Scalable Catalyst-Free Synthesis of 2-Substituted Benzothiazoles for Commercial Pharmaceutical Manufacturing
Scalable Catalyst-Free Synthesis of 2-Substituted Benzothiazoles for Commercial Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more cost-effective manufacturing processes. A pivotal development in this field is documented in Chinese Patent CN111909113A, which discloses a robust method for synthesizing 2-substituted benzothiazole compounds under strictly catalyst-free and additive-free conditions. This technology represents a major leap forward for the fine chemical industry, particularly for those seeking a reliable pharmaceutical intermediate supplier capable of delivering high-purity scaffolds without the burden of heavy metal contamination. The core innovation lies in the utilization of elemental sulfur as an inorganic sulfur source, reacting directly with readily available aromatic amines and benzylamines. This approach not only simplifies the synthetic route but also drastically reduces the environmental footprint associated with traditional thiol-based or metal-catalyzed methodologies. By operating at moderate temperatures between 130°C and 150°C, the process ensures high functional group tolerance while maintaining excellent atom economy. For R&D directors and procurement managers alike, this patent offers a compelling solution to the challenges of sourcing complex heterocycles, promising a pathway to substantial cost savings and enhanced supply chain reliability through the use of commodity chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzothiazoles has relied heavily on methods that present significant logistical and economic hurdles for large-scale production. Traditional routes often utilize thiols or thioamides as organic sulfur sources, which are notoriously difficult to handle due to their unpleasant odors, high toxicity, and instability upon storage. Furthermore, many established protocols depend on transition metal catalysis involving ortho-haloanilines, which introduces the risk of residual metal impurities that are strictly regulated in pharmaceutical applications. The removal of these metals requires additional purification steps, such as scavenging or recrystallization, which inevitably lowers the overall yield and increases the cost of goods sold. Additionally, the starting materials for these conventional methods, particularly substituted ortho-haloanilines, can be expensive and limited in structural diversity, restricting the chemical space accessible to medicinal chemists. These factors combined create a bottleneck in the supply chain, leading to longer lead times and higher prices for the final active pharmaceutical ingredients or agrochemical intermediates derived from these scaffolds.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a streamlined, direct multicomponent reaction that bypasses these traditional pitfalls entirely. By employing elemental sulfur, a cheap and abundant inorganic reagent, the process eliminates the need for malodorous thiols and expensive metal catalysts. The reaction proceeds through a cascade sequence where benzylamine is initially oxidized to an aldehyde, which then condenses with the aromatic amine to form a Schiff base intermediate before undergoing cyclization with sulfur. This atom-economical pathway allows for the direct construction of the benzothiazole core from simple, commercially available building blocks. The absence of additives and catalysts means the workup procedure is significantly simplified, often requiring only standard extraction and chromatography to achieve high purity. This novel approach not only enhances the safety profile of the manufacturing process but also opens up new possibilities for introducing diverse substituents on the aromatic rings, thereby expanding the utility of these compounds in drug discovery and material science applications.
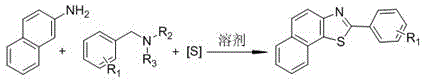
Mechanistic Insights into Metal-Free Multicomponent Cyclization
The mechanistic pathway of this catalyst-free synthesis is a fascinating example of oxidative cyclization driven by the solvent system itself. The reaction initiates with the oxidation of the benzylamine substrate to its corresponding aldehyde, a transformation facilitated by the oxidizing nature of dimethyl sulfoxide (DMSO) at elevated temperatures. This in situ generation of the aldehyde is critical as it immediately reacts with the aromatic amine component to form a Schiff base intermediate. Unlike metal-catalyzed C-H activation which often requires harsh conditions or specific directing groups, this condensation occurs readily under the thermal conditions provided. The subsequent step involves the nucleophilic attack of the imine nitrogen or the aromatic ring on the elemental sulfur species, leading to the formation of the C-S bond. The final aromatization yields the stable 2-substituted benzothiazole ring system. This mechanism highlights the dual role of DMSO as both a high-boiling polar solvent and a mild oxidant, eliminating the need for external oxidizing agents that could generate hazardous waste streams.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring product quality. Since no transition metals are introduced into the reaction matrix, the risk of metal-catalyzed side reactions, such as homocoupling or over-oxidation, is virtually eliminated. The primary byproducts are typically derived from the over-oxidation of the amine or incomplete cyclization, both of which are easily separated during the aqueous workup and column chromatography stages. The high functional group tolerance observed in the patent examples, including compatibility with halogens, trifluoromethyl groups, and methoxy substituents, suggests that the reaction conditions are mild enough to preserve sensitive moieties. This level of selectivity is paramount for pharmaceutical manufacturers who must adhere to strict impurity profiles. The ability to achieve yields as high as 90% in model reactions indicates a highly efficient conversion that minimizes the formation of complex impurity mixtures, thereby simplifying the downstream purification process and ensuring a consistent supply of high-purity intermediates.
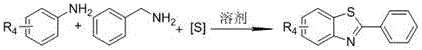
How to Synthesize 2-Substituted Benzothiazoles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to maximize yield and safety. The process is designed to be operationally simple, utilizing standard glassware and heating equipment without the need for specialized high-pressure reactors. The key to success lies in maintaining the correct molar ratios, specifically using an excess of benzylamine and elemental sulfur relative to the aromatic amine to drive the equilibrium towards the product. The reaction temperature must be tightly controlled within the 130°C to 150°C range; temperatures below this threshold may result in incomplete conversion, while excessive heat could lead to decomposition of the sensitive Schiff base intermediate. Detailed standardized synthetic steps see the guide below.
- Charge a dry reaction vessel with aromatic amine, benzylamine, and elemental sulfur in a molar ratio of approximately 1: 2:3.
- Add dimethyl sulfoxide (DMSO) as the solvent and heat the mixture to 130-150°C under nitrogen atmosphere.
- Stir for 9-15 hours, cool to room temperature, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the reduction of raw material costs, as the process replaces expensive palladium or copper catalysts and specialized organosulfur reagents with commodity-grade elemental sulfur and bulk amines. This shift significantly lowers the barrier to entry for producing these valuable heterocycles, allowing for more competitive pricing in the final API market. Furthermore, the simplified workflow reduces the dependency on complex purification infrastructure, such as metal scavenging columns or extensive recrystallization setups, which streamlines the manufacturing timeline. The robustness of the reaction conditions also implies a lower risk of batch failure due to catalyst deactivation or sensitivity to moisture and oxygen, thereby enhancing the overall reliability of the supply chain.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major cost driver from the bill of materials, while the use of inexpensive elemental sulfur further drives down the variable costs per kilogram. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with purification, leading to substantial operational expenditure savings. By avoiding the need for specialized handling equipment for toxic thiols, the facility can also reduce capital expenditure on safety infrastructure and waste treatment systems. These cumulative efficiencies result in a leaner manufacturing process that is highly resilient to fluctuations in the prices of specialty chemicals.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on widely available starting materials like benzylamines and anilines, which are produced on a massive scale globally. This abundance ensures that production schedules are not held hostage by the limited availability of niche reagents or the long lead times often associated with custom-synthesized catalysts. The stability of the reagents also allows for longer storage periods without degradation, enabling manufacturers to maintain strategic stockpiles and buffer against market volatility. Consequently, the risk of supply disruption is minimized, ensuring a continuous flow of intermediates to downstream customers.
- Scalability and Environmental Compliance: The atom-economic nature of this reaction aligns perfectly with modern green chemistry principles, generating minimal waste and avoiding the discharge of heavy metals into the environment. This compliance simplifies the regulatory approval process for new manufacturing sites and reduces the costs associated with hazardous waste disposal. The process has already been demonstrated to work effectively on a gram scale with good yields, suggesting a straightforward path to kilogram and ton-scale production without the need for extensive re-optimization. This scalability ensures that the technology can meet the growing demand for benzothiazole derivatives in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, providing clarity for stakeholders evaluating its implementation. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for making informed decisions about process adoption and integration into existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free benzothiazole synthesis?
A: The primary advantages include the elimination of expensive transition metal catalysts, the use of cheap and stable elemental sulfur instead of smelly thiols, and mild reaction conditions that tolerate diverse functional groups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful gram-scale experiments with high yields and simple workup procedures involving standard extraction and chromatography, indicating strong potential for commercial scale-up.
Q: What is the role of DMSO in this reaction mechanism?
A: DMSO serves a dual role as both the reaction solvent and an oxidant, facilitating the initial oxidation of benzylamine to the corresponding aldehyde which is crucial for the subsequent condensation and cyclization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzothiazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis technology for the global pharmaceutical and agrochemical industries. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that consistency is key in fine chemical manufacturing, and our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of sulfur-based chemistries with precision and care.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the future of sustainable chemical manufacturing.
