Revolutionizing Isonitrile Production: A Safe, One-Step Difluorocarbene Strategy for Commercial Scale
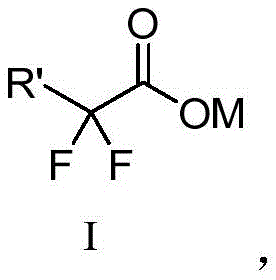
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and materials science, particularly in the realm of isonitrile chemistry. Patent CN112279789B introduces a transformative methodology for the preparation of isonitrile compounds, addressing long-standing challenges regarding safety, efficiency, and environmental impact. This innovation leverages sodium difluorochloroacetate or potassium difluorobromoacetate as a precise difluoromethyl source, enabling the direct conversion of primary amines into target isonitriles through a streamlined one-step condensation. For R&D directors and process chemists, this represents a significant departure from hazardous traditional routes, offering a robust platform for generating high-purity intermediates essential for Ugi multicomponent reactions and polymer construction. The technical breakthrough lies in the in-situ generation of difluorocarbene, which reacts selectively with the amine nitrogen, thereby bypassing the need for pre-synthesized formamides and toxic dehydrating agents.
From a commercial perspective, the implications of this technology extend far beyond the laboratory bench, offering substantial value to procurement and supply chain stakeholders. The reliance on simple, widely available industrial commodities as starting materials ensures a stable supply chain, mitigating the risks associated with sourcing specialized or regulated reagents. Furthermore, the operational simplicity of the process—requiring standard heating and filtration equipment without the need for cryogenic conditions or exotic catalysts—translates directly into reduced capital expenditure and lower operational costs. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is crucial for evaluating the long-term viability and cost-effectiveness of integrating this synthetic route into large-scale production pipelines for complex drug candidates and advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isonitrile compounds has been plagued by significant safety hazards and operational inefficiencies that hinder large-scale adoption. The most predominant conventional method involves the dehydration of formamides, a process that necessitates the prior synthesis and isolation of the formamide intermediate, often requiring multiple reaction steps that degrade overall atom economy. More critically, the dehydration step itself typically employs excessive amounts of highly toxic and corrosive reagents such as phosgene, phosphorus trichloride, or sulfonyl chlorides. These substances pose severe health risks to personnel and require specialized containment infrastructure, driving up both capital and operational expenditures. Additionally, the reaction mixtures resulting from these dehydration protocols are often complex and difficult to separate, leading to challenging purification processes that reduce final yields and increase waste generation. Another traditional approach, the Hofmann-type degradation of primary amines using chloroform and strong bases like sodium hydroxide, suffers from poor functional group tolerance due to the harsh alkaline conditions, frequently resulting in unwanted side reactions and limited substrate scope.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a difluorocarbene precursor strategy that fundamentally simplifies the synthetic pathway while enhancing safety profiles. By employing sodium difluorochloroacetate or potassium difluorobromoacetate, the process generates the reactive carbene species in situ under relatively mild thermal conditions, eliminating the need for handling gaseous phosgene or isolating unstable intermediates. This one-step condensation with primary amines proceeds efficiently in common polar aprotic solvents such as DMF or DMSO, using inexpensive inorganic bases like potassium carbonate. The result is a cleaner reaction profile that facilitates easier downstream processing, typically requiring only standard extraction and chromatography to achieve high purity. This methodological shift not only improves the safety of the manufacturing environment by removing toxic reagents but also enhances the economic feasibility of producing isonitrile derivatives, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Difluorocarbene-Mediated Isonitrile Formation
The core chemical innovation of this process revolves around the controlled generation and insertion of difluorocarbene (:CF2) into the nitrogen-hydrogen bond of the primary amine. Under the influence of a base at elevated temperatures (typically 80-130°C), the difluorohaloacetate salt undergoes decarboxylation and elimination of the halide ion to release the reactive difluorocarbene species. This electrophilic carbene then attacks the nucleophilic nitrogen of the primary amine, forming an intermediate that subsequently rearranges or eliminates hydrogen fluoride to yield the stable isonitrile functionality (-NC). This mechanism is distinct from the nucleophilic substitution pathways seen in other halogenated reactions, offering a unique route to the isocyanide motif that preserves the integrity of sensitive functional groups on the aromatic or aliphatic backbone of the amine substrate. The use of difluoroacetates acts as a solid, easy-to-handle "carbene equivalent," providing a safer alternative to generating carbenes from diazo compounds or other hazardous precursors.
Impurity control in this system is inherently superior due to the specificity of the carbene insertion and the mildness of the basic conditions employed. Unlike the strong base conditions of the Hofmann degradation which can promote elimination reactions or hydrolysis of sensitive esters and amides, the carbonate or bicarbonate bases used here maintain a pH environment that favors the desired transformation while suppressing competitive degradation pathways. Furthermore, the byproducts of the reaction, primarily carbon dioxide and inorganic salts, are easily removed during the aqueous workup phase, leaving the organic layer enriched with the target isonitrile. This clean impurity profile is critical for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals are enforced, ensuring that the final high-purity isonitrile compounds meet rigorous regulatory standards without the need for extensive and yield-eroding purification steps.
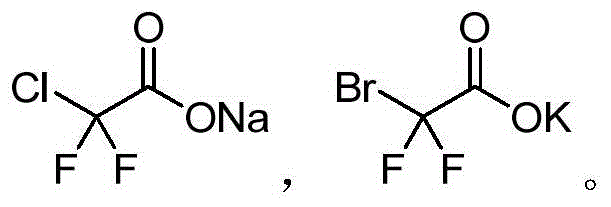
How to Synthesize Isonitrile Compounds Efficiently
The practical execution of this synthesis is designed for straightforward implementation in standard chemical manufacturing facilities, requiring no specialized high-pressure or cryogenic equipment. The protocol involves charging a reaction vessel with the difluorohaloacetate salt, the primary amine substrate, and a stoichiometric amount of base, followed by the addition of a polar aprotic solvent under an inert nitrogen atmosphere to prevent moisture interference. The mixture is then heated to the optimal temperature range, allowing the reaction to proceed to completion over a period of 8 to 16 hours, after which the product is isolated through a simple workup involving dilution, extraction, and drying. This operational simplicity makes the technology highly accessible for process chemists looking to optimize their workflows, and the detailed standardized synthesis steps provided below offer a clear roadmap for replicating these results in a pilot or production setting.
- Charge a reaction vessel with sodium difluorochloroacetate, a suitable inorganic or organic base (such as K2CO3), and the primary amine substrate under an inert atmosphere.
- Inject anhydrous polar aprotic solvent (e.g., DMF) and heat the mixture to 80-130°C for 8-16 hours to facilitate in-situ difluorocarbene generation and insertion.
- Cool the reaction to room temperature, dilute with water and dichloromethane, separate the organic layer, and purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling strategic advantages centered around risk mitigation and cost efficiency. The primary driver for cost optimization is the elimination of expensive and hazardous reagents; by replacing phosgene and phosphorus chlorides with stable acetate salts, the process removes the need for costly safety infrastructure, specialized storage tanks, and complex waste treatment systems associated with toxic gas handling. This shift significantly lowers the barrier to entry for manufacturing isonitriles and reduces the overall cost of goods sold (COGS) by streamlining the raw material portfolio to include only common, bulk-available chemicals. Moreover, the simplified workup procedure reduces solvent consumption and processing time, further contributing to operational savings without compromising on the quality or yield of the final product.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the synthetic route and the substitution of high-cost, high-risk reagents with commodity chemicals. By utilizing sodium difluorochloroacetate, which is an industrial commodity with a stable price point, manufacturers can avoid the volatile pricing and supply constraints often associated with specialized dehydrating agents. The one-step nature of the reaction also reduces labor costs and energy consumption by eliminating intermediate isolation steps, leading to a more lean and efficient production cycle that maximizes throughput while minimizing resource utilization.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of raw materials that are widely produced and globally available. Unlike niche reagents that may be subject to single-source bottlenecks or geopolitical trade restrictions, the key components of this reaction—difluoroacetates, carbonates, and common solvents—are part of the standard inventory for most fine chemical suppliers. This ubiquity ensures a continuous and reliable flow of materials, reducing the risk of production stoppages due to raw material shortages and allowing for more accurate forecasting and inventory management strategies.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than traditional methods, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. The absence of halogenated waste streams from dehydrating agents and the generation of benign inorganic byproducts simplify the effluent treatment process, reducing the cost and complexity of environmental compliance. Furthermore, the robustness of the reaction conditions allows for seamless scale-up from gram-scale laboratory synthesis to multi-ton commercial production, ensuring that the process remains efficient and controllable regardless of the batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isonitrile synthesis technology, drawing directly from the experimental data and scope defined in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this method for their specific manufacturing needs. Understanding these details is essential for assessing the feasibility of adopting this route for your specific product portfolio.
Q: What are the primary safety advantages of this isonitrile synthesis method compared to traditional dehydration routes?
A: Unlike conventional methods that require highly toxic dehydrating agents like phosgene or phosphorus oxychloride, this patented process utilizes stable, commercially available difluoroacetate salts. This eliminates the need for handling hazardous gases and significantly reduces the risk profile associated with large-scale manufacturing.
Q: Can this method accommodate primary amines with sensitive functional groups?
A: Yes, the reaction conditions are relatively mild compared to strong base-promoted Hofmann degradations. The use of moderate bases like potassium carbonate in polar aprotic solvents allows for better compatibility with various substituents, including ethers and halogens, minimizing side reactions and improving overall yield.
Q: Is this process suitable for industrial scale-up and continuous production?
A: The process is designed for scalability, utilizing simple unit operations such as heating, filtration, and extraction. The raw materials are industrial commodities with wide availability, and the absence of specialized storage requirements for reagents facilitates seamless integration into existing commercial manufacturing lines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isonitrile Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient and safe synthetic methodologies play in the development of next-generation pharmaceuticals and advanced materials. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the difluorocarbene insertion method can be successfully translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex fluorinated intermediates positions us as a strategic partner for companies seeking to secure their supply chains for high-value isonitrile derivatives.
We invite you to collaborate with us to explore how this advanced synthesis technology can be integrated into your production workflow to drive efficiency and reduce costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial goals, ensuring a reliable supply of high-quality intermediates for your most critical projects.
