Advanced Silver-Catalyzed Synthesis of 4-Methylenepyrrolidine-2-Thiones for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for nitrogen-containing heterocycles, particularly those serving as privileged scaffolds in drug discovery. Patent CN114262290A introduces a groundbreaking methodology for the synthesis of 4-methylenepyrrolidine-2-thione compounds, a class of molecules with significant potential in developing anti-inflammatory, antibacterial, and anticancer agents. This innovation addresses critical bottlenecks in existing synthetic strategies by leveraging a silver-catalyzed tandem cyclization that ensures high regioselectivity and chemical efficiency. The core structural motif, characterized by a five-membered ring containing both nitrogen and sulfur atoms with an exocyclic double bond, is notoriously difficult to construct with high purity using traditional methods. By utilizing N-propargyl-N-alkyl/arylamine thioacyl fluorides as key precursors, this technology enables the direct assembly of complex thiopyrrolidone derivatives in a single operational step.
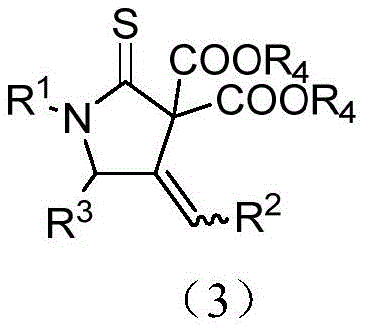
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the production of high-value intermediates. The described compounds are not merely academic curiosities but serve as essential building blocks for next-generation therapeutics. The patent details a versatile approach where the substitution patterns on the nitrogen atom and the exocyclic double bond can be precisely tuned, offering a wide chemical space for medicinal chemistry optimization. This level of control is paramount when developing candidates that require specific steric and electronic properties to interact with biological targets. Furthermore, the method's reliance on easily accessible starting materials reduces the dependency on exotic reagents, thereby simplifying the overall supply chain logistics for pharmaceutical manufacturers seeking reliable sources of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 4-methylenepyrrolidine-2-thione scaffolds has been plagued by inefficient multi-step sequences that suffer from poor atom economy and low overall yields. Traditional routes often require the pre-functionalization of starting materials with protecting groups, followed by harsh cyclization conditions that can lead to decomposition or the formation of unwanted regioisomers. The use of stoichiometric amounts of heavy metal reagents or strong bases in older methodologies frequently results in significant waste generation and complicates the downstream purification process. Additionally, controlling the stereochemistry of the exocyclic double bond has been a persistent challenge, often necessitating costly separation techniques to isolate the desired E or Z isomer. These inefficiencies translate directly into higher production costs and longer lead times, creating substantial barriers for the commercial viability of drugs relying on this chemical core.
The Novel Approach
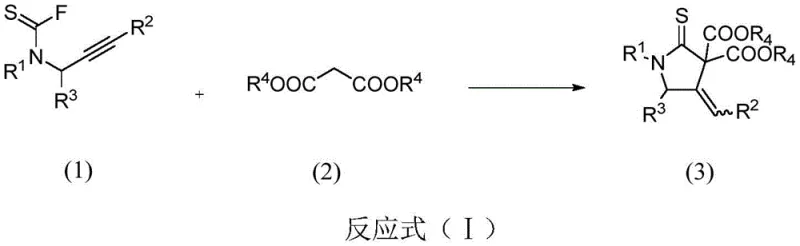
In stark contrast, the novel approach detailed in CN114262290A utilizes a silver-catalyzed intermolecular tandem reaction that dramatically simplifies the synthetic landscape. By reacting N-propargyl thioacyl fluorides with malonate esters in the presence of a silver catalyst and an inorganic base, the process achieves a direct carbon-terminal addition to the alkyne followed by spontaneous cyclization. This one-pot transformation eliminates the need for intermediate isolation and protection-deprotection steps, significantly reducing the operational complexity. The reaction proceeds under mild thermal conditions, typically between 40°C and 82°C, which preserves the integrity of sensitive functional groups that might otherwise degrade under harsher regimes. The use of catalytic amounts of silver salts, such as AgOTf or AgNTf2, ensures that the process remains cost-effective while maintaining high levels of conversion and selectivity.
Mechanistic Insights into Silver-Catalyzed Tandem Cyclization
The mechanistic elegance of this transformation relies on the unique ability of the silver cation to activate the alkyne moiety through pi-coordination. Upon coordination, the electron density of the triple bond is polarized, rendering the terminal carbon more electrophilic and susceptible to nucleophilic attack. The active methylene group of the malonate ester, deprotonated by the inorganic base such as cesium carbonate, acts as the nucleophile that initiates the cascade. This initial addition generates a vinyl-silver intermediate, which subsequently undergoes an intramolecular nucleophilic attack by the sulfur atom of the thioacyl group. This cyclization step closes the five-membered ring and releases the silver catalyst back into the cycle, allowing it to facilitate further reactions. The precise tuning of the silver counterion and the base strength is critical for balancing the reaction kinetics and preventing side reactions such as polymerization or hydrolysis of the thioacyl fluoride.
From an impurity control perspective, this catalytic system offers distinct advantages by promoting a highly specific reaction pathway. The chemoselectivity of the silver catalyst ensures that the reaction occurs exclusively at the alkyne terminus, minimizing the formation of regioisomeric byproducts that are common in uncatalyzed thermal cyclizations. Furthermore, the mild reaction conditions help to suppress the degradation of the thioamide functionality, which is prone to oxidation or hydrolysis under acidic or strongly basic environments. The resulting crude reaction mixtures are typically cleaner, facilitating easier purification via standard silica gel chromatography. This high level of purity is essential for pharmaceutical applications, where strict limits on impurities are enforced by regulatory bodies. The ability to consistently produce high-purity intermediates reduces the risk of batch failures and ensures the quality of the final active pharmaceutical ingredient.
How to Synthesize 4-Methylenepyrrolidine-2-Thione Efficiently
The practical implementation of this synthesis protocol is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves charging a dried reaction vessel with the silver catalyst and inorganic base, followed by the addition of the thioacyl fluoride substrate and the malonate ester in an anhydrous organic solvent such as 1,2-dichloroethane. The mixture is then heated to the optimal temperature range, typically around 60°C, and monitored via thin-layer chromatography until the starting material is fully consumed. Following the reaction, the workup is straightforward, involving filtration to remove inorganic salts and concentration of the filtrate under reduced pressure. The detailed standardized synthesis steps for this process are outlined in the guide below.
- Combine N-propargyl-N-alkyl/arylamine thioacyl fluoride and dimethyl malonate with a silver catalyst and inorganic base in anhydrous solvent.
- Heat the reaction mixture to 40-82°C and monitor progress via TLC until the starting thioacyl fluoride is fully consumed.
- Filter off precipitates, concentrate the filtrate under reduced pressure, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of complex multi-step sequences and the use of commercially available, off-the-shelf reagents significantly lower the raw material costs associated with producing these specialized intermediates. By reducing the number of unit operations required, manufacturers can achieve substantial savings in labor, energy consumption, and equipment usage. The robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, leading to more consistent batch-to-batch performance and reducing the risk of production delays. This stability is crucial for maintaining a steady supply of critical intermediates to downstream drug manufacturing facilities.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis directly translates to lower operational expenditures by removing the need for expensive protecting group chemistry and intermediate purification stages. The use of catalytic rather than stoichiometric amounts of silver further optimizes the cost structure, making the process economically viable for large-scale production. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a more sustainable and cost-effective manufacturing footprint. These cumulative efficiencies allow for a more competitive pricing strategy for the final pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as malonate esters and simple amines mitigates the risk of supply chain disruptions caused by the scarcity of exotic reagents. The mild reaction temperatures and standard solvent systems mean that the process can be easily transferred between different manufacturing sites without requiring specialized equipment or extensive re-validation. This flexibility enhances the resilience of the supply chain, ensuring that production can continue uninterrupted even in the face of logistical challenges. Furthermore, the high yield and selectivity of the reaction maximize the output from each batch, ensuring that demand can be met consistently and reliably.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this methodology, such as high atom economy and reduced waste generation, align perfectly with modern environmental regulations and corporate sustainability goals. The process avoids the use of toxic heavy metals in stoichiometric quantities and minimizes the generation of hazardous byproducts, simplifying compliance with environmental discharge standards. The scalability of the reaction has been demonstrated across a wide range of substrate scopes, indicating that it can be successfully scaled from gram-scale laboratory experiments to multi-ton commercial production. This scalability ensures that the technology can support the growing demand for these intermediates as drug candidates progress through clinical trials to market launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 4-methylenepyrrolidine-2-thione compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for making informed decisions about integrating this technology into your existing manufacturing workflows.
Q: What are the key advantages of this silver-catalyzed method over traditional synthesis routes?
A: This method offers superior atom economy and step efficiency by utilizing a one-pot tandem reaction that avoids harsh conditions and expensive transition metals often required in conventional multi-step sequences.
Q: Is this synthesis protocol suitable for large-scale commercial production?
A: Yes, the process utilizes readily available silver catalysts and operates at mild temperatures (40-82°C) in common organic solvents, making it highly amenable to scale-up from kilogram to multi-ton manufacturing.
Q: What is the substrate scope for the N-substituent in the thioacyl fluoride precursor?
A: The methodology demonstrates broad compatibility, successfully accommodating various aryl, alkyl, and heteroaryl groups on the nitrogen atom, allowing for diverse functionalization of the final thiopyrrolidone scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methylenepyrrolidine-2-Thione Supplier
As a leader in the fine chemical and pharmaceutical intermediate sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthetic technology for our global clients. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-methylenepyrrolidine-2-thione derivatives meets the highest quality standards required by the pharmaceutical industry. Our commitment to excellence extends beyond mere production; we act as a strategic partner in optimizing your supply chain for maximum efficiency and reliability.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the potential economic advantages of switching to this silver-catalyzed method. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us help you secure a stable and cost-effective supply of high-purity pharmaceutical intermediates for your next generation of therapeutic innovations.
