Advanced Manufacturing of C-Glucoside Derivative Impurities for Global Pharmaceutical Compliance
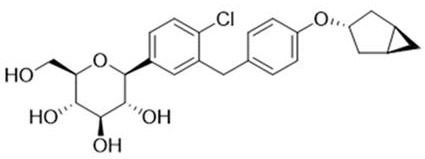
The pharmaceutical industry's relentless pursuit of safer and more effective antidiabetic medications has placed SGLT-2 inhibitors at the forefront of therapeutic innovation. However, the clinical success of these C-glucoside derivatives relies heavily on rigorous impurity profiling to ensure patient safety and regulatory compliance. Patent CN113372315B introduces a groundbreaking methodology for the scalable synthesis of a specific C-glucoside impurity, designated as Formula (I), which is critical for the quality control of next-generation diabetes treatments. This technical breakthrough addresses the historical bottleneck of producing sufficient quantities of reference standards, enabling pharmaceutical manufacturers to meet stringent pharmacopoeial requirements with greater efficiency. By establishing a robust synthetic route, this patent empowers global supply chains to maintain the highest standards of drug purity and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex C-glycoside impurities has been plagued by low yields, cumbersome purification steps, and an inability to scale beyond milligram quantities. Traditional routes often rely on non-selective glycosylation or harsh deprotection conditions that degrade the sensitive sugar moiety, leading to a complex mixture of byproducts that are difficult to separate. Furthermore, existing methods frequently fail to provide the structural fidelity required for accurate analytical method validation, forcing quality control laboratories to rely on scarce and expensive imported reference materials. The lack of a dedicated, scalable process for these specific impurities has created a significant vulnerability in the supply chain for SGLT-2 inhibitor manufacturers, delaying drug approval processes and increasing the cost of goods sold due to inefficient sourcing strategies.
The Novel Approach
The methodology disclosed in Patent CN113372315B represents a paradigm shift by utilizing a convergent synthetic strategy that prioritizes both scalability and selectivity. Instead of attempting to isolate the impurity as a minor byproduct of the main drug synthesis, this approach constructs the impurity directly from a protected precursor through a series of highly controlled transformations. The process leverages specific Lewis acid catalysis and mild etherification conditions to preserve the stereochemical integrity of the glucoside core while introducing the necessary structural modifications. This targeted synthesis not only ensures a consistent supply of high-purity reference standards but also drastically simplifies the downstream purification workflow. By solving the problem of large-scale production required for 10g grade reference substances, this novel approach provides a reliable foundation for comprehensive impurity analysis and quality control.

Mechanistic Insights into Lewis Acid-Mediated Ether Cleavage and Methylene Bridging
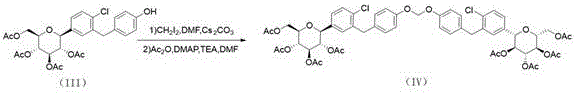
The core of this synthetic innovation lies in the precise manipulation of protecting groups and the formation of the methylene bridge, which defines the unique structure of the impurity. The first critical step involves the use of aluminum chloride (AlCl3) in conjunction with 1-dodecyl mercaptan in dichloromethane. This Lewis acid-mediated reaction facilitates the selective cleavage of specific ether bonds without compromising the stability of the glycosidic linkage. The mechanism likely proceeds through the coordination of the aluminum species to the oxygen atoms, increasing their electrophilicity and allowing the thiol to act as a nucleophile or scavenger, effectively removing the protecting group to reveal the reactive phenolic or alcoholic sites necessary for the subsequent coupling. This step is performed at controlled temperatures between 10-15°C during addition and 25°C during reaction to minimize side reactions and ensure high conversion rates.
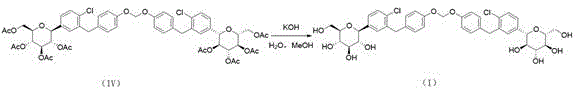
Following the deprotection, the synthesis advances to a sophisticated methylene bridging reaction using diiodomethane and cesium carbonate in dimethylformamide (DMF). This step is pivotal for constructing the characteristic diarylmethane or ether linkage found in the impurity structure. The use of cesium carbonate as a base is particularly advantageous due to its high solubility in organic solvents and its ability to promote SN2-type substitutions efficiently. The reaction is conducted under a nitrogen atmosphere at elevated temperatures of 85-95°C to overcome the activation energy barrier for the formation of the carbon-oxygen or carbon-carbon bond. Subsequent acetylation with acetic anhydride, triethylamine, and DMAP protects the newly formed hydroxyl groups, stabilizing the intermediate for the final hydrolysis step. This multi-step sequence demonstrates a deep understanding of carbohydrate chemistry and protective group orthogonality.
How to Synthesize C-Glucoside Impurity Efficiently
The synthesis of this high-value impurity requires strict adherence to the optimized reaction parameters outlined in the patent to ensure reproducibility and high purity. The process begins with the preparation of Intermediate 1, followed by the crucial bridging and acetylation steps to form Intermediate 2, and concludes with a global deprotection to yield the final target. Each stage demands precise control over stoichiometry, temperature, and workup procedures to maximize yield and minimize the formation of regioisomers. For detailed operational protocols, including specific solvent volumes, stirring rates, and crystallization techniques, please refer to the standardized guide below which encapsulates the industrial best practices derived from this patent.
- Perform Lewis acid-mediated ether cleavage using AlCl3 and 1-dodecyl mercaptan in dichloromethane to generate Intermediate 1.
- Execute methylene bridging with diiodomethane and Cs2CO3 in DMF, followed by acetylation to form Intermediate 2.
- Conduct final hydrolysis using aqueous KOH in methanol to deprotect acetyl groups and isolate the target impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers substantial strategic benefits beyond mere technical feasibility. The ability to produce reference standards in 10g batches internally or through a specialized partner eliminates the dependency on unpredictable external suppliers who may struggle with the complexity of C-glycoside chemistry. This vertical integration capability significantly reduces lead times for critical quality control materials, ensuring that API manufacturing lines are never halted due to a lack of validated testing standards. Moreover, the use of commercially available raw materials such as aluminum chloride, cesium carbonate, and common solvents like dichloromethane and DMF ensures that the supply chain remains resilient against raw material shortages. The streamlined nature of the three-step process also implies a reduction in overall processing time and labor costs, contributing to a more lean and efficient operation.
- Cost Reduction in Manufacturing: The elimination of complex, low-yielding isolation steps traditionally associated with impurity synthesis leads to a significant optimization of production costs. By utilizing a direct synthetic route with defined intermediates, the process minimizes waste generation and solvent consumption, which are major cost drivers in fine chemical manufacturing. The high selectivity of the Lewis acid cleavage and the efficient methylene bridging reaction reduce the need for extensive chromatographic purification, allowing for simpler crystallization or precipitation techniques that are far more economical at scale. This efficiency translates directly into a lower cost of goods for the reference standards, freeing up budget for other critical R&D initiatives.
- Enhanced Supply Chain Reliability: Securing a stable source of high-purity impurities is essential for maintaining uninterrupted API production and regulatory compliance. This method provides a robust framework for scaling production from gram to kilogram levels as demand for SGLT-2 inhibitors grows globally. The reliance on standard industrial reagents and equipment means that the process can be easily transferred to multiple manufacturing sites, mitigating the risk of single-source supply disruptions. Furthermore, the predictable reaction outcomes ensure consistent quality batch after batch, reducing the variability that often plagues complex organic syntheses and strengthening the overall reliability of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are readily adaptable to large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The use of aqueous workups and standard solvent systems facilitates effective waste management and recycling, aligning with modern green chemistry principles. By avoiding the use of heavy metal catalysts or highly toxic reagents, the synthesis profile is environmentally friendlier, simplifying the regulatory burden associated with waste disposal and worker safety. This alignment with environmental, social, and governance (ESG) goals enhances the corporate reputation of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams planning to integrate this impurity standard into their quality control workflows and for procurement specialists evaluating the long-term viability of the supply source. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: What is the primary application of the compound synthesized in Patent CN113372315B?
A: The compound serves as a critical reference standard (impurity) for quality control and safety assessment of C-glucoside derivative drugs, specifically SGLT-2 inhibitors used in treating type 2 diabetes.
Q: How does this synthesis method improve upon previous techniques for impurity production?
A: The patented method addresses the challenge of large-scale production (10g grade) required for reference substances, offering improved reaction yields and a more streamlined purification process compared to prior art.
Q: What are the key reaction conditions for the methylene bridging step?
A: The methylene bridging step utilizes diiodomethane and cesium carbonate in DMF under nitrogen protection, heated to 85-95°C, ensuring efficient formation of the ether linkage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-Glucoside Impurity Supplier
As the global demand for SGLT-2 inhibitors continues to surge, the need for high-quality, compliant impurity standards has never been more critical. NINGBO INNO PHARMCHEM stands ready to support your pharmaceutical development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the nuanced chemistry of C-glucoside derivatives, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that the integrity of your final drug product depends on the accuracy of your reference materials, and we are committed to delivering excellence in every gram we produce.
We invite you to collaborate with us to optimize your supply chain and accelerate your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for immediate validation or comprehensive route feasibility assessments for future projects, our experts are here to provide the insights and solutions you need to succeed in the competitive landscape of antidiabetic drug manufacturing.
