Advanced Synthesis of 5,5-Difluoro-3-Substituted Piperidine Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for fluorinated heterocycles, particularly 5,5-difluoro-3-substituted piperidine derivatives, due to their profound impact on metabolic stability and receptor binding affinity in modern drug candidates. Patent CN102731368A introduces a groundbreaking preparation method that addresses critical bottlenecks in the existing manufacturing landscape, offering a pathway that is both economically viable and technically superior for large-scale production. This innovation specifically targets the synthesis of complex piperidine scaffolds where the introduction of fluorine atoms at the 5-position has historically been plagued by low yields, hazardous reagents, and difficult purification processes. By leveraging a strategic sequence of reduction, dehydration, and catalytic hydrogenation, this technology enables the reliable production of high-purity pharmaceutical intermediates that are essential for the development of next-generation therapeutics targeting central nervous system disorders and other critical indications. The technical breakthrough lies not merely in the chemical transformation but in the holistic redesign of the process flow to prioritize safety, cost-efficiency, and environmental compliance, making it an ideal candidate for integration into global supply chains seeking reliable pharmaceutical intermediates supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
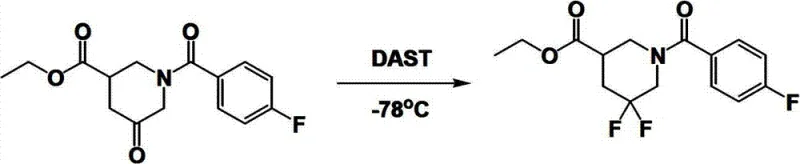
Historically, the synthesis of 5,5-difluoro piperidine structures has relied heavily on direct fluorination strategies using reagents such as DAST (Diethylaminosulfur Trifluoride), which present severe limitations for industrial application. As illustrated in prior art reaction schemes, the use of DAST often requires cryogenic conditions, such as minus 78 degrees Celsius, which imposes significant energy costs and operational complexity on manufacturing facilities. Furthermore, these conventional methods frequently generate alkene fluoride byproducts that are structurally similar to the desired product, making separation extremely difficult and resulting in substantial material loss during purification. The hazardous nature of fluorinating agents also introduces significant safety risks, requiring specialized containment and handling procedures that drive up capital expenditure and operational overhead. Additionally, alternative routes involving azide intermediates pose explosion hazards and require rigorous safety protocols, further complicating the scale-up process for commercial manufacturing. These cumulative factors render traditional methods unsuitable for the high-volume, cost-sensitive production required by the modern pharmaceutical market.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a mild, multi-step sequence that circumvents the need for dangerous fluorinating agents entirely. By starting with a pre-fluorinated ketone precursor, the process employs standard reduction chemistry to establish the necessary stereochemistry before proceeding to a controlled dehydration step. This strategy effectively bypasses the formation of problematic alkene fluoride impurities, ensuring a cleaner reaction profile that simplifies downstream processing. The use of catalytic hydrogenation for the final deprotection and saturation steps further enhances the green chemistry profile of the synthesis, utilizing hydrogen gas and palladium on carbon which are standard, safe, and easily manageable reagents in any GMP facility. This shift from hazardous direct fluorination to a constructive build-up approach represents a paradigm shift in process chemistry, offering a route that is inherently safer, more predictable, and significantly more amenable to optimization for commercial scale-up of complex pharmaceutical intermediates.
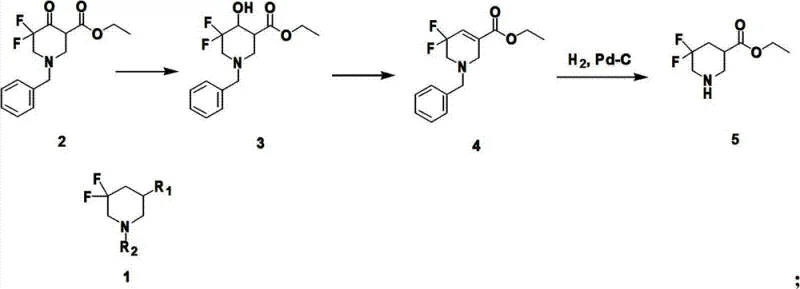
Mechanistic Insights into Pd-C Catalyzed Hydrogenation and Dehydration
The core of this synthetic innovation lies in the precise control of the dehydration and hydrogenation steps, which dictate the overall efficiency and purity of the final 5,5-difluoro-3-substituted piperidine derivative. The mechanism begins with the reduction of the 4-carbonyl group to a hydroxyl group, setting the stage for a regioselective elimination. This dehydration is achieved either through mesylation followed by base treatment or via direct acid-catalyzed elimination, both of which are carefully tuned to prevent over-reaction or degradation of the sensitive difluoro motif. The resulting tetrahydropyridine intermediate is then subjected to Pd-C catalysis under hydrogen pressure, which simultaneously removes the benzyl protecting group and saturates the double bond. This dual-function step is critical, as it consolidates two transformations into a single operational unit, reducing processing time and solvent consumption. The catalytic cycle ensures high turnover and selectivity, minimizing the formation of over-reduced or de-fluorinated side products that could compromise the quality of the active pharmaceutical ingredient. Understanding these mechanistic nuances is vital for R&D teams aiming to replicate this success in their own process development workflows.
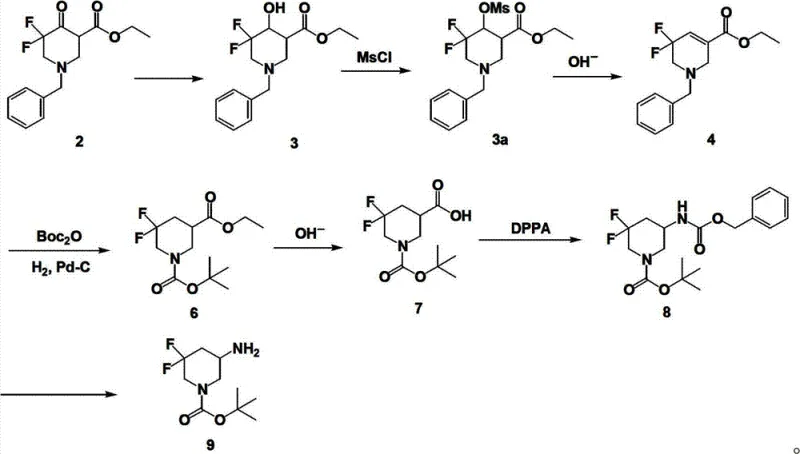
Furthermore, the versatility of this mechanism allows for the introduction of various protecting groups and substituents at the 3-position, enabling the synthesis of a diverse library of derivatives from a common intermediate. For instance, the pathway can be adapted to produce Boc-protected amines or Cbz-protected esters depending on the specific requirements of the downstream coupling reactions. This modularity is achieved by adjusting the reagents used in the final functionalization steps, such as using di-tert-butyl dicarbonate for Boc protection or benzyl chloroformate for Cbz protection. The ability to toggle between these protecting groups without altering the core fluorinated scaffold provides medicinal chemists with significant flexibility in designing synthesis routes for complex drug molecules. Impurity control is maintained throughout this process by leveraging the crystallinity of intermediates, allowing for purification via recrystallization rather than relying solely on chromatography, which is a key factor in reducing manufacturing costs and improving throughput for high-purity pharmaceutical intermediates.
How to Synthesize 5,5-Difluoro-3-Substituted Piperidine Efficiently
To implement this synthesis effectively, manufacturers must adhere to a standardized protocol that emphasizes temperature control and reagent quality to ensure consistent batch-to-batch performance. The process begins with the reduction of the ketone starting material using sodium borohydride in a suitable solvent system, followed by a carefully monitored dehydration step to generate the alkene intermediate. Detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, reaction times, and workup procedures required to achieve optimal yields. This structured approach ensures that the critical quality attributes of the intermediate are maintained, facilitating a smooth transition to the final hydrogenation and derivatization stages. By following these established parameters, production teams can minimize variability and maximize the efficiency of the overall manufacturing campaign.
- Reduce N-benzyl-5,5-difluoro-4-carbonyl-nipecotic acid ethyl ester using sodium borohydride to form the hydroxyl intermediate.
- Dehydrate the hydroxyl intermediate via mesylation and base treatment to generate the tetrahydropyridine alkene structure.
- Perform Pd-C catalyzed hydrogenation to remove the benzyl group and saturate the ring, yielding the core piperidine ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The elimination of expensive and hazardous fluorinating reagents like DAST translates directly into a significantly reduced cost of goods sold, as the raw material costs are lower and the safety infrastructure requirements are less burdensome. Moreover, the avoidance of difficult-to-separate byproducts means that yield losses during purification are drastically minimized, leading to higher overall material efficiency and reduced waste disposal costs. This efficiency gain is critical for maintaining competitive pricing in the global market for pharmaceutical intermediates, where margin pressure is constant. The use of common, commercially available reagents such as sodium borohydride and palladium on carbon also mitigates supply chain risks, as these materials are not subject to the same regulatory restrictions or availability fluctuations as specialized fluorinating agents. Consequently, manufacturers can secure a more stable and predictable supply of raw materials, ensuring continuity of production even in volatile market conditions.
- Cost Reduction in Manufacturing: The process eliminates the need for costly fluorinating reagents and reduces waste generation, leading to substantial cost savings in raw material procurement and waste management. By avoiding complex purification steps required to remove alkene fluoride byproducts, the overall processing time is shortened, which further lowers utility and labor costs associated with production. This economic efficiency makes the technology highly attractive for large-scale manufacturing where even marginal cost improvements can result in significant financial benefits over the product lifecycle.
- Enhanced Supply Chain Reliability: Relying on standard reagents like sodium borohydride and hydrogen gas ensures that the supply chain is not vulnerable to the shortages often associated with specialized fluorine chemistry reagents. This reliability allows for better production planning and inventory management, reducing the risk of delays that could impact downstream drug development timelines. The robustness of the process also means that technology transfer between manufacturing sites is smoother, as the required equipment and expertise are widely available in the contract manufacturing organization network.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous reagents make this process inherently safer and easier to scale from kilogram to tonne quantities without requiring major engineering modifications. Furthermore, the reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for manufacturing partners. This sustainability profile is becoming a key differentiator for suppliers seeking to partner with major pharmaceutical companies that prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity for stakeholders evaluating its potential for their specific projects. These answers are derived directly from the patent specifications and experimental data, ensuring that the information provided is accurate and relevant to real-world manufacturing scenarios. Understanding these details is crucial for making informed decisions about process adoption and supply chain integration.
Q: Why is this method superior to DAST fluorination for industrial production?
A: This method avoids the use of expensive and hazardous DAST reagents, eliminating the formation of difficult-to-separate alkene fluoride byproducts and significantly improving process safety and scalability.
Q: What is the purity profile of the resulting 5,5-difluoro piperidine derivatives?
A: The process yields a single major product with high selectivity, avoiding complex impurity profiles associated with direct fluorination, thus simplifying downstream purification and ensuring stringent purity specifications.
Q: Can this synthesis route be scaled for commercial tonnage production?
A: Yes, the reaction conditions are mild (0-80°C) and utilize common reagents like sodium borohydride and Pd-C, making the process highly suitable for large-scale commercial manufacturing without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,5-Difluoro-3-Substituted Piperidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from laboratory bench to full-scale manufacturing. Our team of expert chemists is well-versed in the nuances of fluorinated chemistry and can leverage this patented technology to deliver high-purity pharmaceutical intermediates that meet your stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the identity and quality of every batch, providing you with the confidence and documentation needed for regulatory filings. Our commitment to technical excellence and operational reliability makes us the ideal partner for companies seeking to secure a stable supply of critical building blocks for their drug development pipelines.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis tailored to your project volume and timeline. By collaborating with us, you can access specific COA data and route feasibility assessments that will help you optimize your supply chain and reduce time to market. Let us help you navigate the complexities of fine chemical manufacturing with a partner who understands the importance of quality, cost, and continuity in the pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
