Scalable Manufacturing of High-Purity Garcinoside via Novel Catalytic Esterification
Scalable Manufacturing of High-Purity Garcinoside via Novel Catalytic Esterification
The pharmaceutical industry constantly seeks robust synthetic routes for potent anticancer agents, and the production technology detailed in patent CN102532212B represents a significant leap forward in the manufacturing of Garcinoside. This innovative process addresses the critical bottlenecks of traditional synthesis by utilizing a mild, three-step catalytic sequence that replaces labor-intensive purification methods with efficient extraction and crystallization techniques. By leveraging specific condensing agents like EDCI and precise deacetylation reagents, this methodology ensures that the delicate polyprenylated xanthone core of Gambogic Acid remains intact while achieving exceptional purity levels suitable for clinical applications. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex glycosides like Garcinoside has been plagued by reliance on silica gel column chromatography for purification, a method that is inherently inefficient for industrial scale-up. Traditional protocols often suffer from prolonged production cycles due to the slow elution rates and extensive solvent consumption required to separate the target compound from impurities and by-products. Furthermore, the interaction of sensitive natural product derivatives with acidic silica surfaces can lead to decomposition or isomerization, resulting in reduced overall yields and compromised product quality. These operational inefficiencies translate directly into higher manufacturing costs and unpredictable lead times, creating substantial risks for supply chain managers who require consistent volumes of active pharmaceutical ingredients for downstream drug development.
The Novel Approach
In stark contrast, the novel approach outlined in the patent introduces a streamlined workflow that completely eliminates the need for silica gel column separation,取而代之 by a sophisticated combination of liquid-liquid extraction and recrystallization. This strategic shift allows for the rapid isolation of intermediates such as Methyl Gambogic Acid and Tetraacetyl Garcinoside with high recovery rates, drastically shortening the total production time. The use of mild reaction conditions, specifically maintaining temperatures between 10–70°C under nitrogen protection, preserves the structural integrity of the molecule while facilitating high conversion rates. This methodological overhaul not only enhances the economic viability of the process but also aligns with modern green chemistry principles by reducing solvent waste, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Catalytic Esterification and Glycosylation
The core of this synthetic breakthrough lies in the precise control of reaction kinetics during the esterification and glycosylation steps, which are critical for maintaining the stereochemistry and functionality of the final product. In the initial step, Gambogic Acid undergoes esterification with methanol in the presence of condensing agents such as 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI) or N,N'-dicyclohexylcarbodiimide (DCC), coupled with catalysts like 4-dimethylaminopyridine (DMAP). This activation strategy ensures the efficient formation of the methyl ester without inducing side reactions on the sensitive conjugated system. Subsequently, the introduction of the sugar moiety via reaction with 4-(3,4,5,6-tetra-O-acetyl-β-D-allopyranosyl)benzoyl chloride is meticulously controlled at 0-40°C in dichloromethane, preventing thermal degradation while ensuring complete acylation of the phenolic hydroxyl group.
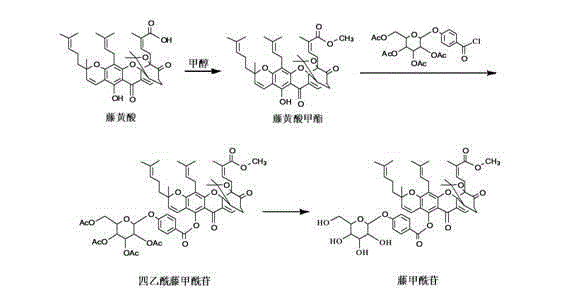
Following the formation of the tetraacetyl intermediate, the final deacetylation step is executed using reagents such as sodium methoxide or dibutyltin oxide in a methanol solvent at moderate temperatures of 20-65°C. This specific choice of deacetylation conditions is crucial for selectively removing the acetyl protecting groups without hydrolyzing the newly formed glycosidic bond or the methyl ester. The mechanism relies on the nucleophilic attack of the alkoxide on the carbonyl carbon of the acetate groups, regenerating the free hydroxyls on the sugar ring which are essential for the compound's water solubility and bioavailability. The subsequent purification via activated carbon adsorption and recrystallization effectively removes trace metal catalysts and organic impurities, resulting in a final product with purity exceeding 99.8%, a specification that meets the rigorous demands of high-purity pharmaceutical intermediates.
How to Synthesize Garcinoside Efficiently
The synthesis of Garcinoside described in this patent offers a reproducible and scalable pathway that can be readily adapted for commercial production facilities. The process begins with the activation of Gambogic Acid followed by sequential functionalization and deprotection, each step optimized for maximum yield and minimal environmental impact. Operators must strictly adhere to the specified temperature ranges and nitrogen atmospheres to prevent oxidation of the prenyl chains, which are susceptible to degradation under harsh conditions. The detailed standardized synthesis steps below outline the precise stoichiometry and workup procedures required to achieve the reported high yields and purity specifications.
- Perform esterification of Gambogic Acid with methanol using EDCI/DMAP catalysts at 10-70°C to form Methyl Gambogic Acid.
- React Methyl Gambogic Acid with tetra-O-acetyl sugar chloride in dichloromethane at 0-40°C to synthesize Tetraacetyl Garcinoside.
- Execute deacetylation using sodium methoxide or dibutyltin oxide in methanol at 20-65°C, followed by recrystallization to achieve >99.8% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional chromatographic methods to this extraction-based protocol offers profound economic and logistical benefits that extend far beyond simple yield improvements. The elimination of silica gel columns removes a major bottleneck in production throughput, allowing for larger batch sizes and faster turnover rates without the need for expensive chromatography equipment or vast quantities of silica. This operational simplification leads to substantial cost savings in terms of both raw materials and labor, as the process requires fewer unit operations and less skilled intervention for purification. Additionally, the robustness of the reaction conditions ensures consistent quality across different production runs, reducing the risk of batch failures and ensuring a steady flow of materials for downstream formulation.
- Cost Reduction in Manufacturing: The removal of silica gel chromatography significantly lowers the cost of goods sold by eliminating the expense of stationary phases and the large volumes of organic solvents typically required for elution. Furthermore, the ability to recover and recycle solvents like ethyl acetate and dichloromethane through standard distillation processes further enhances the economic efficiency of the plant. By optimizing the catalyst loading and reaction times, the process minimizes energy consumption associated with heating and cooling, contributing to a leaner and more cost-effective manufacturing profile that supports competitive pricing strategies.
- Enhanced Supply Chain Reliability: The simplified workflow reduces the overall production cycle time, enabling manufacturers to respond more rapidly to fluctuations in market demand and urgent orders. The use of commercially available and stable reagents such as EDCI and sodium methoxide ensures that the supply of raw materials is secure and not subject to the volatility often seen with specialized custom reagents. This reliability is critical for maintaining continuous production schedules and meeting the strict delivery timelines required by global pharmaceutical partners, thereby strengthening the overall resilience of the supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving seamlessly from laboratory bench scales to multi-ton commercial production without the geometric limitations imposed by column chromatography. The reduction in hazardous waste generation, particularly silica waste and mixed solvent streams, simplifies compliance with increasingly stringent environmental regulations and lowers waste disposal costs. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing site against potential regulatory changes regarding solvent emissions and solid waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Garcinoside, providing clarity on the feasibility and advantages of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, offering a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding the adoption of this synthesis route for their specific application needs.
Q: How does this process improve purity compared to traditional methods?
A: By replacing traditional silica gel column chromatography with liquid-liquid extraction and recrystallization, the process achieves purity levels exceeding 99.8% while significantly reducing production time and solvent waste.
Q: What are the key advantages for large-scale supply chain reliability?
A: The elimination of batch-limited column separation allows for continuous processing and easier scale-up from 100 kgs to multi-ton production, ensuring consistent supply continuity for pharmaceutical manufacturers.
Q: Does the mild reaction condition impact the stability of the final product?
A: No, the mild temperature ranges (10-70°C) and nitrogen protection preserve the sensitive polyprenylated xanthone structure of Gambogic Acid, preventing degradation and ensuring high bioactivity in the final Garcinoside.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Garcinoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a partner who can translate complex patent technologies into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of Garcinoside meets the >99.8% purity benchmark required for high-value pharmaceutical applications. Our commitment to quality and consistency makes us the preferred choice for companies seeking a reliable Garcinoside supplier.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this process offers for your specific supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in securing a high-quality supply of this vital anticancer intermediate.
