Advanced Silver-Catalyzed Synthesis of 4-Methylene Pyrrolidine-2-Thiones for Pharmaceutical Applications
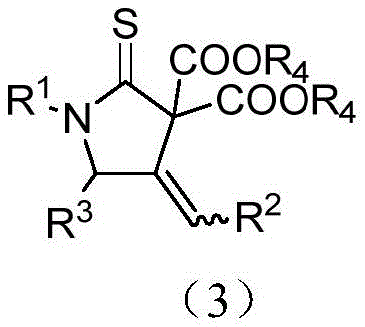
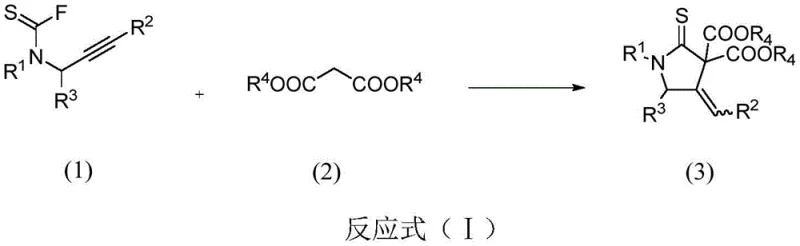
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, which serve as foundational scaffolds for bioactive molecules. Patent CN114262290A introduces a groundbreaking approach for synthesizing 4-methylenepyrrolidine-2-thione compounds, a class of structures with significant potential in anti-inflammatory, bactericidal, and anticancer drug development. This innovation leverages a silver-catalyzed tandem reaction between N-propargyl-N-alkyl/arylamine thioacyl fluorides and malonate esters, achieving high atom and step economy. Unlike traditional multi-step syntheses that often suffer from low overall yields and harsh conditions, this method operates under mild thermal parameters, typically between 40-82°C, utilizing accessible inorganic bases and silver salts. The versatility of this chemical transformation allows for the precise tuning of positional and chemical selectivity, providing a reliable pathway for generating complex heterocyclic libraries essential for modern drug discovery pipelines.
For procurement managers and supply chain directors, the implications of adopting this technology are profound. The ability to access high-purity pharmaceutical intermediates through a streamlined, one-pot process directly translates to cost reduction in API manufacturing. By minimizing the number of isolation steps and reducing solvent consumption, the overall environmental footprint is significantly lowered, aligning with global sustainability goals. Furthermore, the broad substrate scope ensures that supply chains remain resilient against raw material fluctuations, as various substituted aryl and alkyl precursors can be effectively utilized without compromising reaction efficiency. This positions the technology as a strategic asset for securing long-term supply continuity of critical drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrrolidine-2-thione cores has relied on laborious multi-step sequences involving the protection and deprotection of amine functionalities, followed by separate cyclization events. These conventional routes often necessitate the use of hazardous reagents, such as carbon disulfide or thiophosgene derivatives, which pose significant safety risks and require specialized handling infrastructure. Additionally, traditional methods frequently struggle with regioselectivity issues, leading to complex mixtures of isomers that are difficult and costly to separate. The cumulative effect of these inefficiencies results in prolonged lead times for high-purity pharmaceutical intermediates and inflated production costs. Moreover, the reliance on stoichiometric amounts of activating agents generates substantial chemical waste, creating disposal challenges that further burden the operational budget and environmental compliance protocols of manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN114262290A represents a paradigm shift towards green and efficient synthesis. By employing a silver-catalyzed intermolecular tandem addition-cyclization strategy, the process consolidates multiple bond-forming events into a single operational step. This novel approach eliminates the need for pre-functionalized cyclic precursors, instead building the heterocyclic ring directly from linear acyclic starting materials. The use of thioacyl fluorides as stable yet reactive electrophiles allows for controlled reactivity, minimizing side reactions and improving the purity profile of the crude product. The reaction proceeds smoothly in common organic solvents like 1,2-dichloroethane or acetonitrile, avoiding the need for exotic or prohibitively expensive media. This simplification of the synthetic route not only accelerates the timeline from bench to pilot plant but also enhances the overall mass balance, making it an economically superior choice for industrial applications.
Mechanistic Insights into Silver-Catalyzed Cyclization
The core of this transformative chemistry lies in the unique activation mode provided by the silver catalyst. Mechanistically, the silver cation coordinates with the alkyne moiety of the N-propargyl thioacyl fluoride, increasing its electrophilicity and facilitating nucleophilic attack by the active methylene group of the malonate ester. This initial addition generates a vinyl-silver intermediate, which subsequently undergoes an intramolecular nucleophilic attack by the nitrogen atom onto the thioacyl carbon. This cyclization step is driven by the formation of the stable five-membered thione ring and the release of the silver species, which re-enters the catalytic cycle. The presence of an inorganic base, such as cesium carbonate, is crucial for deprotonating the malonate to generate the nucleophilic enolate and neutralizing the hydrogen fluoride byproduct released during the reaction. Understanding this catalytic cycle is vital for R&D directors aiming to optimize reaction parameters for specific substrates, as fine-tuning the electronic properties of the silver ligand can further enhance turnover frequencies.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The high chemoselectivity of the silver catalyst ensures that the reaction proceeds primarily through the desired carbon-terminal addition pathway, suppressing competing side reactions such as polymerization of the alkyne or hydrolysis of the thioacyl fluoride. The mild reaction conditions (40-82°C) prevent thermal degradation of sensitive functional groups, thereby maintaining the integrity of the molecular scaffold. Furthermore, the use of thioacyl fluorides, which are more stable than their acid chloride counterparts, reduces the formation of hydrolysis byproducts during storage and handling. For quality assurance teams, this means a cleaner crude reaction profile, which simplifies downstream purification processes like column chromatography or crystallization. The result is a final product with stringent purity specifications, ready for subsequent coupling reactions in complex drug synthesis without requiring extensive remediation.
How to Synthesize 4-Methylene Pyrrolidine-2-Thione Efficiently
Implementing this synthesis in a laboratory or pilot setting requires careful attention to reagent quality and reaction monitoring. The process begins with the preparation of anhydrous reaction conditions to prevent catalyst deactivation by moisture. A typical protocol involves charging a dried reaction vessel with the silver catalyst and inorganic base, followed by the addition of the solvent and substrates. The mixture is then heated to the optimal temperature range, with reaction progress tracked via thin-layer chromatography (TLC) to ensure complete consumption of the limiting reagent. Detailed standardized synthesis steps see the guide below.
- Combine N-propargyl-N-alkyl/arylamine thioacyl fluoride and dimethyl malonate with a silver catalyst (e.g., AgNTf2) and inorganic base (e.g., Cs2CO3) in anhydrous 1,2-dichloroethane.
- Heat the reaction mixture to 60°C and monitor progress via TLC until the starting thioacyl fluoride is fully consumed, typically requiring 4 to 24 hours.
- Filter off precipitates, concentrate the filtrate under reduced pressure, and purify the residue via silica gel column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this silver-catalyzed methodology offers tangible benefits for the commercial viability of pharmaceutical projects. From a procurement perspective, the reliance on commercially available silver salts and simple malonate esters reduces dependency on custom-synthesized building blocks, which often carry high price tags and long lead times. The elimination of transition metal catalysts that require rigorous removal steps, such as palladium, simplifies the purification workflow and lowers the cost of goods sold. While silver is a precious metal, the catalytic loading is low, and recovery protocols can be implemented to further mitigate material costs. This economic efficiency is compounded by the high yields observed across a wide range of substrates, ensuring that raw material inputs are converted into valuable product with minimal waste.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction significantly reduces operational expenditures associated with multiple reactor charges, workups, and intermediate isolations. By consolidating the synthesis into a single step, labor costs and utility consumption are drastically minimized. Furthermore, the avoidance of hazardous reagents reduces the need for specialized safety equipment and waste treatment protocols, contributing to substantial cost savings. The high atom economy ensures that the majority of the mass of the starting materials ends up in the final product, optimizing the utilization of expensive precursors and reducing the overall material intensity of the process.
- Enhanced Supply Chain Reliability: The robustness of this chemical platform against variations in substrate structure provides a buffer against supply chain disruptions. If a specific aryl precursor becomes unavailable, the protocol's flexibility allows for the rapid substitution with alternative analogues without the need for extensive process re-validation. This adaptability ensures continuous production schedules and prevents bottlenecks in the manufacturing pipeline. Additionally, the use of stable thioacyl fluoride intermediates allows for inventory buffering, as these materials can be stored for extended periods without significant degradation, unlike more reactive acid chlorides. This stability enhances the predictability of raw material availability and supports just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the mild thermal requirements and the use of standard organic solvents. The absence of exothermic hazards associated with traditional cyclization reagents makes the process safer to operate on a large scale. From an environmental standpoint, the generation of inorganic salts as the primary byproduct simplifies waste stream management compared to processes generating organic sludge. The green chemistry metrics of this method, including high E-factors and low process mass intensity, align with increasingly strict regulatory standards for pharmaceutical manufacturing. This compliance reduces the risk of regulatory delays and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and scope limitations defined in the patent literature. They serve as a preliminary guide for process chemists evaluating the feasibility of this technology for their specific projects. For more detailed technical support, direct consultation with our engineering team is recommended.
Q: What are the optimal reaction conditions for this silver-catalyzed cyclization?
A: The optimal conditions involve using AgNTf2 as the catalyst and Cs2CO3 as the base in anhydrous 1,2-dichloroethane at 60°C, yielding up to 90% conversion.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent tolerance for various substituents including halogens (F, Cl, Br, I), electron-donating groups (OMe), and electron-withdrawing groups (CF3, CN).
Q: Is this synthesis suitable for large-scale pharmaceutical manufacturing?
A: Absolutely. The process utilizes readily available reagents, operates at moderate temperatures (40-82°C), and avoids complex multi-step sequences, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methylene Pyrrolidine-2-Thione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex heterocyclic chemistry safely and efficiently, providing you with a dependable source for your key building blocks.
We invite you to collaborate with us to leverage this innovative silver-catalyzed technology for your next pharmaceutical program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time to market with our superior manufacturing capabilities and dedication to quality excellence.
