Scalable Manufacturing of High-Purity 2-Methyl-5-Pyrazine Formate via Novel Catalytic Route
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally compliant pathways for critical intermediates, and the synthesis of 2-methyl-5-pyrazine formate stands as a prime example of this evolution. As detailed in the groundbreaking patent CN109369545B, a novel four-step synthetic strategy has been developed that fundamentally restructures the production landscape for this vital hypoglycemic agent precursor. This innovative approach bypasses the historical reliance on hazardous oxidants and complex purification sequences, offering a streamlined route that begins with the condensation of methylglyoxal and 2-amino malonamide. For R&D directors and procurement specialists alike, this patent represents a significant leap forward, transitioning from pollution-heavy oxidation methods to a cleaner, high-yield sequence involving cyclization, hydrolysis, halogenation, and catalytic reduction. The implications for supply chain stability and cost efficiency in the manufacturing of antidiabetic drugs like glipizide are profound, marking a shift towards sustainable industrial chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-methyl-5-pyrazine formate has been plagued by severe environmental and efficiency bottlenecks, primarily stemming from two dominant synthetic strategies that are increasingly untenable for modern green manufacturing standards. The first conventional route involves the preparation of 2-methyl-5,6-dicarboxypyrazine followed by a decarboxylation step, often utilizing potassium permanganate as a harsh oxidant. This method not only suffers from inherently low yields during the oxidation and decarboxylation phases but also generates substantial quantities of manganese-containing waste residues, creating a heavy burden on wastewater treatment facilities and escalating disposal costs. Furthermore, the symmetry of the starting material 2,5-dimethylpyrazine in alternative oxidation routes leads to poor selectivity, resulting in significant formation of double oxidation byproducts and isolating yields as low as 5.8%, which is commercially disastrous for large-scale operations.

The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a paradigm shift by utilizing methylglyoxal and 2-amino malonamide as inexpensive, readily accessible feedstocks to construct the pyrazine ring directly. This new pathway elegantly sidesteps the need for destructive oxidation steps entirely, instead relying on a constructive cyclization followed by functional group manipulation. By avoiding the use of potassium permanganate, the process eliminates the generation of toxic manganese sludge, aligning perfectly with stringent global environmental regulations. Moreover, the strategic design of this route ensures high regioselectivity, effectively suppressing the formation of difficult-to-separate isomers that have traditionally plagued the purification process. The result is a synthesis that is not only chemically superior in terms of yield and purity but also economically advantageous due to the drastic reduction in waste treatment overhead and raw material costs.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this technological breakthrough lies in the precise control of reaction conditions during the initial cyclization and subsequent functionalization steps, which dictates the overall success of the manufacturing campaign. The process initiates with the reaction of methylglyoxal and 2-amino malonamide in an alkaline medium, specifically sodium hydroxide, maintained at a critically low temperature range of 5 to 10°C. This thermal control is paramount; operating within this narrow window suppresses the formation of the unwanted 2-formamide-3-hydroxy-6-methylpyrazine isomer, ensuring that the crude 3-hydroxy-5-methylpyrazine-2-formamide achieves an LC purity of not less than 95%. Following this, the intermediate undergoes hydrolysis in 50% sulfuric acid under reflux, converting the amide to the corresponding carboxylic acid with high efficiency. The subsequent halogenation step, typically employing thionyl chloride in a xylene solvent system with DMF catalysis, activates the hydroxyl position for the final reductive removal, creating a robust leaving group that facilitates the terminal transformation.
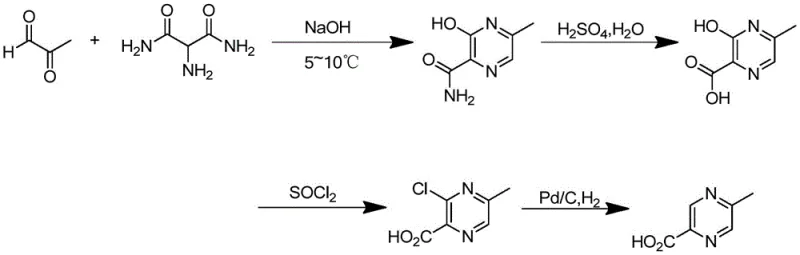
Impurity control is rigorously managed throughout this cascade, particularly during the final catalytic hydrogenation stage where the halogenated intermediate is reduced to the target molecule. The use of a palladium-on-carbon catalyst under controlled hydrogen pressure (1.8 to 2.2 MPa) and moderate temperatures (58-62°C) ensures complete dehalogenation without over-reduction of the pyrazine ring or other sensitive functionalities. The inclusion of an acid-binding agent, such as sodium hydroxide, during this reduction phase is a critical mechanistic detail that neutralizes the hydrogen halide byproduct in situ, preventing potential degradation of the product or corrosion of the reactor equipment. This careful orchestration of chemical steps results in a final product with an LC purity reaching 99%, demonstrating that high-quality pharmaceutical intermediates can be achieved through intelligent process design rather than exhaustive downstream purification.
How to Synthesize 2-Methyl-5-Pyrazine Formate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and reaction times to maximize yield and minimize byproduct formation. The protocol dictates a sequential addition of reagents where thermal management is the primary variable controlling selectivity, particularly in the initial exothermic cyclization. Operators must ensure the internal temperature remains between 5°C and 10°C during the base addition to prevent isomerization, followed by a controlled hydrolysis period of approximately 12 hours under acidic reflux. The subsequent halogenation and reduction steps require standard safety protocols for handling thionyl chloride and high-pressure hydrogenation, respectively, but offer straightforward workup procedures involving simple extraction and recrystallization. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the standardized procedure below.
- Cyclization of methylglyoxal and 2-amino malonamide in alkaline solution at 5-10°C to form 3-hydroxy-5-methylpyrazine-2-formamide.
- Acidic hydrolysis using 50% sulfuric acid under reflux to convert the formamide to 3-hydroxy-5-methylpyrazine-2-carboxylic acid.
- Halogenation with thionyl chloride in xylene/DMF followed by catalytic hydrogenation using Pd/C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational efficiencies and risk mitigation strategies that enhance the overall resilience of the supply network. By shifting away from the traditional permanganate oxidation methods, manufacturers can eliminate the complex and costly logistics associated with the disposal of heavy metal waste, which often constitutes a significant portion of the variable production costs in fine chemical manufacturing. The reliance on commodity chemicals like methylglyoxal and 2-amino malonamide ensures a stable and diversified supply base, reducing the vulnerability to price volatility often seen with specialized or scarce reagents like diaminomaleonitrile. Furthermore, the high intrinsic purity of the crude intermediates reduces the demand for extensive chromatographic purification, shortening the overall cycle time and increasing the throughput capacity of existing production facilities without the need for major capital expenditure.
- Cost Reduction in Manufacturing: The elimination of potassium permanganate and the associated waste treatment infrastructure leads to substantial cost savings, as the process no longer incurs the heavy financial penalties of managing hazardous manganese sludge. Additionally, the high yield and selectivity of the new route mean that less raw material is wasted on byproducts, effectively lowering the cost of goods sold (COGS) per kilogram of active pharmaceutical ingredient produced. The simplified purification workflow, driven by the high quality of the crude product, further reduces solvent consumption and energy usage, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly improved because the key starting materials are bulk chemicals with established global supply chains, unlike the niche precursors required for older methods. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages, providing a consistent and reliable flow of intermediates to downstream API manufacturers. The robustness of the process also means that scale-up from pilot to commercial production is more predictable, allowing for better inventory planning and shorter lead times for fulfilling large-volume orders from international pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard unit operations such as stirred tank reactors and filtration systems that are common in multipurpose chemical plants. From a regulatory standpoint, the absence of heavy metal oxidants simplifies the environmental permitting process and ensures compliance with increasingly strict global discharge standards, such as those imposed by the EPA and ECHA. This "green chemistry" profile not only future-proofs the manufacturing site against tightening regulations but also enhances the brand reputation of the supplier as a responsible partner in the sustainable development of pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers negotiating supply contracts. The answers reflect the consensus on how this methodology resolves historical pain points in pyrazine chemistry.
Q: Why is the new synthesis route for 2-methyl-5-pyrazine formate considered more environmentally friendly?
A: The novel process described in patent CN109369545B completely eliminates the use of potassium permanganate (KMnO4), a strong oxidant that generates massive amounts of hazardous manganese-containing waste residues and wastewater, thereby significantly reducing environmental treatment costs and compliance risks.
Q: How does the new method improve product purity compared to traditional oxidation routes?
A: By utilizing a controlled low-temperature cyclization at 5-10°C, the new method suppresses the formation of isomeric byproducts like 2-formamide-3-hydroxy-6-methylpyrazine, achieving crude purities exceeding 95% LC and final product purities of 99% without complex chromatographic separation.
Q: What are the key raw materials required for this scalable synthesis?
A: The process relies on cheap and readily available starting materials, specifically methylglyoxal and 2-amino malonamide, avoiding the use of expensive and difficult-to-synthesize precursors like diaminomaleonitrile found in older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-5-Pyrazine Formate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the long-term viability of the pharmaceutical supply chain. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN109369545B are fully realized in practical, industrial settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methyl-5-pyrazine formate meets the exacting standards required for the synthesis of life-saving medications like glipizide and acipimox. Our commitment to quality and consistency makes us a trusted partner for global innovators seeking to optimize their API manufacturing processes.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your competitive position in the global market.
