Scalable One-Pot Synthesis of 1-(2-Quinolinyl)-β-Carboline Natural Products for Commercial API Manufacturing
The pharmaceutical industry continuously seeks efficient pathways to access bioactive alkaloids, particularly the 1-(2-quinolinyl)-β-carboline scaffold found in natural products like Nitramarine and Nitraridine. Patent CN110776510A discloses a groundbreaking preparation method that addresses the longstanding challenges of low yields and complex multi-step sequences associated with these compounds. This innovation leverages a tandem reaction strategy where substituted methylquinoline derivatives and tryptophan or tryptamine derivatives undergo a direct contact reaction in the presence of elemental iodine. The significance of this technology lies in its ability to construct the complex heterocyclic framework in a single pot, drastically simplifying the manufacturing workflow. For R&D directors and process chemists, this represents a pivotal shift from laborious classical syntheses to a streamlined, high-efficiency protocol that maintains high purity standards while minimizing operational hazards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(2-quinolinyl)-β-carboline natural products has been plagued by inefficiencies that hinder commercial viability. Early methodologies, such as those reported by Satoshi Hibino in 1986, relied on intramolecular thermal cyclization of azatrienes, necessitating the use of indole formaldehyde substrates and multiple protection-deprotection steps involving N-benzenesulfinyl groups. These routes often suffered from poor atom economy and required harsh reagents like strong bases and hydroxylamine hydrochloride for oxime cyclization. Furthermore, subsequent improvements by Tulyaganov in 2006, which utilized high-temperature condensation of tryptamine with quinoline carboxylic acids or aldehydes, still yielded disappointing results with isolated yields hovering around 9% to 16%. Such low efficiency translates directly into excessive raw material consumption and difficult purification processes, making cost reduction in pharmaceutical intermediate manufacturing nearly impossible with these legacy techniques.
The Novel Approach
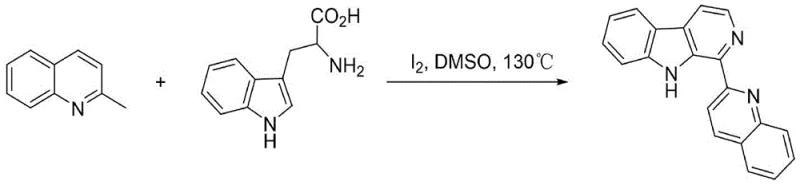
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated iodine-mediated cascade reaction that integrates functionalization, cyclization, and oxidation into a unified process. By employing inexpensive methylquinoline derivatives and readily available tryptophan or tryptamine precursors, the method bypasses the need for pre-functionalized aldehydes or acids. The reaction proceeds smoothly in dimethyl sulfoxide (DMSO) at moderate temperatures, typically around 130°C, achieving yields as high as 90% in optimized examples. This drastic improvement in efficiency is not merely incremental; it fundamentally alters the economic landscape of producing these valuable alkaloids. The one-pot nature eliminates the need for isolating sensitive intermediates, thereby reducing solvent usage and processing time, which are critical factors for enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Iodine-Catalyzed Tandem Cyclization
The mechanistic elegance of this transformation relies on the dual role of elemental iodine acting simultaneously as a catalyst and an oxidant within the DMSO medium. Detailed mechanistic studies indicate that the reaction initiates with the iodination of the methyl group on the quinoline ring to generate a 2-(iodomethyl)-quinoline intermediate. This species is highly reactive and can further oxidize to form 2-formylquinoline in situ. These transient intermediates then engage with the amino acid or amine functionality of the tryptophan derivative through a Pictet-Spengler-type cyclization. The presence of iodine ensures that the resulting tetrahydro-β-carboline intermediate undergoes rapid oxidative dehydrogenation to yield the fully aromatic 1-(2-quinolinyl)-β-carboline core. This seamless integration of steps prevents the accumulation of side products and drives the equilibrium towards the desired thermodynamic product.
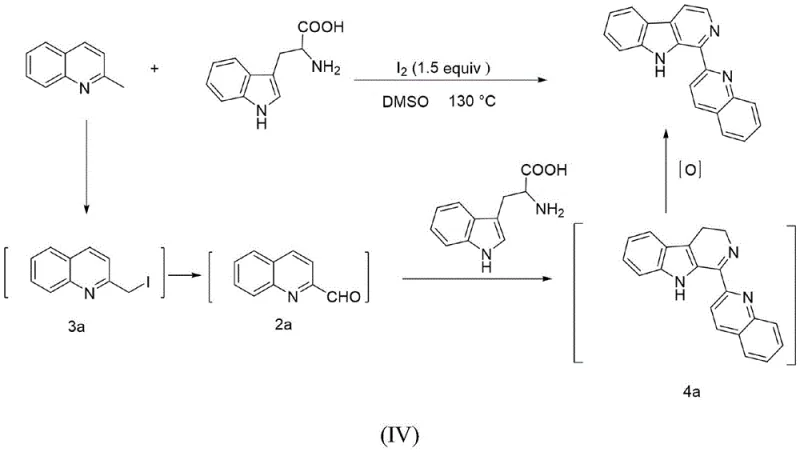
Understanding the impurity profile is crucial for regulatory compliance, and this mechanism offers inherent advantages in impurity control. Because the reactive intermediates such as 2-(iodomethyl)-quinoline and 2-formylquinoline are generated in situ and consumed immediately, their steady-state concentrations remain low, minimizing the potential for polymerization or non-specific side reactions that often plague stepwise syntheses. Additionally, the oxidative power of the iodine-DMSO system is sufficiently selective to aromatize the carboline ring without over-oxidizing sensitive functional groups on the quinoline or indole moieties, such as methoxy or halogen substituents. This selectivity ensures that the final crude product contains fewer structurally related impurities, simplifying the downstream purification burden and ensuring that the stringent purity specifications required for API intermediates can be met with standard chromatographic techniques.
How to Synthesize Nitramarine Efficiently
The practical implementation of this synthesis is straightforward and robust, making it ideal for both laboratory scale-up and industrial production. The general procedure involves charging a pressure-resistant tube with the specific methylquinoline derivative, the corresponding tryptophan or tryptamine salt, and elemental iodine in a molar ratio that favors complete conversion, typically around 1:1.4:1.5. The mixture is suspended in DMSO and heated to 130°C with magnetic stirring for 6 to 10 hours. Following the reaction, the workup is remarkably simple: the mixture is cooled, diluted with water, and extracted with an organic solvent like dichloromethane or ethyl acetate. A key step involves washing the organic layer with sodium thiosulfate to quench any unreacted iodine, followed by drying and concentration. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Combine substituted methylquinoline derivatives, tryptophan or tryptamine derivatives, and elemental iodine in dimethyl sulfoxide (DMSO) solvent within a pressure-resistant vessel.
- Heat the reaction mixture to a temperature range of 50-150°C, preferably 130°C, and maintain magnetic stirring for a duration of 6 to 10 hours to facilitate the tandem cyclization and oxidation.
- Upon completion, perform an aqueous workup involving extraction with organic solvents such as dichloromethane, wash with sodium thiosulfate to remove excess iodine, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed methodology presents a compelling value proposition centered on cost efficiency and operational stability. Traditional routes for synthesizing β-carboline alkaloids often rely on precious metal catalysts or exotic reagents that are subject to volatile market pricing and supply constraints. By shifting to a base-metal-free system utilizing abundant iodine and commodity solvents like DMSO, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the reduction in unit operations—from multiple isolated steps to a single pot—drastically lowers energy consumption and labor costs associated with intermediate handling and quality control testing. This streamlined process directly contributes to cost reduction in pharmaceutical intermediate manufacturing by maximizing throughput per batch and minimizing waste disposal expenses.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction of synthetic steps from five or six down to a single operation significantly lowers the overall cost of goods sold. Without the need for rigorous heavy metal removal processes, which often require specialized scavengers and additional filtration units, the downstream processing becomes far more economical. The high yields reported, often exceeding 80%, mean that less starting material is wasted, improving the overall atom economy and reducing the environmental footprint associated with raw material sourcing and disposal.
- Enhanced Supply Chain Reliability: The reliance on cheap and easily obtainable raw materials such as methylquinolines and tryptophan derivatives ensures a stable supply chain that is less susceptible to geopolitical disruptions or supplier bottlenecks. Unlike specialized reagents that may have long lead times, the inputs for this reaction are commodity chemicals available from multiple global vendors. This diversification of supply sources enhances the resilience of the manufacturing process, ensuring consistent delivery schedules and reducing the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: The use of DMSO as a solvent and iodine as a reagent aligns well with green chemistry principles when managed correctly, as it avoids the generation of toxic heavy metal waste streams. The reaction conditions are mild enough to be safely scaled from gram to kilogram quantities without requiring specialized high-pressure equipment beyond standard reactors. This scalability facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing producers to respond quickly to market demand while maintaining compliance with increasingly strict environmental regulations regarding hazardous waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this process into existing production lines or new product development pipelines.
Q: What are the primary advantages of the iodine-catalyzed method over traditional multi-step syntheses?
A: The iodine-catalyzed method described in patent CN110776510A significantly reduces synthetic complexity by integrating multiple reaction steps into a single one-pot process. Unlike traditional methods that require isolation of unstable intermediates and harsh conditions, this approach utilizes mild temperatures and commercially available reagents, resulting in higher overall yields and reduced waste generation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up due to its operational simplicity and the use of robust reagents like DMSO and elemental iodine. The elimination of sensitive catalysts and the ability to tolerate various substituents on the quinoline and indole rings make it a reliable choice for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of elemental iodine in this reaction mechanism?
A: Elemental iodine serves a dual function as both a catalyst and an oxidant. It facilitates the initial functionalization of the methylquinoline to form reactive intermediates like 2-(iodomethyl)-quinoline and 2-formylquinoline in situ, and subsequently drives the oxidative dehydrogenation required to aromatize the final β-carboline core without needing external oxidizing agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitramarine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed one-pot synthesis for producing high-value β-carboline alkaloids. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Nitramarine or its derivatives meets the highest international standards for pharmaceutical applications. We are committed to leveraging this advanced technology to deliver superior quality intermediates that empower your drug discovery and development efforts.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for your proprietary derivatives. Let us be your trusted partner in bringing these potent bioactive molecules to the global market efficiently and sustainably.
