Advanced Liquid-Phase Synthesis of 5-Ester 1,3-Aryl Pyrazolines for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic methodologies that balance structural diversity with manufacturing efficiency. Patent CN1515557A introduces a transformative approach to the synthesis of 1,3-aryl substituted pyrazolines containing ester groups at the 5-position, utilizing a novel liquid-phase synthesis strategy. This technology addresses the critical bottlenecks associated with traditional solution-phase reactions by employing soluble polyethylene glycol (PEG) resins as supports. By integrating the kinetic benefits of homogeneous catalysis with the separation simplicity of heterogeneous systems, this method enables the rapid construction of molecular libraries essential for drug discovery. The process eliminates the need for complex purification steps typically required in solution chemistry, thereby streamlining the path from bench-scale experimentation to commercial production. For R&D directors and procurement specialists alike, this represents a significant leap forward in generating high-value heterocyclic scaffolds with enhanced cost-effectiveness and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1,3-aryl substituted pyrazolines have historically relied on solution-phase reactions that present substantial operational challenges for large-scale manufacturing. Conventional methods often involve multi-step sequences, such as the preparation of chlorohydrazones followed by cycloaddition with alkenes, or the oxidation of hydrazones prior to reaction with olefins. These processes frequently necessitate the use of expensive reagents, harsh reaction conditions, and rigorous purification protocols like column chromatography to achieve acceptable purity levels. Furthermore, the inability to directly utilize commercially available starting materials in a convergent manner often leads to lower overall yields and increased waste generation. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times, while R&D teams struggle with the time-consuming nature of optimizing each discrete step for new analogues. The lack of a unified, one-pot protocol makes parallel synthesis and library generation prohibitively difficult, hindering the rapid structure-activity relationship studies required in modern drug development.
The Novel Approach
In stark contrast, the liquid-phase synthesis method described in the patent leverages soluble polymer-supported technology to overcome these historical limitations. By anchoring the acrylate component onto a soluble PEG resin, the reaction proceeds under homogeneous conditions, ensuring excellent mass transfer and reaction kinetics similar to solution chemistry. However, the unique solubility profile of the PEG support—soluble in dichloromethane and methanol but insoluble in ether and isopropanol—allows for facile product isolation through simple precipitation and filtration. This one-pot cyclization strategy enables the direct condensation of aromatic aldehydes and hydrazine salts followed by immediate ring closure with the resin-bound acrylate and an oxidant. This convergence not only drastically reduces the number of unit operations but also minimizes solvent consumption and waste disposal costs. For supply chain heads, this translates to a more streamlined manufacturing process that is inherently safer and more environmentally compliant, offering a sustainable pathway for producing complex pyrazoline derivatives at scale.
Mechanistic Insights into Soluble Resin-Supported Cyclization
The core of this innovative synthesis lies in the strategic use of soluble polyethylene glycol (PEG) as a phase-tag that dictates the reaction environment and workup procedure. The mechanism initiates with the covalent attachment of substituted acryloyl chloride to the hydroxyl termini of the PEG chain in the presence of an organic base, creating a soluble macromolecular reagent. Subsequently, the formation of the hydrazone intermediate occurs in situ from equimolar amounts of aromatic aldehyde and hydrazine hydrochloride, facilitated by an organic base. Upon addition of the resin-supported acrylate and a mild oxidizing agent such as Chloramine T, a 1,3-dipolar cycloaddition cascade is triggered. The oxidant plays a crucial role in generating the reactive nitrile imine or similar dipole species necessary for the ring-closing step, ensuring regioselective formation of the pyrazoline core. Because the entire reaction mixture remains in a single liquid phase during the transformation, diffusion limitations common in solid-phase synthesis are entirely avoided, leading to superior conversion rates and reaction homogeneity.
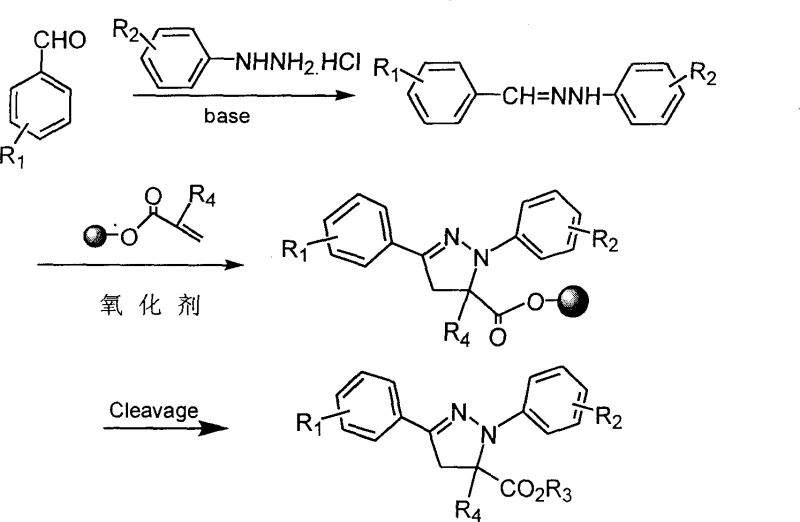
Following the cyclization, the final critical step involves the cleavage of the product from the polymer support. This is achieved through a transesterification or nucleophilic displacement mechanism using sodium alkoxide in an alcoholic solvent. The choice of alkoxide corresponds to the desired ester group in the final product, allowing for modular diversification of the 5-position substituent. Once cleaved, the small molecule product remains in solution while the PEG resin precipitates out upon the addition of a non-solvent like diethyl ether. This physical separation is driven by the distinct solubility parameters of the polymer versus the small molecule, effectively acting as a "filter-free" purification step. For quality control teams, this mechanism ensures that metal catalysts or heavy oxidant byproducts associated with the resin can be easily washed away, resulting in crude products with purity levels often exceeding 90%, which is sufficient for initial biological screening without further chromatographic refinement.
How to Synthesize 5-Ester 1,3-Aryl Pyrazolines Efficiently
The synthesis of these valuable heterocyclic intermediates follows a logical three-stage workflow designed for maximum operational simplicity and yield optimization. The process begins with the preparation of the soluble resin support, followed by the one-pot assembly of the pyrazoline core, and concludes with the release of the final esterified product. This standardized protocol is highly adaptable to various substituents on the aromatic rings, making it ideal for generating diverse compound libraries. Detailed standard operating procedures for each stage, including specific temperature controls and stoichiometric ratios, are outlined in the technical guide below to ensure reproducibility and safety during scale-up.
- Load substituted acryloyl chloride onto soluble PEG resin in an organic solvent with a base at 0-35°C.
- Perform a one-pot cyclization by reacting aromatic aldehydes and hydrazine salts to form hydrazones, then adding the resin-supported acrylate and an oxidant like Chloramine T.
- Cleave the final product from the resin using sodium alkoxide in an alcohol solvent, followed by crystallization to isolate the pure pyrazoline ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this soluble resin-supported technology offers compelling economic and logistical benefits over legacy manufacturing routes. The shift from multi-step solution synthesis to a convergent one-pot process fundamentally alters the cost structure of producing pyrazoline intermediates. By eliminating intermediate isolation steps and reducing the reliance on expensive purification media like silica gel, the overall cost of goods sold is significantly reduced. Furthermore, the use of commodity chemicals such as polyethylene glycol, common organic solvents, and readily available aromatic aldehydes ensures a stable and resilient supply chain less susceptible to market volatility. The simplified workup procedure also drastically shortens the production cycle time, allowing facilities to increase throughput without requiring additional capital investment in reactor capacity. This efficiency gain is critical for meeting tight delivery schedules in the fast-paced pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in solvent usage directly contribute to substantial cost savings. Traditional methods often require expensive reagents and extensive chromatographic purification, which are both cost-prohibitive and time-consuming. In this novel process, the soluble resin acts as a recyclable or disposable scaffold that simplifies downstream processing, removing the need for complex distillation or column chromatography. The ability to achieve high crude purity through simple crystallization means that resources previously allocated to purification can be redirected towards increasing batch sizes or developing new analogues. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further lowering the operational expenditure associated with utility costs in large-scale production facilities.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted benzaldehydes and hydrazine salts mitigates the risk of supply disruptions. Unlike specialized reagents that may have long lead times or single-source suppliers, the inputs for this synthesis are commodity chemicals with robust global supply networks. The simplicity of the reaction setup also reduces the dependency on highly specialized equipment or skilled labor for complex manipulations, making it easier to qualify multiple contract manufacturing organizations (CMOs) for production. This redundancy in the supply base ensures business continuity and allows procurement teams to negotiate better terms due to the flexibility of sourcing. Moreover, the stability of the resin-supported intermediates allows for potential stockpiling of key reagents, buffering against short-term market fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often introduces unforeseen challenges, particularly regarding heat transfer and mixing efficiency. This liquid-phase synthesis operates under homogeneous conditions, which inherently scales better than heterogeneous solid-phase reactions where diffusion limits can become problematic at larger volumes. The use of mild oxidants like Chloramine T avoids the generation of hazardous waste streams associated with stronger oxidizing agents, simplifying effluent treatment and regulatory compliance. The precipitation-based workup minimizes the volume of organic waste solvents generated, aligning with green chemistry principles and reducing disposal costs. For environmental health and safety officers, this process presents a lower risk profile, facilitating faster regulatory approvals and smoother audits during the technology transfer phase.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this soluble resin-supported synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility of adopting this method for your specific application needs. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of using soluble PEG resin over traditional solution-phase synthesis for pyrazolines?
A: Soluble PEG resin combines the kinetic advantages of homogeneous solution chemistry with the separation ease of solid-phase synthesis. It allows for simplified purification through precipitation, significantly reducing solvent usage and processing time compared to traditional multi-step solution methods.
Q: How does this liquid-phase method impact the purity of the final pharmaceutical intermediates?
A: The method utilizes the differential solubility of the PEG support to facilitate easy removal of impurities. Crude products often achieve purity levels exceeding 90% after simple crystallization and washing, making them immediately suitable for biological screening without extensive chromatography.
Q: Is this synthesis route scalable for industrial production of agrochemical intermediates?
A: Yes, the process uses common organic solvents and mild reaction conditions (0-80°C), which are highly amenable to scale-up. The one-pot nature of the cyclization step minimizes unit operations, enhancing throughput and reducing overall manufacturing costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Aryl Pyrazolines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics and agrochemicals. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 1,3-aryl pyrazolines that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement advanced technologies like soluble polymer-supported synthesis allows us to offer cost-effective solutions without compromising on quality or delivery timelines, positioning us as a strategic partner for your long-term growth.
We invite you to collaborate with us to leverage these technological advancements for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this optimized synthesis route can reduce your overall manufacturing expenses. Please contact us today to request specific COA data for our existing inventory or to discuss route feasibility assessments for custom derivatives. Let us help you accelerate your development pipeline with reliable, high-quality chemical intermediates delivered with precision and professionalism.
