Advanced Chiral Ferrocene-Biphenol PNO Ligands for Scalable Asymmetric Hydrogenation
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel tridentate ligand architectures that address long-standing limitations in stability and selectivity. Patent CN113004341A discloses a groundbreaking class of PNO ligands containing both facial chiral ferrocene and axial chiral biphenol moieties, offering a robust solution for the enantioselective synthesis of high-value pharmaceutical intermediates. Unlike traditional bidentate systems, these tridentate ligands create a more defined chiral environment around the transition metal center, significantly enhancing catalytic performance in asymmetric hydrogenation reactions. The structural versatility allows for fine-tuning of electronic and steric properties, making them applicable to a wide range of substrates including functionalized ketones and imines. 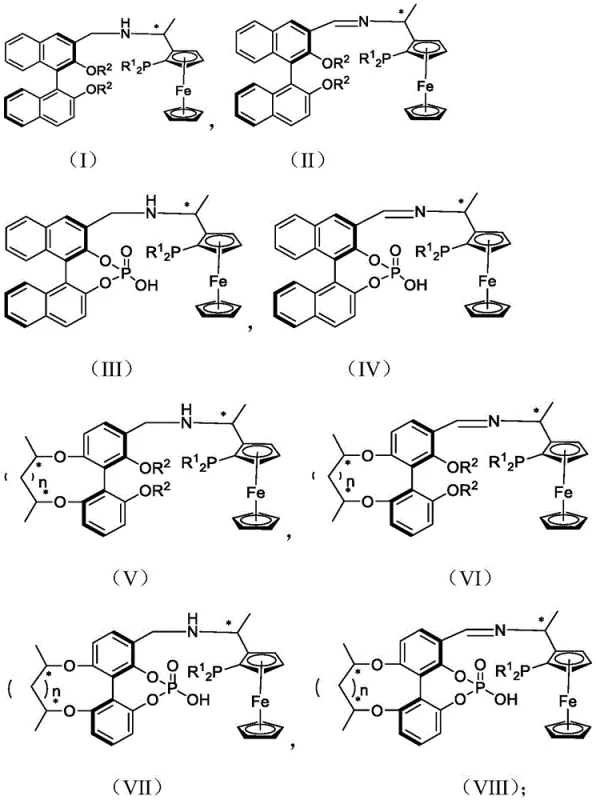
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient chiral ligands has been hindered by complex synthetic routes and limited substrate scope. Earlier generations of tridentate ligands, such as SpiroPAP, while effective, often require multi-step syntheses that are difficult to scale and involve expensive chiral precursors. Furthermore, many existing PNN or PNO ligands suffer from stability issues under rigorous reaction conditions, leading to catalyst decomposition and reduced turnover numbers. The reliance on alkyl alcohol hydroxyl groups for coordination in some prior art ligands results in weaker acidity and less effective bifunctional catalysis, limiting their ability to activate polar double bonds efficiently. These factors collectively increase the cost of goods and complicate the supply chain for critical chiral building blocks.
The Novel Approach
The innovative strategy presented in this patent overcomes these hurdles by integrating axially chiral binaphthol or biphenol units with planar chiral ferrocene backbones. This dual-chirality design not only simplifies the synthetic pathway by utilizing commercially available bulk chiral raw materials but also enhances the rigidity and stability of the ligand framework. The introduction of phenolic hydroxyl groups provides stronger acidity compared to alkyl alcohols, facilitating a more potent bifunctional catalytic mechanism where the hydroxyl proton interacts directly with the substrate. This approach ensures excellent selectivity and high catalytic activity, as evidenced by the successful application in synthesizing bioactive molecules like Woody acetate and various enzyme inhibitors. 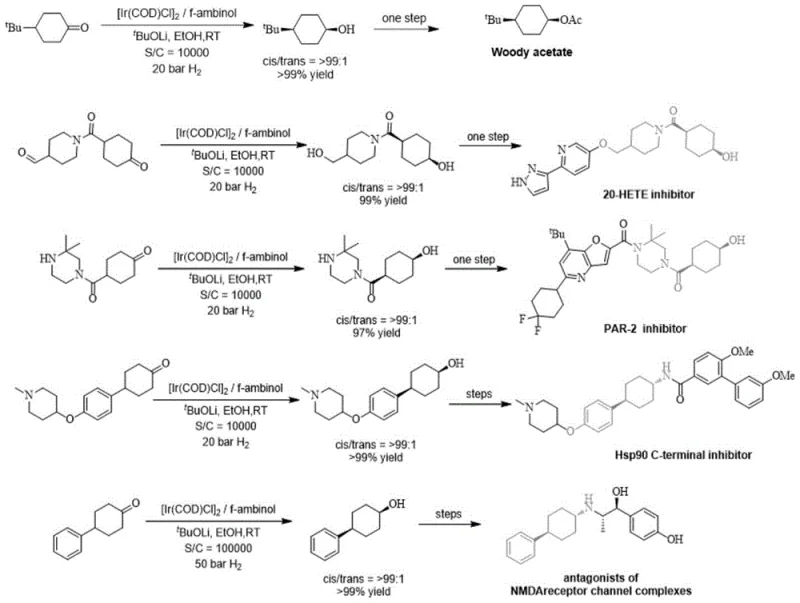
Mechanistic Insights into Tridentate PNO Coordination and Bifunctional Catalysis
The exceptional performance of these ligands stems from their unique coordination mode and electronic properties. In the catalytic cycle, the tridentate PNO ligand coordinates to the transition metal center (such as Iridium, Ruthenium, or Rhodium) through the phosphorus, nitrogen, and oxygen atoms, forming a highly stable octahedral or square planar complex. This tridentate binding prevents ligand dissociation, a common failure mode in bidentate systems, thereby extending catalyst lifetime. The oxygen atom from the axial chiral biphenol participates in coordination, which increases the electron density on the metal center, making it more prone to generating hydride species essential for hydrogenation. Simultaneously, the acidic proton of the phenolic hydroxyl group engages in hydrogen bonding with the substrate's polar functionality, lowering the activation energy for hydride transfer.
Impurity control is inherently improved due to the high diastereoselectivity and enantioselectivity driven by the rigid chiral pocket. The steric bulk provided by the ferrocene and biphenol units effectively blocks unfavorable approach trajectories for the substrate, ensuring that only the desired enantiomer is formed. This precision minimizes the formation of unwanted isomers, simplifying downstream purification processes and reducing waste. The patent data indicates that these catalysts can achieve cis/trans ratios greater than 20:1 and enantiomeric excess values exceeding 99% in the hydrogenation of substituted cyclohexanones, demonstrating superior control over stereochemical outcomes compared to conventional catalysts.
How to Synthesize (S,R)-f-ambinol Efficiently
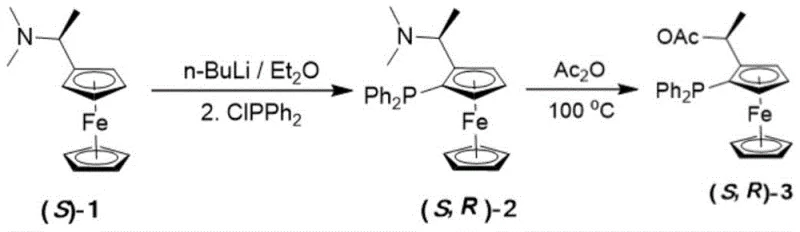
The synthesis of the representative ligand (S,R)-f-ambinol (L1) exemplifies the practicality and scalability of this technology. The process begins with the preparation of a planar chiral ferrocene amine precursor, followed by the modification of an axially chiral binaphthol derivative. Key steps include lithiation, phosphorylation, and a condensation-reduction sequence that links the two chiral fragments. The use of standard reagents like n-butyllithium, chlorodiphenylphosphine, and sodium borohydride ensures that the process is compatible with existing fine chemical manufacturing infrastructure. Detailed standardized synthetic steps for producing high-purity ligands are outlined in the guide below.
- Preparation of chiral ferrocene amine precursor via lithiation of planar chiral ferrocene and reaction with chlorodiphenylphosphine.
- Protection and formylation of axially chiral binaphthol or biphenol backbones to generate key aldehyde intermediates.
- Condensation of the ferrocene amine with the chiral aldehyde followed by reduction and deprotection to yield the final PNO ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this ligand technology translates into tangible strategic benefits centered around cost efficiency and supply reliability. The synthetic route relies on chiral raw materials that are commercial bulk products, eliminating the need for custom synthesis of exotic starting materials which often carry long lead times and high price volatility. By simplifying the ligand synthesis to fewer steps with high yields, the overall production cost of the catalyst is significantly reduced. This cost reduction in pharmaceutical intermediate manufacturing allows for more competitive pricing of the final active pharmaceutical ingredients without compromising on quality or purity standards.
- Cost Reduction in Manufacturing: The streamlined synthesis avoids expensive and hazardous reagents, utilizing common laboratory chemicals that are easily sourced globally. The high catalytic efficiency, with Turnover Numbers (TON) reaching up to 100,000 as demonstrated in the patent examples, means that significantly less catalyst is required per kilogram of product. This drastic reduction in catalyst loading directly lowers the raw material cost per batch and minimizes the burden on waste treatment systems associated with heavy metal disposal.
- Enhanced Supply Chain Reliability: Since the core chiral building blocks like BINOL and ferrocene derivatives are produced on a large industrial scale, the risk of supply disruption is minimal. The robustness of the ligand structure ensures that the catalyst maintains its activity over extended periods, reducing the frequency of catalyst replenishment. This stability supports continuous manufacturing processes and ensures consistent product quality, which is critical for maintaining regulatory compliance and meeting delivery schedules for global clients.
- Scalability and Environmental Compliance: The reaction conditions described, such as room temperature hydrogenation and the use of ethanol as a solvent, align well with green chemistry principles. The high selectivity reduces the generation of isomeric byproducts, simplifying purification and reducing solvent consumption. This environmental compatibility facilitates easier regulatory approval and supports corporate sustainability goals, making the technology attractive for long-term investment in green manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these chiral ferrocene-biphenol PNO ligands in industrial processes. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this technology for adoption.
Q: What distinguishes these PNO ligands from traditional bidentate ligands?
A: These tridentate PNO ligands provide a deeper and more rigid chiral pocket due to the combination of facial chiral ferrocene and axial chiral biphenol, resulting in superior stereoselectivity and catalyst stability compared to bidentate systems.
Q: Can these ligands be used for large-scale industrial hydrogenation?
A: Yes, the patent demonstrates Turnover Numbers (TON) reaching 100,000 with excellent yields and selectivity, indicating high catalyst efficiency suitable for commercial scale-up.
Q: Are the raw materials for these ligands commercially available?
A: The synthesis utilizes commercial bulk chiral starting materials such as BINOL and planar chiral ferrocene derivatives, ensuring supply chain reliability and cost-effectiveness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable f-ambinol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the enantiomeric excess and chemical purity of complex chiral ligands and their metal complexes to meet the highest international pharmacopoeia standards.
We invite you to collaborate with our technical team to explore how this cutting-edge ligand technology can optimize your specific synthesis routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our reliable f-ambinol supplier capabilities can drive efficiency and innovation in your supply chain.
