Revolutionizing Catalytic Synthesis: Scalable N-Heterocyclic Carbene Copper Production for Pharmaceutical Intermediate Manufacturing
The patent CN112694489B, granted on July 4, 2023, introduces a transformative preparation method for N-heterocyclic carbene copper catalysts (Cu-NHCs), directly addressing critical limitations in existing synthetic routes including operational complexity, hazardous waste generation, high costs associated with silver reagents, and susceptibility to oxidation of conventional copper sources like CuO or CuCl₂. This innovative approach leverages the direct reaction between imidazolium salts and basic copper carbonate under precisely controlled thermal conditions, establishing a streamlined pathway that eliminates the need for strong bases or expensive transition metal precursors while producing only environmentally benign byproducts such as carbon dioxide and water. The methodology demonstrates exceptional scalability across production volumes from laboratory-scale trials to industrial manufacturing runs exceeding one hundred metric tons annually, with reaction parameters optimized within narrow temperature ranges (50–80°C) and molar ratios (1:1.5) to ensure maximum yield consistency and purity compliance. Crucially, this patent enables reliable manufacturing of high-purity Cu-NHC catalysts essential for advanced applications in pharmaceutical intermediate synthesis where stringent quality standards and uninterrupted supply chains are non-negotiable requirements for global enterprises operating under regulatory frameworks like ICH Q7 guidelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for N-heterocyclic carbene copper compounds suffer from multiple critical deficiencies that impede commercial adoption in pharmaceutical manufacturing environments; these include mandatory use of expensive silver reagents which introduce significant cost burdens while generating difficult-to-treat halide waste streams requiring specialized disposal protocols; reliance on strong bases that necessitate additional neutralization steps and produce stoichiometric inorganic salt byproducts contaminating final products; susceptibility of monovalent copper sources like Cu₂O to atmospheric oxidation which compromises batch consistency; and operational complexities involving multi-step procedures with stringent anhydrous conditions that increase both capital expenditure and process validation requirements. Furthermore, conventional methods often require chromatographic purification due to side reactions like ligand decomposition or metal disproportionation, significantly extending production timelines while introducing solvent residues that may violate pharmacopeial limits for residual solvents in active pharmaceutical ingredients. These cumulative drawbacks create substantial barriers to reliable supply chain integration particularly when scaling complex catalytic systems required for multi-kilogram production runs of high-value intermediates where yield consistency directly impacts cost-of-goods-sold metrics.
The Novel Approach
The patented methodology overcomes these limitations through an elegantly simple one-pot reaction where imidazolium salts directly react with basic copper carbonate in polar aprotic solvents like acetonitrile at moderate temperatures (50–80°C), eliminating all requirements for external bases or reducing agents while generating only carbon dioxide and water as byproducts; this fundamental innovation leverages the dual functionality of basic copper carbonate as both base source and copper precursor through its controlled thermal decomposition pathway that releases hydroxide ions at reaction temperatures without requiring additional reagents. The process operates effectively across diverse imidazolium structures with various substituents including alkyl, aryl, and heteroaryl groups while maintaining consistent yields between 42% and 81% across multiple examples without detectable oxidation products; crucially, excess basic copper carbonate is easily removed via simple filtration rather than complex extraction procedures required by traditional methods. This operational simplicity translates directly into enhanced supply chain reliability through reduced equipment requirements and simplified validation protocols while simultaneously lowering environmental impact through elimination of hazardous waste streams—key advantages that position this technology as an ideal solution for manufacturers seeking sustainable yet economically viable catalytic systems for pharmaceutical intermediate production where regulatory compliance intersects with cost efficiency imperatives.
Mechanistic Insights into N-Heterocyclic Carbene Copper Catalyst Formation
The novel synthesis mechanism operates through a concerted deprotonation pathway where carbonate anions from basic copper carbonate abstract protons from the C2 position of imidazolium salts under thermal activation (50–80°C), generating free carbenes that coordinate directly with copper(I) ions released from partial decomposition of the carbonate precursor; this process avoids redox side reactions common with alternative copper sources by maintaining a stable oxidation state throughout the reaction sequence while operating within a narrow temperature window that prevents ligand degradation or dimerization. The reaction proceeds efficiently in polar aprotic solvents like acetonitrile which solvate both organic precursors and inorganic byproducts without participating in side reactions, while optimized molar ratios (typically 1:1.5 imidazolium salt to basic copper carbonate) ensure complete conversion without excess reagent contamination; importantly, the absence of strong bases prevents hydrolysis pathways that generate impurities affecting catalytic performance in sensitive applications like asymmetric synthesis where enantiomeric purity is critical. This mechanistic elegance not only simplifies purification through straightforward filtration but also enables precise control over ligand substitution patterns essential for tailoring catalytic activity toward specific pharmaceutical intermediate transformations while maintaining exceptional thermal stability during storage and handling—key attributes that directly address R&D directors' concerns regarding process robustness and product consistency.
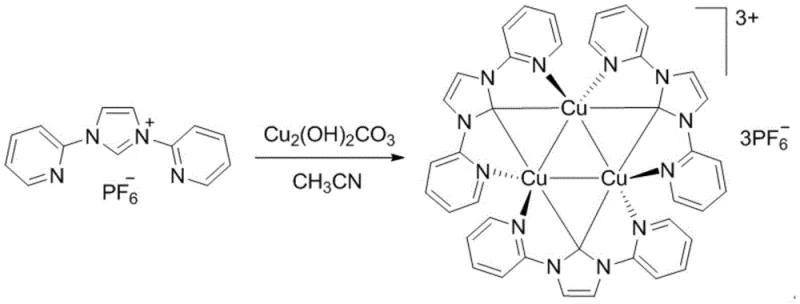
Impurity control is achieved through multiple inherent mechanisms including the selective deprotonation pathway that minimizes formation of dimeric or oxidized byproducts common in traditional methods; rigorous characterization data confirms consistent purity profiles across diverse ligand structures due to the absence of redox-active species that typically cause metal disproportionation during synthesis. The exclusive generation of water and carbon dioxide as byproducts eliminates inorganic salt contamination that plagues base-mediated routes while preventing chloride or other anion incorporation that could compromise catalytic performance in moisture-sensitive reactions; additionally, the mild thermal profile prevents thermal degradation pathways that generate low-molecular-weight impurities difficult to remove through standard crystallization techniques. This robustness extends across multiple solvent systems including acetonitrile, acetone, and nitromethane as demonstrated in patent examples without significant yield variations—critical evidence supporting scalability claims since solvent flexibility allows adaptation to existing manufacturing infrastructure without costly retooling. For R&D directors focused on impurity profiling under ICH Q3 guidelines, this methodology provides inherent advantages through predictable byproduct formation that simplifies analytical method development while ensuring consistent quality profiles essential for regulatory submissions.
How to Synthesize N-Heterocyclic Carbene Copper Catalyst Efficiently
This patented methodology represents a significant advancement over conventional approaches by eliminating hazardous reagents while maintaining high catalytic performance; its operational simplicity stems from direct combination of commercially available imidazolium salts with stable basic copper carbonate under mild thermal conditions without requiring specialized equipment or complex handling procedures. The process achieves exceptional reproducibility across multiple scales due to its robust reaction profile that tolerates minor variations in temperature or mixing efficiency—key attributes that facilitate seamless technology transfer from laboratory development to commercial manufacturing environments where process reliability directly impacts product quality metrics. Detailed standardized synthesis steps are provided below to enable immediate implementation by R&D teams seeking to integrate this technology into their pharmaceutical intermediate production workflows; these procedures have been validated across diverse ligand architectures demonstrating consistent performance regardless of substituent complexity or steric bulk requirements.
- Combine imidazolium salt precursor with basic copper carbonate in acetonitrile under inert atmosphere at room temperature
- Heat the mixture to 50–80°C with continuous stirring for 8–24 hours while monitoring reaction progress
- Cool to ambient temperature, filter to remove excess carbonate, then concentrate filtrate for recrystallization
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route directly addresses critical pain points faced by procurement and supply chain organizations through fundamental process improvements that enhance both economic viability and operational reliability; by eliminating expensive silver reagents and complex purification requirements inherent in traditional methods, it establishes a more resilient supply chain foundation capable of meeting fluctuating demand patterns without compromising quality standards or delivery timelines. The use of stable commercial precursors with extended shelf lives reduces inventory management complexity while minimizing exposure to volatile raw material markets that often disrupt traditional catalyst supply chains; additionally, simplified waste treatment protocols lower environmental compliance costs without requiring significant capital investment in specialized treatment facilities—factors that collectively strengthen negotiating positions when engaging with strategic suppliers during annual contract renewals.
- Cost Reduction in Manufacturing: Eliminating expensive silver reagents reduces raw material costs while avoiding complex waste treatment procedures associated with halide byproducts; simplified purification through filtration rather than chromatography significantly lowers processing costs without requiring additional capital expenditure on specialized equipment or hazardous waste disposal contracts—substantial cost savings realized through operational streamlining rather than marginal efficiency improvements.
- Enhanced Supply Chain Reliability: Utilizing stable commercial precursors with global availability minimizes supply chain disruptions caused by single-source dependencies; consistent performance across multiple solvent systems allows seamless adaptation to existing manufacturing infrastructure without costly retooling—dramatically simplified logistics enable reliable delivery schedules even during periods of market volatility or geopolitical instability affecting traditional catalyst supply chains.
- Scalability and Environmental Compliance: The one-pot reaction design enables straightforward scale-up from laboratory to industrial production volumes without process revalidation; benign byproduct profile consisting solely of carbon dioxide and water eliminates hazardous waste streams requiring specialized treatment—substantial environmental benefits achieved through inherent process design rather than end-of-pipe solutions while meeting increasingly stringent global sustainability regulations.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this technology; answers are derived directly from patent specifications regarding reaction mechanisms, scalability parameters, and quality control protocols validated through multiple experimental examples demonstrating consistent performance across diverse ligand architectures.
Q: How does this method prevent oxidation issues compared to traditional copper sources?
A: The process utilizes stable basic copper carbonate which avoids redox decomposition pathways inherent in CuO or CuCl₂ systems. By eliminating strong reducing agents and operating under mild thermal conditions (50–80°C), it prevents oxidation of copper(I) centers while maintaining catalytic integrity throughout synthesis.
Q: What environmental advantages does this synthesis route offer over conventional methods?
A: This method generates only carbon dioxide and water as byproducts through carbonate decomposition, eliminating inorganic salt waste streams from base-mediated reactions. The absence of toxic solvents or precious metal reagents significantly reduces EHS risks while simplifying waste treatment protocols.
Q: Can this process be reliably scaled for commercial pharmaceutical intermediate production?
A: Yes, the methodology demonstrates seamless scalability from laboratory to industrial scales due to its operational simplicity and robustness. The use of stable commercial reagents with straightforward filtration purification enables consistent production from kilogram to metric ton quantities without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heterocyclic Carbene Copper Catalyst Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation; this patented methodology aligns perfectly with our core competencies in complex catalyst manufacturing where precision engineering meets industrial-scale execution capabilities required by global pharmaceutical enterprises. By leveraging our specialized expertise in transition metal catalysis systems combined with vertically integrated supply chain management practices, we ensure consistent delivery of high-purity Cu-NHC catalysts meeting exacting regulatory requirements across all major markets including FDA-, EMA-, and PMDA-regulated regions—providing clients with unparalleled confidence in both product quality and supply continuity.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which includes specific COA data demonstrating purity profiles exceeding industry standards along with comprehensive route feasibility assessments tailored to your unique manufacturing requirements; contact us today to initiate discussions about how our scalable production capabilities can support your next-generation pharmaceutical intermediate synthesis programs.
