Scalable Green Synthesis of 2-Substituted 3-Methylindole Derivatives Using Metal-Free Photocatalysis
The landscape of fine chemical synthesis is undergoing a paradigm shift towards sustainability, driven by the urgent need to eliminate toxic heavy metals and reduce energy consumption in industrial processes. A groundbreaking development in this arena is detailed in patent CN114031541B, which discloses a novel preparation method for 2-substituted 3-methylindole derivatives utilizing graphitic carbon nitride (g-C3N4) as a heterogeneous photocatalyst. This technology addresses critical pain points in the production of high-value pharmaceutical intermediates, specifically targeting the C2-alkylation of indoles, a transformation traditionally plagued by the use of expensive noble metals and harsh reaction conditions. By leveraging visible light irradiation and an environmentally benign solvent system, this method offers a robust pathway for generating complex indole scaffolds that are essential building blocks for drugs ranging from antihypertensives to anticancer agents. The strategic adoption of this metal-free protocol allows manufacturers to align their production capabilities with increasingly stringent global regulatory standards regarding residual solvents and heavy metal impurities.
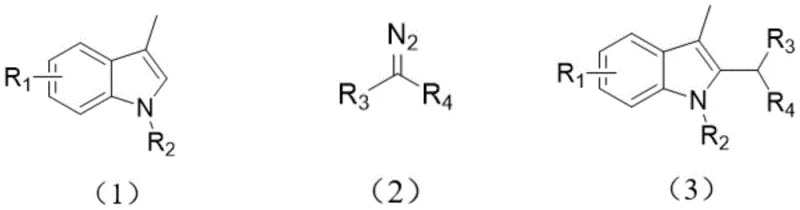
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the C2-position alkylation of indole derivatives has been predominantly achieved through transition metal-catalyzed carbene insertion reactions, often relying on rhodium or manganese complexes. These conventional methodologies, while effective in specific contexts, suffer from significant drawbacks that hinder their scalability and economic viability for large-scale manufacturing. The reliance on noble metal catalysts such as rhodium introduces substantial raw material costs and creates a persistent risk of metal contamination in the final active pharmaceutical ingredient (API), necessitating expensive and complex purification steps to meet safety specifications. Furthermore, many of these traditional protocols require elevated temperatures, strictly anhydrous conditions, and the use of toxic organic solvents, which collectively increase the environmental footprint and operational hazards of the synthesis. The narrow substrate scope associated with some directing-group-dependent strategies further limits the versatility of these methods, making them unsuitable for the diverse library synthesis required in modern drug discovery pipelines.
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes a pure organic semiconductor, g-C3N4, to drive the alkylation reaction under mild visible light irradiation at room temperature. This approach fundamentally alters the reaction economics by replacing costly transition metals with a catalyst derived from the thermal polymerization of inexpensive urea, which is not only abundant but also non-toxic. The reaction proceeds efficiently in a mixture of ethanol and water, a green solvent system that drastically reduces the volatility and flammability risks associated with traditional organic solvents like dichloromethane or toluene. Crucially, the heterogeneous nature of the g-C3N4 catalyst allows for easy separation and recycling, ensuring that the process remains sustainable over multiple batches without compromising yield or selectivity. This methodology effectively broadens the scope of accessible indole derivatives, accommodating a wide range of electronic substituents while maintaining high atom economy and operational simplicity.
Mechanistic Insights into g-C3N4 Photocatalytic Alkylation
The efficacy of this synthesis relies on the unique electronic properties of graphitic carbon nitride, which functions as a polymer semiconductor with a suitable band gap for visible light absorption. Upon irradiation with blue light (400-450 nm), electrons in the valence band (VB) of the g-C3N4 are excited to the conduction band (CB), generating electron-hole pairs that drive the redox processes necessary for bond formation. The photo-generated electrons interact with the diazo ester substrate, facilitating the extrusion of nitrogen gas and the formation of a reactive radical or carbene-like intermediate. This activated species then undergoes a selective electrophilic attack at the C2 position of the 3-methylindole ring, a regioselectivity that is thermodynamically favored under these specific photocatalytic conditions. The presence of an amine additive, such as triethylamine, plays a crucial role in modulating the reaction environment, potentially acting as a sacrificial electron donor or a base to neutralize acidic byproducts, thereby sustaining the catalytic cycle.
From an impurity control perspective, the metal-free nature of the catalyst ensures that the final product profile is free from transition metal residues, a critical quality attribute for pharmaceutical intermediates intended for human use. The mild reaction temperature (18-30°C) minimizes the formation of thermal degradation byproducts and polymerization side reactions that often plague high-temperature syntheses. Additionally, the use of an aqueous ethanol solvent system enhances the solubility of polar intermediates while suppressing non-polar side reactions, leading to cleaner reaction profiles and simplified downstream processing. The robustness of the g-C3N4 structure under irradiation ensures consistent performance over time, reducing batch-to-batch variability and enhancing the reliability of the manufacturing process for high-purity indole derivatives.
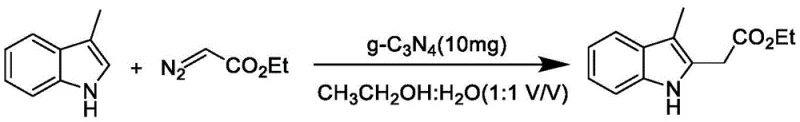
How to Synthesize 2-Substituted 3-Methylindole Efficiently
To implement this advanced photocatalytic protocol in a laboratory or pilot setting, precise control over reaction parameters is essential to maximize yield and reproducibility. The process begins with the careful selection of high-quality 3-methylindole substrates and diazo ester reagents, which are combined in a quartz reaction vessel to ensure optimal transmission of the activating blue light. The addition of the g-C3N4 photocatalyst and a tertiary amine additive must be performed under an inert argon atmosphere to prevent oxidative quenching of the reactive intermediates by atmospheric oxygen. Following the irradiation period, the workup procedure involves simple filtration to recover the solid catalyst, followed by standard extraction and chromatographic purification to isolate the target molecule with high purity. Detailed standardized synthetic steps for this transformation are provided in the guide below to assist technical teams in replicating these results.
- Combine 3-methylindole compound, diazo ester compound, additive (e.g., triethylamine), and g-C3N4 photocatalyst in a quartz vessel with ethanol/water solvent.
- Purge the reaction system with argon gas to create an inert atmosphere and stir the mixture at room temperature (18-30°C).
- Irradiate the reaction mixture with 400-450 nm blue LED light for 12-24 hours, then purify the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this g-C3N4 mediated synthesis represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with critical raw materials. By eliminating the dependency on volatile precious metal markets for catalysts like rhodium or ruthenium, manufacturers can stabilize their production costs and avoid the supply chain disruptions that frequently affect rare earth and noble metal availability. The ability to utilize water-containing solvent systems further reduces the logistical burden and safety costs associated with the storage and disposal of hazardous volatile organic compounds (VOCs). Moreover, the recyclability of the photocatalyst translates directly into lower consumable costs per kilogram of product, enhancing the overall margin profile of the manufacturing operation without sacrificing quality or throughput.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with urea-derived g-C3N4 results in a drastic reduction in raw material expenditure, as the photocatalyst is synthesized from commodity chemicals. This cost benefit is compounded by the elimination of downstream heavy metal scavenging steps, which typically require specialized resins and additional processing time, thereby streamlining the overall production workflow. Furthermore, the energy efficiency of using LED light sources at room temperature significantly lowers utility costs compared to thermal heating methods that require sustained high-temperature inputs. These cumulative savings allow for a more competitive pricing strategy for high-purity pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics ensures a stable supply of diverse indole derivatives, reducing the risk of batch failures due to raw material inconsistencies. Since the catalyst is heterogeneous and easily separable, there is no risk of catalyst leaching affecting subsequent batches, which guarantees consistent product quality and reduces the need for extensive re-testing. The use of common solvents like ethanol and water simplifies procurement logistics, as these materials are globally available and less subject to the regulatory restrictions imposed on chlorinated or aromatic solvents. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this photocatalytic process is inherently safer and more environmentally compliant than traditional methods, as it avoids the generation of heavy metal waste streams that require specialized treatment facilities. The use of visible light allows for the design of continuous flow reactors with high surface-area-to-volume ratios, facilitating the transition from gram-scale laboratory synthesis to multi-ton commercial production with minimal re-optimization. Compliance with green chemistry principles enhances the corporate sustainability profile, making the supply chain more resilient to future environmental regulations and carbon taxation policies. This forward-looking approach ensures long-term operational viability and aligns with the ESG (Environmental, Social, and Governance) goals of modern multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology, based on the detailed experimental data provided in the patent literature. Understanding these nuances is vital for R&D teams evaluating the feasibility of integrating this method into existing manufacturing lines. The answers reflect the specific advantages of the g-C3N4 system in terms of yield, selectivity, and operational safety.
Q: What are the advantages of using g-C3N4 over transition metal catalysts for indole alkylation?
A: g-C3N4 is a metal-free, organic semiconductor photocatalyst that eliminates the risk of heavy metal residues in the final product, which is critical for pharmaceutical applications. It is also derived from cheap urea, is recyclable, and operates under mild visible light conditions compared to the harsh conditions often required for Rh or Mn catalysts.
Q: Can this photocatalytic method tolerate various functional groups on the indole ring?
A: Yes, the method demonstrates broad substrate applicability. It successfully accommodates both electron-withdrawing groups (such as -F, -Cl, -Br) and electron-donating groups (such as -CH3, -OCH3, -OBn) at the C5 position of the indole ring, maintaining high yields across diverse substrates.
Q: Is the g-C3N4 catalyst reusable in this synthesis process?
A: Yes, one of the key green chemistry features of this protocol is the heterogeneity of the g-C3N4 catalyst. It can be recovered from the reaction mixture and recycled for multiple runs without significant loss of catalytic activity, contributing to substantial cost savings and waste reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted 3-Methylindole Supplier
As the demand for high-quality indole intermediates continues to grow across the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and scalable production capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging advanced photocatalytic techniques to deliver products with stringent purity specifications. We operate rigorous QC labs equipped to detect trace impurities and ensure that every batch meets the highest international standards for safety and efficacy. By collaborating with us, clients can accelerate their drug development timelines while benefiting from our commitment to green chemistry and cost-effective manufacturing solutions.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis today to understand the economic impact of switching to this metal-free protocol for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this approach for your target molecules.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
