Advanced Asymmetric Hydrogenation for High-Purity Chiral Dihydro-9-phenanthrene Amines
The pharmaceutical industry continuously seeks efficient pathways to access chiral building blocks, particularly for complex alkaloids and bioactive molecules. Patent CN111825508A introduces a groundbreaking preparation method for dihydro-9-phenanthrene amine compounds and their corresponding chiral products. This technology addresses a long-standing challenge in organic synthesis: the asymmetric catalytic hydrogenation of aromatic amines, which are notoriously difficult to reduce due to their strong aromaticity and tendency to poison transition metal catalysts. By employing specifically designed chiral metal complexes, this invention enables the selective hydrogenation of 9-phenanthrene amine derivatives with exceptional enantioselectivity. For procurement and R&D teams seeking a reliable pharmaceutical intermediates supplier, this patent represents a significant leap forward in accessing high-value chiral amines that serve as precursors for drugs like Nuciferine and Apomorphine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral dihydro-9-phenanthrene amines has been fraught with significant technical hurdles. Simple aromatic amine compounds possess extremely stable aromatic structures, making them resistant to homogeneous asymmetric catalytic hydrogenation. Furthermore, both the aromatic amine reactants and the resulting dihydrogenated products contain amino groups that readily coordinate with central metals like ruthenium, rhodium, or iridium. This coordination often leads to catalyst poisoning and deactivation, halting the reaction progress and resulting in poor yields. Previous literature reports on the catalytic hydrogenation of aromatic amines are scarce, with only limited success achieved through substrate activation strategies that often require harsh conditions or expensive, non-recyclable reagents. These limitations have historically increased the cost and complexity of manufacturing these critical pharmaceutical intermediates.
The Novel Approach
The present invention overcomes these barriers by utilizing a novel class of chiral catalysts represented by Formula (2) in conjunction with specific 9-phenanthrene amine substrates shown in Formula (1). 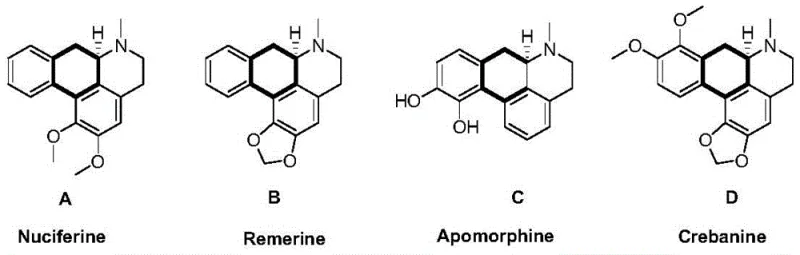 . The inventors discovered that under the specific reaction conditions provided by this system, the aromatic amine substrate can isomerize into an exocyclic imine structure. This transient imine species is then susceptible to asymmetric catalytic hydrogenation by the chiral metal complex. This mechanism effectively bypasses the catalyst poisoning issue associated with direct aromatic amine reduction. The result is a robust process capable of producing a series of dihydro-9-phenanthrene amine compounds with high conversion rates and superior optical purity, offering a streamlined alternative to traditional resolution methods.
. The inventors discovered that under the specific reaction conditions provided by this system, the aromatic amine substrate can isomerize into an exocyclic imine structure. This transient imine species is then susceptible to asymmetric catalytic hydrogenation by the chiral metal complex. This mechanism effectively bypasses the catalyst poisoning issue associated with direct aromatic amine reduction. The result is a robust process capable of producing a series of dihydro-9-phenanthrene amine compounds with high conversion rates and superior optical purity, offering a streamlined alternative to traditional resolution methods.
Mechanistic Insights into Asymmetric Catalytic Hydrogenation
The core of this technology lies in the unique structure of the chiral catalyst, depicted generally in Formula (2). 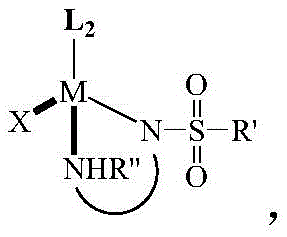 . This complex features a central metal atom M, which can be ruthenium, rhodium, or iridium, coordinated with a substituted eta-6-benzene or eta-5-cyclopentadienyl ligand (L2). Crucially, the catalyst incorporates a chiral diamine ligand formed from a chiral diamine backbone linked to a sulfonyl group. One nitrogen atom of this ligand forms a covalent bond with the metal, while the other forms a coordination bond. This dual-binding mode significantly enhances the chemical stability of the catalyst, preventing the ligand from being displaced by the substrate or product. This stability is paramount for maintaining catalytic activity throughout the reaction cycle, ensuring consistent performance even at lower catalyst loadings.
. This complex features a central metal atom M, which can be ruthenium, rhodium, or iridium, coordinated with a substituted eta-6-benzene or eta-5-cyclopentadienyl ligand (L2). Crucially, the catalyst incorporates a chiral diamine ligand formed from a chiral diamine backbone linked to a sulfonyl group. One nitrogen atom of this ligand forms a covalent bond with the metal, while the other forms a coordination bond. This dual-binding mode significantly enhances the chemical stability of the catalyst, preventing the ligand from being displaced by the substrate or product. This stability is paramount for maintaining catalytic activity throughout the reaction cycle, ensuring consistent performance even at lower catalyst loadings.
The stereochemical outcome of the reaction is dictated by the chirality of the diamine ligand. For instance, using an (R,R)-configured chiral diamine typically favors the formation of the (R)-enantiomer of the dihydro-9-phenanthrene amine product, while an (S,S)-configured ligand yields the (S)-enantiomer. The patent data indicates that specific anions (X), such as triflate [OTf]- or tetraarylborate [BArF]-, further optimize the catalytic performance. The final chiral product, shown in Formula (4), contains a newly formed chiral carbon center. 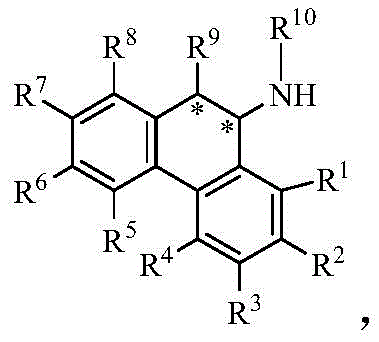 . This precise control over stereochemistry allows manufacturers to target specific enantiomers required for biological activity, eliminating the need for costly downstream chiral separation processes and significantly improving the overall atom economy of the synthesis.
. This precise control over stereochemistry allows manufacturers to target specific enantiomers required for biological activity, eliminating the need for costly downstream chiral separation processes and significantly improving the overall atom economy of the synthesis.
How to Synthesize Dihydro-9-phenanthrene Amines Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable chiral intermediates. The process begins with the preparation of the specific 9-phenanthrene amine substrate, which can be achieved through various routes such as Suzuki coupling of bromobenzonitriles with phenylboronic acids, followed by cyclization and N-alkylation. Once the substrate is prepared, it is subjected to hydrogenation in the presence of the chiral metal catalyst. The reaction is typically conducted in a high-pressure autoclave using solvents like hexafluoroisopropanol (HFIP), which has been shown to enhance enantioselectivity. Detailed operational parameters, including catalyst loading, hydrogen pressure, and temperature, are critical for maximizing yield and ee values.
- Preparation of the chiral catalyst complex involving a metal center (Ru, Rh, or Ir), an arene or Cp ligand, and a chiral diamine ligand modified with a sulfonyl group.
- Synthesis of the 9-phenanthrene amine substrate (Formula 1) via Suzuki coupling or nitration-reduction pathways followed by N-alkylation.
- Execution of the asymmetric hydrogenation reaction under controlled hydrogen pressure (1-100 atm) and temperature (-10 to 100°C) in suitable solvents like HFIP.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric hydrogenation technology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the production workflow. Traditional methods for obtaining chiral amines often rely on classical resolution techniques, which inherently discard up to half of the produced material as the unwanted enantiomer. By achieving high enantiomeric excess directly through catalysis, this method effectively doubles the theoretical yield from the same amount of starting material, leading to significant raw material savings. Furthermore, the use of hydrogen gas as the reducing agent is inherently cost-effective and environmentally benign compared to stoichiometric reducing agents like borohydrides or aluminum hydrides, which generate substantial waste streams.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps translates directly into reduced processing time and lower solvent consumption. Since the catalyst system is highly active and stable, it can potentially be used at lower loadings, reducing the dependency on precious metals like ruthenium or iridium. Additionally, the mild reaction conditions (often between 20-60°C) reduce energy consumption for heating and cooling compared to processes requiring extreme temperatures. The ability to produce high-purity intermediates without extensive purification steps further lowers the operational expenditure associated with waste treatment and quality control testing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted bromobenzonitriles and phenylboronic acids, are commercially available commodity chemicals with stable supply chains. This reduces the risk of bottlenecks associated with sourcing exotic or custom-synthesized precursors. Moreover, the robustness of the catalyst system ensures consistent batch-to-batch reproducibility, which is critical for maintaining supply continuity for downstream drug manufacturers. The scalability of the high-pressure hydrogenation process is well-established in the fine chemical industry, facilitating a smooth transition from pilot scale to commercial production volumes.
- Scalability and Environmental Compliance: From an environmental perspective, this catalytic hydrogenation route aligns with green chemistry principles by maximizing atom economy and minimizing waste generation. The process avoids the use of heavy metal stoichiometric reagents and produces water as the primary byproduct of hydrogenation. This simplified waste profile eases the burden on effluent treatment facilities and helps manufacturers meet increasingly stringent environmental regulations. The potential for catalyst recycling or recovery further enhances the sustainability profile of the process, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented process is essential for stakeholders evaluating its implementation. The following questions address common concerns regarding catalyst selection, reaction optimization, and product specification. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: How does this method overcome catalyst poisoning by aromatic amines?
A: The patent describes a mechanism where the aromatic amine substrate isomerizes to an exocyclic imine structure under reaction conditions, which is then selectively hydrogenated by the specific chiral catalyst, avoiding direct coordination poisoning of the metal center.
Q: What enantiomeric excess (ee) can be achieved with this technology?
A: Experimental data in the patent demonstrates that using optimized catalysts like (R,R)-7a in solvents such as hexafluoroisopropanol (HFIP) can achieve enantiomeric excess values exceeding 98%, ensuring high optical purity for pharmaceutical applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes relatively mild conditions (e.g., 50 atm H2, 50°C) and robust catalysts that maintain activity over time, making it highly adaptable for commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydro-9-phenanthrene Amine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors capable of safely handling the conditions described in patent CN111825508A. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of chiral dihydro-9-phenanthrene amine meets the exacting standards required for pharmaceutical applications. Our team of expert chemists is ready to optimize the catalyst loading and solvent systems to maximize yield and enantiomeric excess for your specific project needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your overall production costs.
