Scalable Copper-Catalyzed Synthesis of (E)-β-Alkyl Styrenes for Advanced Pharmaceutical Intermediates
Scalable Copper-Catalyzed Synthesis of (E)-β-Alkyl Styrenes for Advanced Pharmaceutical Intermediates
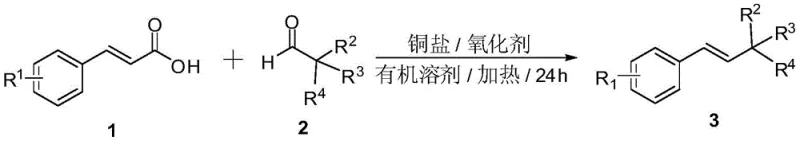
The development of efficient synthetic routes for functionalized styrene derivatives remains a critical priority for the global fine chemical industry, particularly for the production of high-value pharmaceutical intermediates. Patent CN107324976B, published in mid-2020, introduces a transformative methodology for the preparation of (E)-β-alkyl styrene compounds through a copper-catalyzed decarboxylative and decarbonylative cross-coupling reaction. This technology leverages readily available cinnamic acid derivatives and aliphatic aldehydes as primary feedstocks, utilizing a cost-effective copper salt catalyst system in the presence of an organic peroxide oxidant. The significance of this innovation lies in its ability to construct carbon-carbon bonds under relatively mild thermal conditions while maintaining high stereo-selectivity for the (E)-isomer. For R&D teams and process chemists, this represents a substantial advancement over traditional multi-step syntheses, offering a direct pathway to structurally complex scaffolds that are essential precursors in drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of (E)-β-alkyl styrenes was often plagued by significant operational and economic inefficiencies that hindered large-scale adoption. Traditional methodologies frequently relied on the preparation of specialized and often unstable reaction substrates, necessitating complex pre-functionalization steps that increased both material costs and waste generation. Furthermore, earlier literature reports, such as those by Mao Jincheng et al. in 2012 and 2013, described decarboxylative couplings that were strictly limited to benzyl hydrogen compounds, failing to introduce aliphatic hydrocarbon chains at the β-position of the styrene scaffold. These legacy processes typically demanded rigorous inert gas protection to prevent catalyst deactivation and suffered from moderate to low reaction yields, making them unsuitable for the commercial scale-up of complex pharmaceutical intermediates. Additionally, alternative approaches involving dual-metal catalysis systems, such as copper and silver co-catalysis, introduced prohibitive costs due to the use of precious metals and exhibited narrow substrate scope, particularly failing with straight-chain aliphatic carboxylic acids.
The Novel Approach
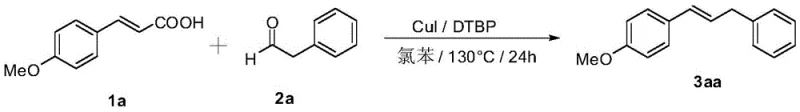
In stark contrast to these historical challenges, the methodology disclosed in CN107324976B utilizes a streamlined single-catalyst system that dramatically simplifies the reaction engineering requirements. By employing inexpensive cinnamic acids and fatty aldehydes in a chlorobenzene solvent system, the process achieves high conversion rates at a manageable temperature of 130°C over a 24-hour period. The optimization data within the patent highlights that cuprous iodide (CuI) serves as the superior catalyst when paired with di-tert-butyl peroxide (DTBP) as the oxidant, delivering yields that significantly exceed those of comparative copper salts like copper chloride or copper carbonate. This novel route eliminates the need for expensive silver co-catalysts and removes the strict requirement for inert atmosphere handling found in previous generations of this chemistry. The result is a robust, operationally simple protocol that facilitates the introduction of diverse aliphatic groups, ranging from branched isopropyl to straight-chain pentyl moieties, thereby expanding the chemical space accessible to medicinal chemists.
Mechanistic Insights into Cu-Catalyzed Decarboxylative Coupling
The core of this technological breakthrough relies on a sophisticated radical-mediated mechanism driven by the synergy between the copper catalyst and the peroxide oxidant. Under the thermal conditions of 130°C, the di-tert-butyl peroxide undergoes homolytic cleavage to generate tert-butoxy radicals, which initiate the catalytic cycle by interacting with the copper species. This interaction facilitates the activation of the cinnamic acid substrate, promoting a sequential decarboxylation and decarbonylation process that generates a reactive vinyl-copper intermediate or a corresponding radical species. Simultaneously, the aliphatic aldehyde undergoes oxidative decarbonylation, likely forming an alkyl radical that couples with the vinyl fragment. This dual activation strategy allows for the precise formation of the C-C bond at the β-position of the styrene framework while preserving the thermodynamically favorable (E)-configuration of the double bond. Understanding this mechanistic pathway is crucial for process chemists aiming to further optimize reaction kinetics or adapt the conditions for continuous flow manufacturing environments.
From an impurity control perspective, the mildness of this reaction system plays a pivotal role in ensuring high product purity, a critical parameter for any reliable pharmaceutical intermediate supplier. The use of chlorobenzene as a high-boiling solvent ensures that the reaction mixture remains homogeneous throughout the 24-hour heating period, preventing localized hot spots that could lead to polymerization or degradation of the sensitive styrene products. Furthermore, the specificity of the CuI/DTBP system minimizes the formation of homocoupling byproducts or over-oxidized species that are common in less selective radical reactions. The post-reaction workup, involving simple extraction and flash column chromatography, effectively removes residual copper salts and organic byproducts, resulting in a final product profile that meets stringent quality specifications. This inherent cleanliness of the reaction reduces the burden on downstream purification units, directly translating to improved overall process efficiency and reduced solvent consumption during isolation.
How to Synthesize (E)-β-Alkyl Styrene Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and pilot-plant production. The standard operating procedure involves charging a pressure-resistant vessel with the cinnamic acid derivative and the chosen aliphatic aldehyde in a molar ratio that favors complete conversion of the acid. Following the addition of the copper catalyst and oxidant, the system is sealed and heated in an oil bath, requiring minimal operator intervention during the reaction phase. The detailed standardized synthesis steps, including specific reagent quantities and safety precautions for handling peroxides at elevated temperatures, are outlined in the guide below.
- Dissolve cinnamic acid derivative and aliphatic aldehyde in chlorobenzene solvent within a pressure-resistant tube.
- Add cuprous iodide (CuI) catalyst and di-tert-butyl peroxide (DTBP) oxidant to the reaction mixture.
- Heat the sealed tube at 130°C for 24 hours, then cool and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards using commodity chemicals like cinnamic acids and simple aldehydes as starting materials fundamentally alters the cost structure of producing these valuable intermediates. By eliminating the dependency on scarce or highly specialized precursors, manufacturers can secure a more stable and predictable supply chain, mitigating the risks associated with raw material volatility. Furthermore, the simplified reaction setup reduces the capital expenditure required for specialized reactor equipment, as the process does not demand high-pressure hydrogenation units or cryogenic cooling systems often associated with alternative synthetic routes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems with abundant copper salts. Unlike previous methods that required dual-metal catalysis involving silver, this approach utilizes cuprous iodide, which is significantly more affordable and widely available in bulk quantities. Additionally, the high atom economy of the decarboxylative coupling means that a larger proportion of the starting mass is converted into the desired product, reducing the cost per kilogram of the final active intermediate. The elimination of inert gas protection requirements further lowers operational expenditures by reducing the consumption of nitrogen or argon and simplifying the reactor sealing mechanisms.
- Enhanced Supply Chain Reliability: The reliance on commercially available feedstocks such as substituted cinnamic acids and fatty aldehydes ensures that production schedules are not held hostage by the lead times of custom-synthesized building blocks. These raw materials are produced globally by numerous chemical suppliers, creating a competitive market that drives down prices and ensures continuity of supply. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive handling requirements, thereby improving the overall reliability of delivery timelines to downstream customers who depend on these intermediates for their own API synthesis campaigns.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with modern green chemistry principles by avoiding the use of toxic heavy metals like palladium or mercury. The solvent system, while requiring proper recovery protocols, is standard in the fine chemical industry, facilitating easy integration into existing waste management infrastructure. The high yields reported in the patent examples suggest that less raw material is wasted as byproduct, which directly correlates to a reduced environmental footprint and lower costs associated with waste disposal and treatment. This makes the technology highly attractive for companies aiming to meet increasingly stringent regulatory standards regarding industrial emissions and chemical safety.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this copper-catalyzed technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is essential for assessing the fit of this chemistry within your specific project pipeline.
Q: What is the optimal catalyst system for this decarboxylative coupling?
A: According to patent CN107324976B, cuprous iodide (CuI) combined with di-tert-butyl peroxide (DTBP) provides the highest yields, outperforming other copper salts like copper chloride or copper carbonate.
Q: Can this method accommodate diverse substrate scopes?
A: Yes, the protocol demonstrates excellent tolerance for various substituents on the cinnamic acid ring, including electron-donating groups like methoxy and electron-withdrawing groups like fluoro, chloro, and trifluoromethyl.
Q: Is inert gas protection required for this reaction?
A: Unlike previous literature methods that strictly require inert atmosphere protection, this novel approach operates effectively in a sealed pressure tube without explicit mention of rigorous inert gas lines, simplifying the operational setup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-β-Alkyl Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced coupling technologies like the one described in CN107324976B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (E)-β-alkyl styrene intermediate meets the exacting standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this copper-catalyzed process for maximum efficiency and cost-effectiveness.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-quality fine chemical intermediates.
