Scalable Synthesis of Ginsenoside Rh3: Advanced Catalytic Strategies for Commercial Production
Scalable Synthesis of Ginsenoside Rh3: Advanced Catalytic Strategies for Commercial Production
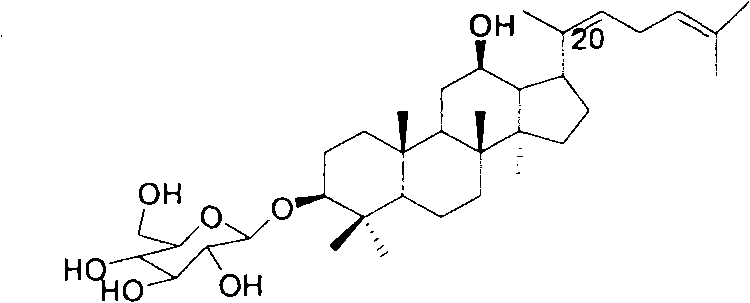
The global demand for rare ginsenosides has surged due to their potent pharmacological profiles, ranging from anti-tumor activity to neuroprotection. Among these, Ginsenoside Rh3 stands out as a critical bioactive marker, yet its natural abundance in Panax ginseng is exceedingly low, necessitating robust semi-synthetic alternatives. A pivotal advancement in this domain is detailed in patent CN102336798A, which outlines a highly efficient, four-step synthetic pathway starting from the readily available Protopanaxadiol. This methodology represents a significant leap forward in process chemistry, addressing the historical bottlenecks of cost and stereoselectivity that have plagued the commercialization of rare saponins. By shifting away from traditional homogeneous catalysis towards greener solid-acid systems, this route offers a compelling value proposition for pharmaceutical manufacturers seeking reliable supply chains for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of ginsenoside analogues has been hindered by reliance on stoichiometric amounts of expensive promoters, particularly silver salts such as silver carbonate. These traditional protocols, while effective on a milligram scale, present severe economic and operational barriers when translated to industrial manufacturing. The primary drawback lies in the cost structure; silver-based reagents are not only prohibitively expensive but also generate substantial heavy metal waste, complicating environmental compliance and disposal logistics. Furthermore, these methods often struggle with stereochemical control at the anomeric center, frequently producing mixtures of alpha and beta glycosidic linkages that require tedious and yield-eroding chromatographic separations. The inability to consistently secure the biologically active beta-configuration without significant loss of material renders these legacy processes economically unviable for large-scale production of rare ginsenosides like Rh3.
The Novel Approach
In stark contrast, the innovative pathway described in the reference patent utilizes a strategic combination of selective protection, dehydration, and solid-acid catalyzed glycosylation to overcome these hurdles. By employing Lewis acids or heterogeneous solid acids like Amberlyst-15 and Nafion-H, the process eliminates the need for costly silver promoters entirely. This shift not only drastically reduces raw material costs but also simplifies the workup procedure, as solid catalysts can be removed via simple filtration rather than complex extraction or quenching sequences. The integration of molecular sieves further enhances the reaction efficiency by scavenging water, driving the equilibrium towards the desired glycoside formation. This streamlined approach ensures high selectivity for the beta-configuration, thereby maximizing the yield of the therapeutically relevant isomer and minimizing downstream purification burdens.
Mechanistic Insights into Solid-Acid Catalyzed Glycosylation
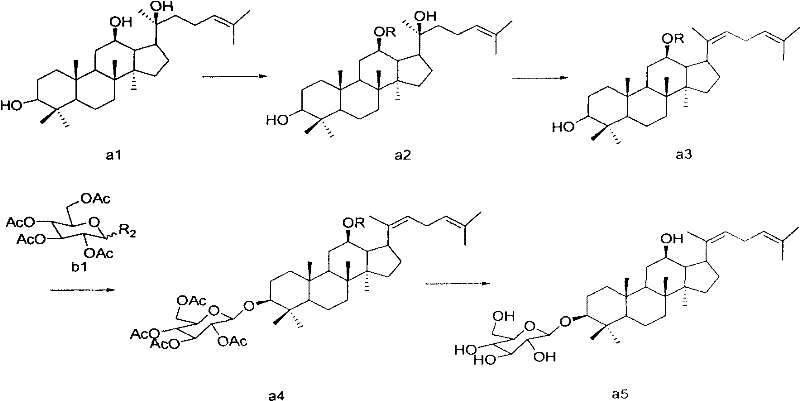
The cornerstone of this synthetic success lies in the mechanistic precision of the glycosylation step, where the interaction between the glucose donor and the aglycone acceptor is meticulously controlled. In the presence of a solid acid catalyst and molecular sieves, the reaction proceeds through a highly organized transition state that favors the formation of the 1,2-trans-glycosidic bond. The solid acid activates the leaving group on the sugar donor, generating an oxocarbenium ion intermediate that is rapidly attacked by the hydroxyl group at the C-3 position of the diene-aglycone. Crucially, the steric environment created by the protecting groups at the C-12 position and the rigid steroid backbone directs the nucleophilic attack from the less hindered face, ensuring high stereoselectivity. This level of control is paramount for maintaining the biological integrity of the final molecule, as the spatial arrangement of the sugar moiety directly influences receptor binding affinity and metabolic stability in vivo.
Beyond stereocontrol, the process incorporates robust mechanisms for impurity management, which is a critical concern for R&D directors focused on regulatory compliance. The initial protection of the 12-hydroxyl group serves a dual purpose: it prevents unwanted side reactions at this site during the subsequent acidic dehydration and glycosylation steps, and it acts as a temporary handle to modulate the solubility and crystallinity of intermediates. By selectively masking this functionality, the synthesis avoids the formation of diglycosylated byproducts or rearrangement products that commonly plague unprotected saponin chemistry. Furthermore, the final deprotection step under mild alkaline conditions is designed to be orthogonal, removing the acyl groups without affecting the sensitive diene system or the newly formed glycosidic bond. This chemoselectivity ensures that the final impurity profile is clean, facilitating easier validation and faster time-to-market for drug development programs.
How to Synthesize Ginsenoside Rh3 Efficiently
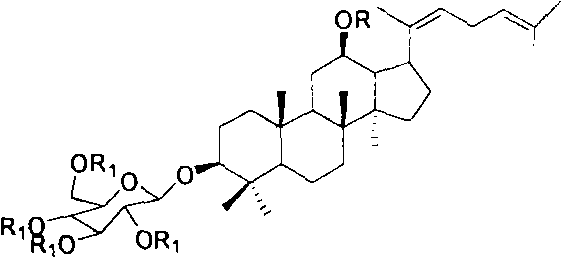
Executing this synthesis requires precise adherence to the optimized reaction parameters defined in the technical literature to ensure reproducibility and high yield. The process begins with the selective acylation of Protopanaxadiol, followed by a controlled dehydration to establish the conjugated diene system essential for Rh3 identity. The subsequent glycosylation is the most critical phase, demanding strict moisture control and the correct loading of solid acid catalyst to maintain reaction kinetics. While the general workflow is straightforward, the devil is in the details regarding solvent choice, temperature ramps, and quenching protocols. For process chemists looking to implement this route, understanding the nuances of each transformation is key to successful technology transfer. The detailed standardized synthesis steps are outlined below to guide your laboratory or pilot plant operations.
- Selective protection of the 12-hydroxyl group on Protopanaxadiol using acyl chlorides or anhydrides to form 12-substituted intermediates.
- Acid-catalyzed dehydration of the 20-hydroxyl group to generate the critical 20(22)-diene system.
- Stereoselective glycosylation using a glucose donor and solid acid catalyst (e.g., Amberlyst-15) with molecular sieves.
- Final deprotection of the ester groups under alkaline conditions to yield high-purity Ginsenoside Rh3.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic advantages beyond mere technical feasibility. The elimination of precious metal catalysts fundamentally alters the cost structure of the manufacturing process, removing exposure to the volatile pricing of silver and reducing the capital tied up in reagent inventory. Moreover, the simplified purification workflow means that production cycles are shorter, allowing for faster turnover and improved responsiveness to market demand fluctuations. The use of robust, commercially available solid acids also mitigates supply risk, as these materials are sourced from stable industrial supply chains rather than niche specialty chemical vendors. This resilience is crucial for maintaining continuity of supply in the face of global logistical disruptions, ensuring that downstream drug manufacturers receive their critical intermediates without delay.
- Cost Reduction in Manufacturing: The substitution of expensive silver carbonate with reusable solid acid catalysts results in substantial cost savings per kilogram of finished product. By avoiding the need for extensive heavy metal scavenging and removal processes, manufacturers can significantly lower their operational expenditures related to waste treatment and quality control testing. The high atom economy of the glycosylation step further contributes to overall process efficiency, ensuring that a greater proportion of raw materials are converted into saleable product rather than lost as waste. These cumulative efficiencies create a more competitive cost base, allowing for better margin management in the final pharmaceutical formulation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acyl chlorides, common organic solvents, and heterogeneous catalysts ensures that the supply chain is not dependent on single-source or geographically constrained raw materials. This diversification of input materials reduces the risk of production stoppages due to raw material shortages. Additionally, the mild reaction conditions reduce the stress on manufacturing equipment, leading to lower maintenance requirements and higher asset availability. For supply chain heads, this means a more predictable production schedule and the ability to commit to longer-term delivery contracts with confidence, knowing that the underlying chemistry is robust and scalable.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a markedly greener profile compared to traditional methods. The absence of heavy metal waste streams simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. The solid acid catalysts can potentially be regenerated and reused multiple times, aligning with principles of green chemistry and sustainability goals. This environmental compatibility not only facilitates easier permitting for new manufacturing facilities but also enhances the corporate social responsibility profile of the supply chain, a factor increasingly weighted by global pharmaceutical buyers during vendor audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of Ginsenoside Rh3. These insights are derived directly from the patented methodology and practical experience in scaling similar saponin syntheses. Understanding these aspects helps stakeholders make informed decisions regarding process adoption and vendor selection. The answers reflect a commitment to transparency and technical accuracy, ensuring that all partners have a clear view of the capabilities and limitations of this advanced synthetic route.
Q: Why is solid acid catalysis preferred over silver carbonate for Ginsenoside Rh3 synthesis?
A: Traditional methods utilizing silver carbonate suffer from high reagent costs and poor stereoselectivity, often yielding mixtures of alpha and beta anomers. The novel solid acid approach eliminates expensive heavy metals, drastically simplifies post-reaction purification, and ensures high beta-configuration selectivity essential for biological activity.
Q: What is the expected purity profile for industrially produced Ginsenoside Rh3 using this route?
A: By employing recrystallization techniques on the final deprotected product, the process consistently achieves high purity levels suitable for pharmaceutical applications. The specific elimination of side reactions during the glycosylation step minimizes difficult-to-remove impurities, streamlining the quality control process.
Q: Is this synthetic pathway scalable for metric-ton production?
A: Yes, the methodology is explicitly designed for industrial suitability. The use of heterogeneous solid acid catalysts allows for easier separation and potential catalyst recycling, while the mild reaction conditions reduce energy consumption and safety risks associated with large-scale exothermic events.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ginsenoside Rh3 Supplier
The synthetic pathway for Ginsenoside Rh3 described herein exemplifies the type of sophisticated chemistry that NINGBO INNO PHARMCHEM specializes in bringing to commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of handling complex natural product derivatives, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications. We understand that consistency is key, and our process engineering team is dedicated to optimizing every unit operation to maximize yield and minimize variability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can be integrated into your supply chain. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need to validate the feasibility of this route for your specific API needs, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your project milestones. Let us collaborate to secure a sustainable and cost-effective supply of this critical intermediate for your next-generation therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
