a significant cost driver and supply risk from the manufacturing equation. Traditional asymmetric synthesis often relies on precious metals that are subject to volatile market pricing and geopolitical supply constraints. By replacing these with abundant organic acids and standard inorganic reagents, the cost structure of the final intermediate is substantially optimized. Furthermore, the robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—reduces the need for specialized high-pressure equipment, thereby lowering capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The replacement of costly chiral catalysts with conventional resolving agents like tartaric acid derivatives leads to a drastic reduction in raw material costs. Additionally, the high regioselectivity of the new route minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification processes such as column chromatography. This streamlining of the workflow translates directly into lower processing costs and higher overall throughput, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted phenethylamines and chroman-3-ones, are commercially available or easily synthesized from bulk chemicals. This ensures a stable and diversified supply base, mitigating the risks associated with single-source dependencies. The scalability of the process, demonstrated in the patent from gram to multi-hundred-gram scales, indicates a clear path to tonnage production without the need for process re-engineering, ensuring consistent delivery timelines for long-term contracts.
- Scalability and Environmental Compliance: The use of common solvents like ethanol, methanol, and ethyl acetate simplifies waste management and solvent recovery systems. Unlike processes requiring chlorinated solvents in large volumes or heavy metal waste streams, this route aligns better with modern green chemistry principles. The simplified workup procedures, often involving simple filtration and crystallization rather than complex extractions, reduce the generation of hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the integration of this route into their existing pipelines and for quality assurance teams establishing specification limits.
Q: How does this method improve upon traditional Mannich reactions for THPBs synthesis?
A: Traditional Mannich reactions often produce 40-60% of unwanted 11-position substituted isomers and are limited to 9-OH compounds. This novel route avoids regioselectivity issues entirely, allowing for the synthesis of 9-alkoxy substituted derivatives with high purity and no positional isomers.
Q: What is the optical purity achievable with this resolution method?
A: Through recrystallization of diastereomeric salts formed with resolving agents like L-tartaric acid or D-dibenzoyltartaric acid, the process achieves optical purity levels exceeding 98%, suitable for pharmaceutical applications.
Q: Is this process scalable for industrial production?
A: Yes, the patent demonstrates successful synthesis at gram to kilogram scales (e.g., 200g batches in examples) using conventional reagents and solvents, avoiding expensive chiral metal catalysts that limit scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stepheridine Intermediate Supplier
As the demand for neurological therapeutics grows, securing a supply of high-purity, optically active intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to ensure optical purity exceeds 98% as required for clinical applications. We understand the critical nature of supply continuity in the pharmaceutical industry and have optimized our processes to deliver consistent quality batch after batch.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring your next-generation neurological drugs reach the market faster and more cost-effectively.
