Scalable Synthesis of Chiral Biotin Intermediates via Cinchona Alkaloid Catalysis for Global Supply Chains
Scalable Synthesis of Chiral Biotin Intermediates via Cinchona Alkaloid Catalysis for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to produce high-value chiral intermediates with absolute stereochemical control. Patent CN101157655A introduces a groundbreaking advancement in the synthesis of (4S, 5R)-half esters, which serve as the pivotal precursors for the industrial manufacturing of d-Biotin (Vitamin H). This technology leverages the power of asymmetric organocatalysis, specifically utilizing cinchona alkaloid derivatives to facilitate the enantioselective ring-opening of cyclic acid anhydrides. Unlike legacy processes that rely on inefficient resolution or expensive enzymatic pathways, this method operates under mild conditions ranging from -80°C to 25°C, delivering total chemical yields exceeding 90 percent and enantiomeric excess (ee) values greater than 98.5 percent. For R&D directors and supply chain managers, this represents a paradigm shift towards more sustainable, cost-effective, and scalable production of complex vitamin intermediates, ensuring a reliable supply chain for downstream nutraceutical and pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (4S, 5R)-half esters has been plagued by significant inefficiencies inherent to classical resolution strategies. Early methodologies, such as those reported by Gerecke et al., relied on the formation of diastereomeric salts using resolving agents like pseudoephedrine or dehydroabietamine, followed by tedious fractional crystallization. These processes are fundamentally flawed because they are limited by a maximum theoretical yield of only 50 percent for the desired enantiomer, necessitating the recycling or disposal of the unwanted isomer. Furthermore, the use of stoichiometric amounts of chiral auxiliaries or resolving agents drastically inflates raw material costs and generates substantial chemical waste. Alternative enzymatic approaches using pig liver esterase, while selective, often suffer from limitations in substrate loading and scalability, making them unsuitable for multi-ton commercial production. The cumulative effect of these drawbacks is a high cost of goods sold (COGS) and a fragile supply chain vulnerable to raw material fluctuations.
The Novel Approach
The methodology disclosed in CN101157655A circumvents these historical bottlenecks by employing a catalytic asymmetric alcoholysis strategy. Instead of resolving a racemic mixture, this process directly constructs the chiral center with high fidelity during the bond-forming event. The reaction involves the treatment of a cyclic anhydride (Formula II) with an alcohol in the presence of a sub-stoichiometric amount of a chiral cinchona alkaloid catalyst. This catalytic cycle allows for the continuous turnover of the chiral inducer, meaning a small quantity of catalyst can generate a large volume of product, thereby decoupling production capacity from the cost of chiral materials. The result is a streamlined workflow that eliminates the need for multiple crystallization steps and resolving agent recovery, significantly simplifying the unit operations required for manufacturing. This direct route not only improves the overall mass balance but also enhances the environmental profile of the synthesis by reducing solvent usage and waste generation.

Mechanistic Insights into Cinchona Alkaloid-Catalyzed Asymmetric Alcoholysis
The success of this synthetic route hinges on the precise molecular recognition and activation capabilities of the cinchona alkaloid catalysts. These naturally derived scaffolds, such as quinine and cinchonidine derivatives (Structure A), possess a rigid bicyclic framework that creates a well-defined chiral environment around the active site. During the reaction, the basic nitrogen atom of the quinoline ring likely activates the alcohol nucleophile through hydrogen bonding or transient ion-pair formation, while the bulky substituents on the catalyst shield one face of the planar cyclic anhydride electrophile. This steric differentiation forces the nucleophilic attack to occur exclusively from the less hindered face, resulting in the formation of the (4S, 5R) configuration with exceptional stereocontrol. The ability to fine-tune the catalyst structure by modifying the R3 and R4 groups (as seen in Structure A) allows chemists to optimize the steric and electronic properties for specific substrates, ensuring that the transition state energy for the desired enantiomer is significantly lower than that of the undesired one.
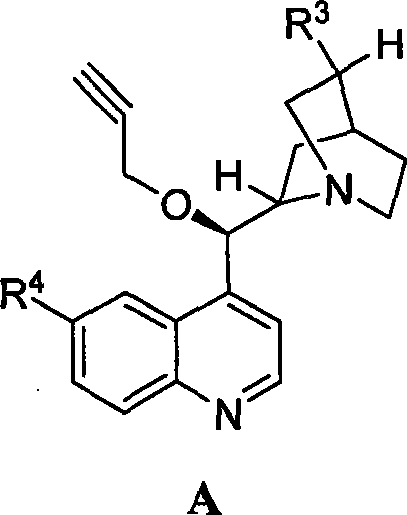
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic methods. Because the chirality is induced kinetically during the ring-opening step, the formation of the opposite enantiomer is suppressed at the source rather than being removed later. This minimizes the burden on downstream purification processes, such as chromatography or repeated recrystallization, which are often required to upgrade the optical purity of resolved mixtures. Furthermore, the mild reaction temperatures (-60°C to -20°C) prevent thermal degradation of the sensitive anhydride ring and minimize side reactions such as polymerization or over-esterification. The robustness of the catalyst also means that it remains stable throughout the reaction duration of 10 to 80 hours, maintaining consistent selectivity without decomposing into species that could contaminate the final product. This high level of process control is essential for meeting the stringent purity specifications required for pharmaceutical intermediates intended for human consumption.
How to Synthesize (4S,5R)-Half Esters Efficiently
Implementing this asymmetric synthesis requires careful attention to reaction parameters to maximize both yield and optical purity. The process begins with the preparation of a dry reaction vessel charged with the cyclic anhydride substrate and the selected cinchona alkaloid catalyst in a suitable organic solvent system, such as a mixture of toluene and carbon tetrachloride. Precise temperature control is critical; the mixture must be cooled to the optimal range of -40°C to -50°C before the slow, dropwise addition of the alcohol reactant to manage the exotherm and maintain stereochemical integrity. Following the addition, the reaction is allowed to stir for an extended period, typically between 60 to 72 hours, to ensure complete conversion of the starting material. Upon completion, the workup involves solvent removal under reduced pressure, followed by an acidic wash to remove basic impurities and catalyst residues. The crude product is then purified via recrystallization from solvents like benzene or toluene to afford the final white crystalline powder with high optical rotation values.
- Prepare the reaction mixture by combining cyclic anhydride II, a specific cinchona alkaloid derivative (e.g., 9-propargyl quinine), and an organic solvent such as toluene or carbon tetrachloride in a dry reactor.
- Cool the reaction system to a temperature range between -80°C and 25°C, preferably maintaining -40°C to -50°C, before slowly adding the alcohol reactant (e.g., methanol) dropwise.
- Stir the mixture for 10 to 80 hours to ensure complete conversion, followed by solvent recovery, acid wash, and recrystallization to isolate the (4S,5R)-half ester with >98.5% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond simple laboratory metrics. The primary economic driver is the drastic reduction in raw material costs associated with the elimination of stoichiometric chiral resolving agents. In traditional resolution processes, more than half of the synthesized material is often discarded or requires expensive recycling, whereas this catalytic method utilizes nearly all starting materials to form the desired product, leading to substantial cost savings in manufacturing. Additionally, the quantitative recovery of the expensive cinchona alkaloid catalyst further lowers the operational expenditure, as the catalyst can be reclaimed from the aqueous layer simply by pH adjustment and filtration. This closed-loop material flow reduces dependency on volatile chiral reagent markets and stabilizes the long-term cost structure of the supply chain.
- Cost Reduction in Manufacturing: The transition from resolution-based methods to direct asymmetric catalysis fundamentally alters the cost equation by removing the 50 percent yield ceiling inherent to racemic splitting. By achieving total yields greater than 90 percent, the effective cost per kilogram of the active intermediate is nearly halved compared to legacy processes. Furthermore, the simplification of the downstream processing—eliminating multiple crystallization steps and salt formation/breaking cycles—reduces labor hours, energy consumption, and solvent usage. These efficiencies compound to offer a highly competitive pricing structure for high-purity pharmaceutical intermediates, allowing buyers to secure better margins in their final vitamin formulations.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step synthesis routes that rely on specialized reagents. This patented method utilizes readily available raw materials, including common alcohols like methanol and commercially sourced cinchona alkaloids, which mitigates the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of pressures and standard organic solvents, ensures that production can be maintained consistently across different manufacturing sites without requiring exotic equipment. This flexibility allows for easier technology transfer and scale-up, ensuring that procurement teams can rely on a steady flow of materials to meet global demand for biotin and related nutraceuticals.
- Scalability and Environmental Compliance: As regulatory pressures regarding chemical waste and environmental impact intensify, this green chemistry approach offers a compliant pathway for industrial expansion. The high atom economy of the reaction means less waste is generated per unit of product, simplifying effluent treatment and reducing disposal costs. The ability to operate at near-ambient pressures and moderate sub-zero temperatures reduces the energy load on cooling systems compared to cryogenic processes. Moreover, the recyclability of both the solvent systems and the catalyst aligns with modern sustainability goals, making this process attractive for companies aiming to reduce their carbon footprint while scaling up production of complex chiral intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and claims within patent CN101157655A, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D teams planning pilot runs and for quality assurance teams establishing specification limits.
Q: What represents the primary advantage of this cinchona alkaloid method over traditional resolution techniques?
A: Unlike traditional resolution methods which suffer from a maximum theoretical yield of 50% and require cumbersome crystallization steps, this asymmetric catalytic approach achieves total yields exceeding 90% with enantiomeric excess greater than 98.5%, effectively doubling production efficiency while eliminating the need for expensive chiral resolving agents.
Q: Is the chiral catalyst recoverable for industrial scale-up?
A: Yes, a critical feature of this process described in patent CN101157655A is the quantitative recovery of the cinchona alkaloid catalyst. By adjusting the pH of the aqueous layer post-reaction, the catalyst precipitates and can be filtered and reused, significantly reducing the cost of goods sold (COGS) for large-scale manufacturing.
Q: What are the optimal reaction conditions for maximizing stereoselectivity?
A: The patent data indicates that maintaining low temperatures between -60°C and -20°C is crucial for high stereocontrol. Additionally, using mixed solvent systems like toluene and carbon tetrachloride, along with specific catalyst derivatives like 9-propargyl quinine, ensures the reaction proceeds with minimal racemization and optimal optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4S,5R)-Half Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team specializes in the process development and scale-up of complex chiral intermediates, including the (4S,5R)-half esters critical for the global biotin market. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and enantioselectivity demonstrated in the lab are faithfully reproduced in our manufacturing plants. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, including precise control over optical rotation and impurity profiles, giving our partners confidence in every batch we deliver.
We invite you to collaborate with us to leverage this advanced catalytic technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate our commitment to quality and reliability. Let us help you secure a sustainable and cost-effective source of high-purity pharmaceutical intermediates for your next generation of products.
