Scalable Metal-Free Ring-Opening of Cyclic Ketones for High-Value Pharmaceutical Intermediates
Scalable Metal-Free Ring-Opening of Cyclic Ketones for High-Value Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving towards greener, more economical methodologies, particularly in the production of complex pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN115611726A, which discloses a novel method for the ring-opening of cyclic ketone compounds mediated by nitrous compounds. This technology addresses a long-standing challenge in organic chemistry: the efficient cleavage of unstrained carbon-carbon single bonds adjacent to carbonyl groups without relying on expensive transition metals or hazardous strong oxidants. By utilizing a catalytic system based on sodium nitrite and concentrated hydrochloric acid, this process achieves the高效 conversion of cyclic ketones into valuable linear keto-acids under remarkably mild conditions. For R&D directors and procurement managers seeking reliable supply chains for high-purity API intermediates, this metal-free approach represents a paradigm shift, offering a pathway to reduce raw material costs while enhancing process safety and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cleavage of carbon-carbon bonds in six-membered rings has been a formidable task due to the thermodynamic stability of unstrained C-C single bonds. Traditional strategies, such as the Baeyer-Villiger oxidation or the Haloform reaction, have served as the primary tools for modifying cyclic ketone frameworks. However, these legacy methods suffer from significant drawbacks that hinder their applicability in modern, large-scale manufacturing. The Baeyer-Villiger reaction, for instance, typically necessitates the use of peracids or hydrogen peroxide with Lewis acid catalysts, posing serious safety risks regarding explosion and handling of unstable oxidants. Furthermore, alternative methods often rely heavily on transition metal catalysts (such as Rhodium, Palladium, or Ruthenium complexes) to activate the C-C bond. These metals are not only prohibitively expensive but also introduce severe contamination risks. For a reliable pharmaceutical intermediate supplier, the presence of residual heavy metals in the final product is a critical failure point, necessitating costly and time-consuming purification steps like scavenging or recrystallization to meet stringent regulatory limits (e.g., ICH Q3D guidelines).
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in CN115611726A offers a streamlined, metal-free alternative that leverages the reactivity of nitrous acid generated in situ. The process initiates by mixing the cyclic ketone substrate with a nitroso compound, specifically sodium nitrite, in a solvent such as tetrahydrofuran (THF). Upon the dropwise addition of concentrated hydrochloric acid at low temperatures (-5°C), the reaction generates the active nitrosating species which facilitates the cleavage of the C-C bond adjacent to the carbonyl group. This novel approach eliminates the need for transition metals entirely, thereby removing the associated supply chain volatility of precious metals and the downstream burden of metal removal. The reaction proceeds smoothly at room temperature after the initial low-temperature initiation, demonstrating robust operational simplicity. This shift from metal-catalyzed to organocatalytic-style conditions drastically simplifies the work-up procedure, allowing for direct extraction and purification, which translates directly into reduced processing time and lower operational expenditures for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Nitrous Acid Mediated C-C Bond Cleavage
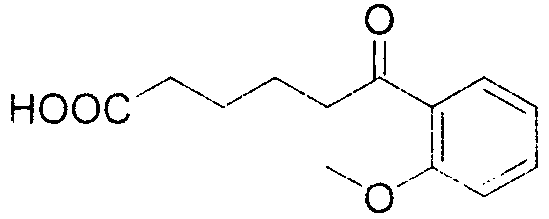
The mechanistic elegance of this transformation lies in the activation of the alpha-position of the ketone through nitrosation. Under acidic conditions, sodium nitrite reacts to form nitrous acid, which subsequently generates the nitrosonium ion (NO+). This electrophilic species attacks the enol tautomer of the cyclic ketone, forming an alpha-nitroso ketone intermediate. This intermediate is highly reactive and undergoes a rearrangement or hydrolysis sequence that ultimately leads to the rupture of the carbon-carbon bond. Unlike radical-based C-C cleavage methods that often lack selectivity and produce complex mixtures of by-products, this ionic pathway is highly controlled. The presence of the carbonyl group activates the adjacent bond, and the introduction of the nitrogenous moiety serves as a leaving group driver, facilitating the ring opening to yield the linear dicarbonyl or keto-acid structure. This mechanism ensures high regioselectivity, preserving the integrity of sensitive functional groups on the aromatic ring, such as methoxy or thioether substituents, which might otherwise be oxidized by harsher traditional reagents.
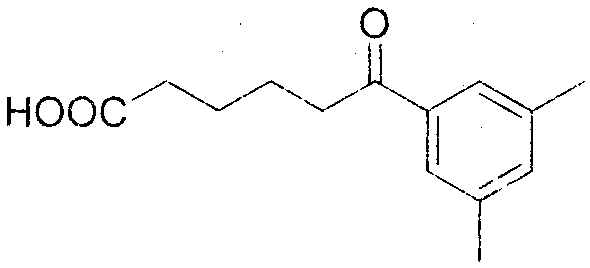
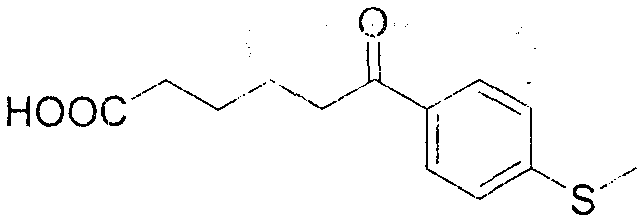
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material precursors or pharmaceutical building blocks. Because the reaction avoids free-radical chains and high-temperature thermal degradation, the formation of polymeric tars or over-oxidized side products is minimized. The patent data indicates that substrates with diverse electronic properties, including electron-donating groups like methoxy and methyl, as well as sulfur-containing groups, react smoothly. For instance, the conversion of 2-methoxyphenylcyclohexanone proceeds with a yield of 75% and HPLC purity of 98%, demonstrating the method's tolerance to ortho-substitution. Similarly, substrates with meta-substitution patterns, such as 3,5-dimethylphenylcyclohexanone, achieve even higher yields of 83%. This broad substrate scope confirms that the electronic nature of the aromatic ring does not significantly impede the nitrosation-driven cleavage, making it a versatile tool for synthesizing a wide array of functionalized keto-acids.
How to Synthesize Keto-Acid Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize side reactions, although the overall protocol is operationally simple. The standard procedure involves dissolving the cyclic ketone and sodium nitrite in THF, cooling to -5°C, and carefully adding concentrated HCl. The reaction is then allowed to warm to room temperature and stir for 6 hours. Detailed standard operating procedures (SOPs) regarding stoichiometry, quenching, and isolation are critical for reproducibility. For process chemists looking to adopt this technology, understanding the nuances of the work-up—specifically the extraction efficiency and column chromatography conditions—is key to achieving the reported 98% purity levels. The detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for scaling this reaction from gram-scale laboratory optimization to multi-kilogram pilot production.
- Dissolve the cyclic ketone substrate and sodium nitrite (1.0-1.2 equiv) in tetrahydrofuran (THF) and cool the mixture to -5°C.
- Slowly add concentrated hydrochloric acid (2.0 equiv) dropwise to the reaction mixture while maintaining low temperature, then allow to warm to room temperature.
- Stir the reaction for 6 hours at room temperature, quench with water, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitrous acid-mediated ring-opening technology offers compelling economic and logistical benefits. The primary driver for cost optimization is the complete elimination of transition metal catalysts. In traditional processes, the cost of the catalyst itself, combined with the specialized ligands and the extensive purification required to remove trace metals, constitutes a significant portion of the Cost of Goods Sold (COGS). By switching to sodium nitrite and hydrochloric acid—two of the most abundant and inexpensive industrial chemicals—the raw material cost base is drastically lowered. Furthermore, the simplified purification process reduces the consumption of solvents and silica gel, contributing to substantial cost savings in waste management and material usage. This aligns perfectly with the strategic goal of cost reduction in pharmaceutical intermediate manufacturing, allowing companies to maintain healthy margins even in competitive markets.
- Cost Reduction in Manufacturing: The economic impact of removing precious metals cannot be overstated. Traditional C-C activation often relies on Palladium or Rhodium, prices for which are subject to extreme market volatility. By utilizing a base-metal-free system, manufacturers insulate their supply chain from these fluctuations. Additionally, the reaction conditions are mild (starting at -5°C and proceeding at room temperature), which significantly reduces energy consumption compared to processes requiring prolonged heating or cryogenic cooling below -40°C. The high atom economy and minimal by-product formation mean that less raw material is wasted, further enhancing the overall process efficiency and profitability.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can sometimes lead to bottlenecks, especially during global supply disruptions. In contrast, sodium nitrite and hydrochloric acid are commodity chemicals available from multiple suppliers globally, ensuring a robust and resilient supply chain. The simplicity of the reagents also means that quality control is straightforward, reducing the risk of batch-to-batch variability caused by catalyst degradation or ligand impurities. This reliability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: As regulatory pressure mounts to reduce the environmental footprint of chemical manufacturing, this green chemistry approach offers a distinct advantage. The absence of heavy metals simplifies wastewater treatment and eliminates the need for hazardous waste disposal associated with spent metal catalysts. The process generates primarily saline waste and organic solvents which can be readily recycled. This ease of waste handling facilitates smoother regulatory approvals and supports sustainability goals. Moreover, the reaction has been demonstrated to scale effectively with consistent yields, indicating that the commercial scale-up of complex keto-acid derivatives is feasible without encountering the heat transfer or mixing issues common in exothermic oxidations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ring-opening technology. These insights are derived directly from the experimental data and beneficial effects outlined in patent CN115611726A, providing clarity on how this method compares to existing industrial standards. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios for high-value intermediates.
Q: What are the advantages of this nitrous acid method over traditional Baeyer-Villiger oxidation?
A: Unlike Baeyer-Villiger oxidation which often requires hazardous peracids or expensive transition metal catalysts, this method utilizes inexpensive sodium nitrite and hydrochloric acid. It operates under mild conditions (-5°C to RT) and avoids the generation of heavy metal waste, significantly simplifying downstream purification and reducing environmental impact.
Q: What is the substrate scope for this ring-opening reaction?
A: The protocol demonstrates excellent compatibility with various substituted cyclic ketones, including ortho-substituted phenyl cyclohexanones, dimethyl-substituted variants, and thioether-containing substrates. Both electron-rich and electron-deficient aromatic rings are tolerated, yielding the corresponding linear keto-acids with high purity (up to 98% by HPLC).
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to the use of commodity chemicals (NaNO2, HCl) and the absence of sensitive transition metal catalysts. The reaction proceeds efficiently at room temperature after initiation, minimizing energy consumption for heating or cryogenic cooling, which is critical for cost-effective manufacturing at the 100 MT scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Keto-Acid Derivatives Supplier
The technological potential of metal-free C-C bond cleavage is immense, offering a cleaner and more economical route to essential building blocks for the life sciences industry. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of keto-acid derivatives meets the highest international standards. We understand the critical nature of supply continuity and quality consistency in the pharmaceutical sector, and our team is dedicated to delivering solutions that optimize both performance and cost.
We invite you to explore how this advanced synthesis method can enhance your product portfolio. Whether you require custom synthesis of specific cyclic ketone derivatives or a full Customized Cost-Saving Analysis for your current supply chain, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to drive efficiency and innovation in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
