Scalable Catalytic Production of Arylsulfonyl Acrylonitriles for Global Pharmaceutical Supply Chains
Scalable Catalytic Production of Arylsulfonyl Acrylonitriles for Global Pharmaceutical Supply Chains
The rapid advancement of medicinal chemistry has placed a premium on versatile building blocks capable of facilitating complex molecular architectures, particularly those incorporating vinylarene sulfonyl moieties. These structural motifs are increasingly recognized for their profound biological significance, serving as potential neuroprotective agents against Parkinson's disease and exhibiting potent antitrypanosomal activity against African sleeping sickness. Furthermore, their ability to inhibit the sortase SrtA isoform makes them critical candidates for combating Staphylococcus aureus infections. In response to the growing demand for these high-value compounds, the technical disclosure within patent CN113039175A presents a groundbreaking preparation method for arylsulfonyl acrylonitriles. This innovation addresses the longstanding challenges associated with synthesizing these Michael acceptors by introducing a robust, transition metal-catalyzed pathway that starts from readily available arylsulfonyl halides. By shifting away from hazardous reagents and inefficient protocols, this technology offers a sustainable and economically viable route for producing high-purity intermediates essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic landscape for vinylarene sulfonyl compounds has been fraught with significant operational and environmental hurdles that severely limit their utility in large-scale manufacturing. Traditional methodologies often rely heavily on the use of prohibited or highly discouraged solvents such as methylene chloride, which poses severe environmental risks due to its volatility and toxicity, or diethyl ether, which carries the danger of forming explosive peroxides. Additionally, the reliance on N-dimethylformamide (DMF) introduces toxicity concerns, while solvents like ethyl acetate and acetonitrile present challenges regarding flammability and cost volatility. Beyond solvent issues, conventional routes frequently suffer from low reactivity, necessitating prolonged reaction times that drive up energy consumption and reduce overall throughput. The use of expensive starting materials and complicated separation procedures further exacerbates the cost burden, creating a bottleneck for procurement teams seeking reliable sources of these critical chemical building blocks for agrochemical and pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a sophisticated transition metal-containing catalyst compound to drive the conversion of arylsulfonyl halides with remarkable efficiency. This methodology enables the use of lower amounts of reactants and significantly shorter reaction times, directly translating into substantial cost savings through reduced waste generation and lower energy requirements. The core of this innovation lies in the ability to synthesize arylsulfonyl acrylonitriles of formula (I) from inexpensive sulfinates or halides using a reaction with suitable vinyl compounds like acrylonitrile. 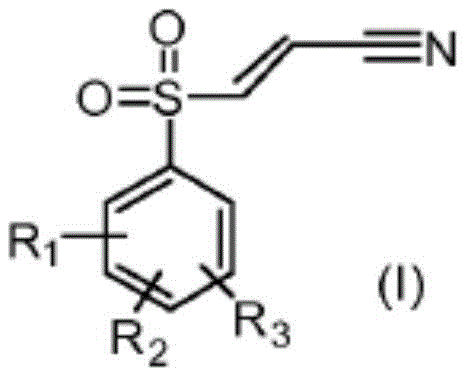 By optimizing the catalytic cycle, the process achieves rapid conversion rates that were previously unattainable, allowing for the production of high-purity products with an improved impurity profile. This shift not only enhances the economic feasibility of manufacturing these compounds but also aligns with modern green chemistry principles by facilitating the use of environmentally benign solvents such as sulfolane, thereby reducing the ecological footprint of the production facility.
By optimizing the catalytic cycle, the process achieves rapid conversion rates that were previously unattainable, allowing for the production of high-purity products with an improved impurity profile. This shift not only enhances the economic feasibility of manufacturing these compounds but also aligns with modern green chemistry principles by facilitating the use of environmentally benign solvents such as sulfolane, thereby reducing the ecological footprint of the production facility.
Mechanistic Insights into Cu-Catalyzed Coupling and Elimination
The mechanistic elegance of this synthesis relies on the formation of a highly active catalyst complex, typically comprising a copper halide and a salt of an organic compound, such as triethylamine hydrochloride. This catalyst complex acts as a powerful driver for the reaction, coordinating with the arylsulfonyl halide and the olefin to facilitate the coupling process under mild thermal conditions. The use of copper iodide, for instance, allows the reaction to proceed at temperatures of 125 °C or less, avoiding the thermal degradation often seen in harsher synthetic routes. The intermediate formed during this catalytic cycle is then subjected to a base-catalyzed elimination step, which is crucial for expelling the halide and establishing the desired double bond geometry. This two-stage mechanism ensures that the final product is obtained in high yield, with the process specifically favoring the formation of the thermodynamically stable (E)-isomer, which is often the biologically active configuration required for downstream pharmaceutical applications.
Furthermore, the control over stereochemistry and impurity profiles is a defining feature of this catalytic system, offering distinct advantages for R&D directors focused on purity specifications. The reaction conditions are tuned to minimize side reactions, resulting in a product mixture with a higher (E)/(Z) isomer ratio compared to previously used processes. In preferred embodiments, such as the synthesis of the compound shown in formula (II), where R1 is a methyl group in the 4-position, the process demonstrates exceptional selectivity. 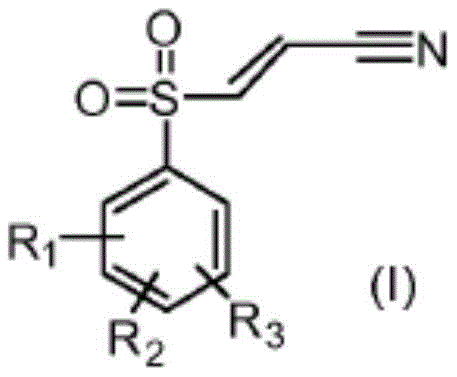 This level of control simplifies the purification process, often allowing for isolation via simple crystallization rather than resource-intensive chromatography. The ability to generate the arylsulfonyl halide in situ further streamlines the workflow, creating a one-pot synthesis that reduces handling steps and potential points of contamination, thereby ensuring a consistent and high-quality output suitable for sensitive biological testing.
This level of control simplifies the purification process, often allowing for isolation via simple crystallization rather than resource-intensive chromatography. The ability to generate the arylsulfonyl halide in situ further streamlines the workflow, creating a one-pot synthesis that reduces handling steps and potential points of contamination, thereby ensuring a consistent and high-quality output suitable for sensitive biological testing.
How to Synthesize 3-Tosylacrylonitrile Efficiently
For technical teams looking to implement this robust synthetic route, the process begins with the careful preparation of the catalyst complex, followed by the controlled addition of reagents to ensure optimal conversion. The protocol is designed to be flexible, accommodating both batch and continuous flow reactor setups, which provides significant versatility for pilot plant and commercial operations. The use of water as a co-solvent in specific amounts can further enhance reaction rates, demonstrating the method's adaptability to different processing conditions. While the general principles are outlined here, precise adherence to the standardized operating procedures is critical for achieving the reported high yields and purity levels. The detailed standardized synthesis steps see the guide below for exact parameters regarding temperature ramps, stirring rates, and workup procedures necessary for reproducible results.
- Prepare the catalyst complex by dissolving copper iodide and triethylamine hydrochloride in acetonitrile at elevated temperatures to form a stable active species.
- Combine the arylsulfonyl halide starting material with acrylonitrile and the prepared catalyst complex in a suitable solvent like sulfolane.
- Heat the reaction mixture to promote coupling, followed by base-catalyzed elimination to isolate the high-purity (E)-isomer product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalytic technology offers profound benefits that extend far beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. By eliminating the dependence on volatile and regulated solvents like methylene chloride, manufacturers can drastically simplify their environmental compliance protocols and reduce the costs associated with hazardous waste disposal. This shift towards greener chemistry not only mitigates regulatory risk but also enhances the long-term sustainability of the supply chain, making it a more attractive partner for multinational corporations with strict ESG mandates. Furthermore, the use of inexpensive and commercially abundant starting materials, such as arylsulfonyl chlorides, ensures that raw material costs remain stable and predictable, shielding the procurement budget from the volatility often seen with exotic reagents.
- Cost Reduction in Manufacturing: The implementation of this efficient catalyst system drives down manufacturing costs by significantly shortening reaction times and improving overall yields, which maximizes the output per batch without requiring additional capital investment in larger reactors. The elimination of expensive transition metal catalysts in favor of more economical copper-based systems, combined with the ability to recycle solvents like sulfolane, leads to substantial operational expenditure savings. Additionally, the simplified purification process, driven by the high selectivity of the reaction, reduces the consumption of silica gel and eluents, further lowering the cost of goods sold for these high-value intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of arylsulfonyl acrylonitriles, as it relies on feedstock that is widely available in the global chemical market. The scalability of the process, proven to work effectively in both batch and continuous flow modes, means that production volumes can be rapidly ramped up to meet surging demand without the lengthy lead times associated with re-engineering legacy processes. This flexibility provides a critical buffer against supply disruptions, ensuring that downstream drug development programs remain on schedule and that commercial launches are not delayed by intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction conditions that are easily managed in standard industrial equipment, avoiding the need for specialized high-pressure or cryogenic infrastructure. The reduced generation of chemical waste and the use of less toxic solvents facilitate easier permitting and operation in regions with stringent environmental regulations. This compliance advantage accelerates the time-to-market for new products and reduces the administrative burden on EHS teams, allowing the organization to focus resources on innovation rather than remediation.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed technical disclosures within the patent literature. These answers address common concerns regarding reaction safety, scalability, and product quality, providing a clear overview of what partners can expect when integrating this synthesis route into their operations. Understanding these technical nuances is essential for assessing the feasibility of adopting this method for the commercial production of complex pharmaceutical intermediates and ensuring alignment with internal quality standards.
Q: What are the primary advantages of the catalytic method described in CN113039175A over conventional synthesis?
A: The patented method eliminates the need for toxic and volatile solvents like methylene chloride and DMF, replacing them with environmentally benign options. It significantly reduces reaction times and improves yields through efficient transition metal catalysis, addressing the low reactivity issues of traditional routes.
Q: How does this process ensure high stereo-selectivity for the (E)-isomer?
A: The process utilizes a specific base-mediated elimination step following the initial catalytic coupling. By carefully selecting bases such as sodium bicarbonate or triethylamine, the reaction favors the formation of the thermodynamically stable (E)-isomer, simplifying downstream purification and enhancing the impurity profile.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. It supports both batch and continuous flow reactor configurations, uses inexpensive and commercially available starting materials like arylsulfonyl halides, and operates at moderate temperatures, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylsulfonyl Acrylonitrile Supplier
As the global demand for specialized pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses deep technical expertise and scalable infrastructure is paramount for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the one described in CN113039175A to deliver superior chemical solutions. Our team boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall project costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets, helping you identify the most economical route to market. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and reliability as your trusted partner in fine chemical synthesis.
