Advanced Asymmetric Synthesis of Axial Chiral 4-Aryl Alpha-Carboline Intermediates for Global Pharma Supply Chains
Advanced Asymmetric Synthesis of Axial Chiral 4-Aryl Alpha-Carboline Intermediates for Global Pharma Supply Chains
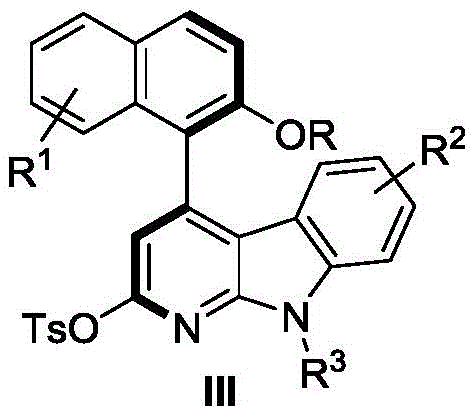
The pharmaceutical industry is constantly seeking robust and scalable methods to access complex chiral scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112898296B introduces a groundbreaking methodology for the asymmetric synthesis of axial chiral 4-aryl alpha-carboline compounds, a structural motif prevalent in numerous bioactive natural products and drug candidates. This technology leverages a sophisticated N-heterocyclic carbene (NHC) organocatalytic system to construct the challenging axially chiral bond with high precision. By utilizing readily available 3-(substituted naphthalen-1-yl) propiolic acid esters and indole-derived sulfonamides, this route offers a direct entry into highly functionalized carboline cores without the need for pre-installed chiral auxiliaries or resolution steps. The ability to control axial chirality through organocatalysis represents a significant leap forward in synthetic efficiency, providing a reliable pathway for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of axially chiral biaryl or heterobiaryl systems has relied heavily on transition metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or oxidative couplings, often requiring stoichiometric chiral ligands or subsequent enzymatic resolution. These conventional pathways frequently suffer from significant drawbacks, including the high cost of precious metal catalysts, the generation of toxic heavy metal waste streams that require rigorous removal to meet regulatory standards, and often modest enantioselectivity that necessitates multiple recrystallization cycles. Furthermore, many existing methods require harsh reaction conditions, such as elevated temperatures or strong bases, which can lead to the decomposition of sensitive functional groups present on the naphthalene or indole rings, thereby limiting the substrate scope and overall yield of the desired pharmacological active ingredients.
The Novel Approach
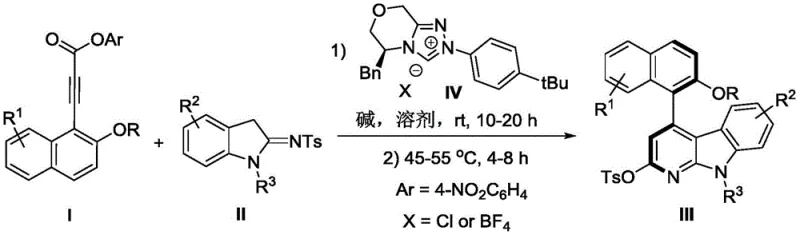
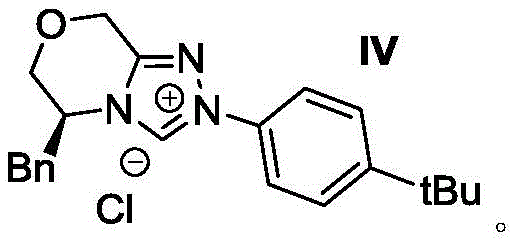
In stark contrast, the methodology disclosed in patent CN112898296B employs a metal-free organocatalytic strategy that fundamentally reshapes the synthetic landscape for these compounds. The core innovation lies in the use of a chiral triazolium salt precursor (Catalyst IV) which generates an active NHC species in situ to activate the alkyne ester towards nucleophilic attack by the indole moiety. This cascade cyclization proceeds under remarkably mild conditions, typically initiating at room temperature and concluding at moderate heating (45-55°C), which preserves the integrity of diverse functional groups. The reaction demonstrates exceptional atom economy and operational simplicity, utilizing acetone as a green solvent and potassium carbonate as a benign base. This novel approach not only streamlines the synthetic sequence but also drastically reduces the environmental footprint and cost of goods, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into NHC-Catalyzed Asymmetric Cyclization
The mechanistic elegance of this transformation is driven by the unique reactivity of the N-heterocyclic carbene catalyst. Upon deprotonation by the base, the chiral triazolium salt generates a nucleophilic carbene that attacks the electron-deficient alkyne of the propiolic acid ester. This forms a zwitterionic allenoate intermediate, which is key to the subsequent stereochemistry-determining step. The bulky chiral environment provided by the catalyst framework effectively shields one face of the reactive intermediate, forcing the intramolecular nucleophilic attack of the indole nitrogen to occur from a specific trajectory. This spatial control is what establishes the axial chirality of the resulting 4-aryl alpha-carboline scaffold. The reaction then proceeds through a proton transfer and elimination of the 4-nitrophenoxide leaving group to restore aromaticity and release the catalyst, completing the cycle. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters for specific substrate analogs.
From an impurity control perspective, this mechanism offers distinct advantages over radical or metal-mediated pathways. The concerted nature of the NHC-catalyzed cycle minimizes the formation of side products typically associated with radical polymerization of alkynes or homocoupling of aryl halides. The high enantiomeric ratios (er) observed, often exceeding 90:10 in optimized examples, indicate that the background racemic reaction is effectively suppressed. This high level of stereocontrol means that the crude reaction mixture contains significantly fewer diastereomeric or enantiomeric impurities, simplifying the downstream purification process. For quality control teams, this translates to a more robust process capable of consistently meeting stringent purity specifications required for clinical grade materials, reducing the risk of batch failures due to chiral impurities.
How to Synthesize Axial Chiral 4-Aryl Alpha-Carboline Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to standard laboratory and pilot plant equipment. The process begins with the precise weighing of the naphthyl propiolate substrate and the indole sulfonamide coupling partner, which are dissolved in dry acetone. The chiral catalyst and inorganic base are added sequentially to initiate the reaction. Maintaining strict temperature control during the two-stage heating profile is critical for maximizing yield and selectivity. Following the reaction period, the workup involves simple concentration and purification via silica gel column chromatography using a petroleum ether and ethyl acetate gradient. For detailed operational parameters and specific stoichiometric ratios tailored to your target molecule, please refer to the standardized synthesis protocol below.
- Combine 3-(substituted naphthalen-1-yl) propiolic acid 4-nitrophenyl ester and substituted indole sulfonamide with chiral NHC catalyst IV and potassium carbonate in acetone solvent.
- Maintain the reaction mixture at room temperature for a duration of 10 to 20 hours to allow for initial activation and intermediate formation.
- Elevate the temperature to 45-55°C for an additional 4 to 8 hours to complete the cyclization, followed by concentration and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed route presents compelling economic and logistical benefits that directly impact the bottom line. The elimination of precious metal catalysts removes a major cost driver and supply chain bottleneck, as the prices of metals like palladium and rhodium are subject to extreme volatility and geopolitical constraints. Furthermore, the use of commodity chemicals such as acetone and potassium carbonate ensures that raw material sourcing is stable and inexpensive, mitigating the risk of production delays due to reagent shortages. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to substantial cost savings in utility expenses over the lifecycle of the product. These factors combine to create a highly competitive cost structure for the manufacturing of these complex intermediates.
- Cost Reduction in Manufacturing: The transition from metal-catalyzed to organocatalytic synthesis eliminates the need for expensive metal scavengers and extensive purification steps required to reduce metal residues to ppm levels. This qualitative shift in process chemistry significantly lowers the cost of goods sold (COGS) by reducing both material inputs and processing time. Additionally, the high yields reported in the patent examples suggest that less starting material is wasted, further enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: By relying on robust organocatalysts and common solvents, the supply chain becomes more resilient to disruptions often seen with specialized reagents. The simplicity of the reaction setup allows for flexible manufacturing across different facilities without the need for specialized corrosion-resistant reactors often required for harsh acidic or basic conditions. This flexibility ensures consistent supply continuity, allowing partners to scale production up or down rapidly in response to market demand fluctuations without compromising quality.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. The absence of heavy metals simplifies regulatory compliance and environmental reporting, accelerating the timeline for regulatory approvals. The scalability is further supported by the exothermic nature of the reaction being manageable under the described mild conditions, allowing for safe scale-up from gram to multi-kilogram batches with predictable outcomes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these axially chiral compounds. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and stakeholders evaluating this synthetic route for their pipeline.
Q: What are the primary advantages of this NHC-catalyzed route over traditional metal-catalyzed methods?
A: This organocatalytic approach eliminates the need for expensive and toxic transition metals like palladium or rhodium, significantly reducing heavy metal residue risks in the final API and lowering raw material costs associated with metal scavenging processes.
Q: How does this synthesis method impact the enantiomeric purity of the final product?
A: The use of the specific chiral triazolium salt catalyst (Formula IV) ensures high enantioselectivity, typically achieving er ratios exceeding 90:10, which minimizes the need for costly downstream chiral resolution steps.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the reaction operates under mild thermal conditions (room temperature to 50°C) using common solvents like acetone, which simplifies reactor requirements and enhances safety profiles for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral 4-Aryl Alpha-Carboline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of innovative medicines. Our team of expert chemists has thoroughly analyzed the synthetic route described in patent CN112898296B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities and rigorous QC labs capable of handling sensitive organocatalytic reactions, ensuring that every batch meets stringent purity specifications and enantiomeric excess requirements. Our commitment to technical excellence allows us to adapt this methodology to produce a wide range of analogs tailored to your specific drug discovery needs.
We invite you to collaborate with us to optimize your supply chain for these valuable intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this organocatalytic route can improve your project economics. Please contact us today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us be your partner in turning complex chemical challenges into commercial successes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
