Efficient Ruthenium-Catalyzed Synthesis of N,N'-Disubstituted Ureas for Commercial Pharmaceutical Applications
Efficient Ruthenium-Catalyzed Synthesis of N,N'-Disubstituted Ureas for Commercial Pharmaceutical Applications
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding the efficient construction of urea linkages which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is documented in patent CN109535037B, which discloses a novel and highly effective method for preparing N,N'-disubstituted urea compounds. This technology leverages a sophisticated ruthenium-catalyzed system to transform N-hydrocarbon acyloxy amides into valuable symmetric ureas with exceptional selectivity. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this methodology represents a paradigm shift away from cumbersome multi-step processes towards a streamlined, atom-economical solution that promises substantial improvements in both yield and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of urea derivatives has been plagued by significant inefficiencies that hinder large-scale production. Traditional routes often necessitate the use of multiple distinct raw materials and aggressive chemical reagents, leading to complex reaction mixtures that are difficult to purify. These legacy methods frequently suffer from low atom economy and generate substantial waste streams, posing challenges for environmental compliance and cost management. Furthermore, achieving high selectivity for symmetric N,N'-disubstituted ureas specifically, rather than asymmetric variants or mixed products, has proven technically demanding. The requirement for harsh reaction conditions in many classical protocols also limits the substrate scope, preventing the incorporation of sensitive functional groups that are often critical in modern drug design. Consequently, manufacturers face inflated production costs and extended lead times when relying on these outdated synthetic strategies.
The Novel Approach
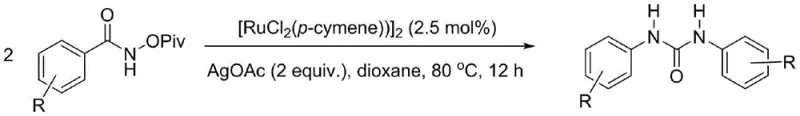
In stark contrast to these historical limitations, the invention described in CN109535037B introduces a remarkably elegant solution that utilizes N-hydrocarbon acyloxy amides as the sole organic starting material. This innovative route operates under mild reaction conditions, typically requiring temperatures around 80 °C, which preserves the integrity of sensitive functional groups on the aromatic rings. The process is catalyzed by a dichloro(p-methylcumene)ruthenium(II) dimer complex in the presence of silver acetate, a combination that drives the reaction with high efficiency and selectivity. By simplifying the reactant profile to a single amide derivative, the method drastically reduces the complexity of the supply chain and minimizes the potential for side reactions. This streamlined approach not only enhances the overall yield but also simplifies the downstream purification process, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ruthenium-Catalyzed Urea Formation
The core of this technological advancement lies in the unique reactivity of the ruthenium catalyst system towards the N-O bond of the acyloxy amide substrate. The mechanism likely involves the activation of the N-hydrocarbon acyloxy amide by the [RuCl2(p-cymene)]2 catalyst, facilitated by the silver acetate additive which may act as a halide scavenger or Lewis acid to promote bond cleavage. This activation generates a reactive nitrenoid or similar intermediate that subsequently undergoes coupling to form the central urea carbonyl linkage. The beauty of this mechanism is its ability to proceed under aerobic conditions, eliminating the need for stringent inert atmospheres that often complicate industrial reactors. The catalytic cycle is robust enough to tolerate a wide variety of substituents on the phenyl ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens, demonstrating the versatility required for diverse medicinal chemistry applications.
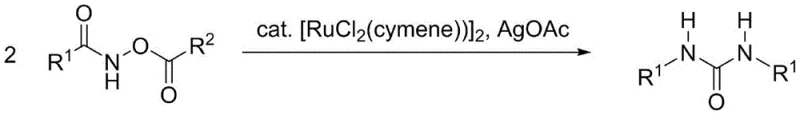
From an impurity control perspective, this catalytic system offers distinct advantages due to its high chemoselectivity. The reaction pathway is directed specifically towards the formation of the symmetric urea structure, minimizing the formation of asymmetric byproducts or unreacted starting materials that typically plague non-catalytic thermal decompositions. The use of a defined transition metal complex ensures that the reaction kinetics are controlled, preventing runaway exotherms or the generation of tarry decomposition products. This level of control is critical for maintaining a clean impurity profile, which is a primary concern for high-purity pharmaceutical intermediates destined for clinical use. The ability to achieve such high selectivity with a relatively low catalyst loading further underscores the efficiency of the process, reducing the burden on metal removal steps during final purification.
How to Synthesize N,N'-Disubstituted Urea Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The general protocol involves dissolving the N-hydrocarbon acyloxy amide substrate in a suitable organic solvent, such as dioxane, and adding the ruthenium catalyst and silver acetate additive in precise molar ratios. The mixture is then heated to the optimal temperature range, allowing the catalytic cycle to proceed to completion. While the patent details various specific examples with different substrates, the underlying principle remains consistent: the careful balance of catalyst, additive, and thermal energy drives the transformation. For detailed operational specifics, please refer to the standardized guide below which outlines the critical steps for successful execution.
- Combine N-hydrocarbon acyloxy amide substrate with [RuCl2(p-cymene)]2 catalyst and AgOAc additive in an organic solvent such as dioxane.
- Heat the reaction mixture to a temperature between 60 °C and 100 °C, preferably 80 °C, and maintain for 1 to 24 hours under air atmosphere.
- Upon completion, isolate the N,N'-disubstituted urea product via column chromatography or recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed methodology translates into tangible strategic benefits that extend beyond mere chemical curiosity. The simplification of the raw material list to a single amide derivative significantly de-risks the supply chain by reducing the number of vendors and quality control checkpoints required. This consolidation of inputs leads to a more resilient manufacturing process that is less susceptible to disruptions in the availability of specialized reagents. Furthermore, the mild reaction conditions imply lower energy consumption compared to high-temperature or high-pressure alternatives, contributing to a reduced carbon footprint and lower utility costs. These factors combined create a compelling economic case for integrating this technology into existing production lines for urea-based active ingredients.
- Cost Reduction in Manufacturing: The elimination of multiple reagents and the use of a highly selective catalyst system inherently lowers the cost of goods sold. By avoiding the need for expensive coupling agents or protecting group strategies often seen in traditional urea synthesis, the process achieves significant cost optimization. The high selectivity also means less material is lost to byproduct formation, improving the overall mass balance and reducing waste disposal costs. Additionally, the catalyst loading can be optimized to very low levels while maintaining efficiency, further driving down the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The substrates used in this method, such as substituted benzamides, are generally commercially available or easily synthesized from commodity chemicals. This accessibility ensures a stable supply of starting materials, mitigating the risk of production delays caused by raw material shortages. The robustness of the reaction under air atmosphere also removes the dependency on specialized inert gas infrastructure, allowing for more flexible manufacturing scheduling. This reliability is crucial for maintaining continuous supply to downstream customers in the pharmaceutical sector who demand strict adherence to delivery timelines.
- Scalability and Environmental Compliance: The operational simplicity of this method makes it highly amenable to scale-up from gram to ton quantities without significant re-engineering of the process. The use of common solvents like dioxane and the absence of hazardous reagents simplify waste treatment and regulatory compliance. The mild thermal profile reduces the safety risks associated with exothermic reactions, facilitating safer operations in large-scale reactors. These attributes collectively support the sustainable growth of production capacity to meet increasing market demand for high-quality urea intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis for their specific projects, we have compiled answers to common inquiries based on the patent data. These responses address key concerns regarding reaction conditions, catalyst systems, and product scope. Understanding these technical nuances is essential for making informed decisions about process adoption and resource allocation. The following insights are derived directly from the experimental data provided in the intellectual property documentation.
Q: What are the primary advantages of this ruthenium-catalyzed urea synthesis over conventional methods?
A: Unlike traditional methods that often require multiple reagents and harsh conditions, this novel approach utilizes a single amide derivative raw material under mild conditions (e.g., 80 °C). It offers high product selectivity for symmetric ureas and simplifies the operational workflow significantly.
Q: What is the specific catalyst system used in this patented process?
A: The process employs dichloro(p-methylcumene)ruthenium(II) dimer, denoted as [RuCl2(p-cymene)]2, as the primary catalyst. This is used in conjunction with silver acetate (AgOAc) as an additive to facilitate the reaction efficiently.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up. The reaction operates at moderate temperatures (60-100 °C) and uses readily available solvents like dioxane. The simple workup procedures, such as column chromatography or recrystallization, support robust commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Disubstituted Urea Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the one described in CN109535037B for enhancing the efficiency of pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs to guarantee that every batch of N,N'-disubstituted urea meets the highest industry standards. We are equipped to handle the complexities of transition metal catalysis, including efficient metal scavenging and purification protocols.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce costs while maintaining the highest levels of product integrity and regulatory compliance.
