Advanced Retinal Manufacturing via Stable Diol Intermediates for Commercial Scale-up
The pharmaceutical and nutraceutical industries rely heavily on the consistent supply of high-purity carotenoids, with retinal (Vitamin A aldehyde) serving as a critical foundational building block. Patent CN1277960A introduces a transformative methodology for preparing retinal that circumvents the historical limitations of processing unstable retinol. By leveraging stable diol derivatives synthesized from inexpensive C10 compounds such as linalool or geraniol, this technology offers a robust alternative to traditional oxidation pathways. The innovation lies in the strategic use of sulfone coupling reactions to construct the carbon skeleton, followed by selective oxidation and dehydration steps that preserve the integrity of the sensitive polyene chain. This approach not only enhances the safety profile of the manufacturing process by eliminating the need to handle oxygen-sensitive retinol but also opens new avenues for cost-effective commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of retinal has been plagued by significant technical hurdles, primarily revolving around the instability of the starting materials. Conventional methods often involve the direct oxidation of retinol, a process that demands rigorous exclusion of oxygen, light, and heat to prevent rapid degradation of the substrate. This sensitivity creates substantial bottlenecks in manufacturing, requiring specialized equipment and inert atmospheres that drive up capital expenditure and operational complexity. Furthermore, alternative synthetic routes utilizing beta-ionone or citral as starting materials suffer from high raw material costs and multi-step sequences that accumulate yield losses. The economic burden of these inefficiencies is compounded by the difficulty in controlling impurity profiles, as the fragile nature of the intermediates often leads to the formation of cis-trans isomers and oxidative byproducts that are difficult to separate, ultimately compromising the quality of the final active pharmaceutical ingredient.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing a stable diol derivative as the central intermediate, effectively decoupling the synthesis from the volatility of retinol. This novel approach initiates with the coupling of a cyclic sulfone derivative and an allyl halide derivative, both of which can be derived from readily available and cost-effective C10 terpenes. The resulting sulfone intermediate is then treated with a base to generate the diol derivative, which serves as a robust platform for subsequent functionalization. By stabilizing the reactive centers through the diol structure, the process allows for standard handling and storage conditions, significantly reducing the risk of batch failure. This strategic redesign of the synthetic route ensures that the delicate conjugated double bond system is constructed under controlled conditions, thereby enhancing the overall reliability and reproducibility of the manufacturing process for high-purity OLED material and pharmaceutical precursors.
Mechanistic Insights into Sulfone-Mediated Polyene Construction
The core of this synthetic strategy relies on the precise manipulation of sulfur chemistry to assemble the carbon backbone, a technique that offers superior control over regioselectivity and stereochemistry compared to traditional aldol condensations. The process begins with the generation of a sulfone anion from a cyclic sulfone derivative, which acts as a nucleophile in a coupling reaction with an allyl halide. This Julia-type olefination logic allows for the formation of the critical carbon-carbon double bonds with high fidelity. Following the coupling, the sulfone group serves as a leaving group or is transformed, facilitating the construction of the extended polyene chain found in retinal. The subsequent oxidation of the diol intermediate to a hydroxyaldehyde is typically achieved using selective oxidants such as manganese dioxide, which targets the allylic alcohol functionality without affecting the sensitive double bonds. This chemoselectivity is paramount for maintaining the structural integrity of the molecule and minimizing the formation of over-oxidized impurities that could complicate downstream purification efforts.
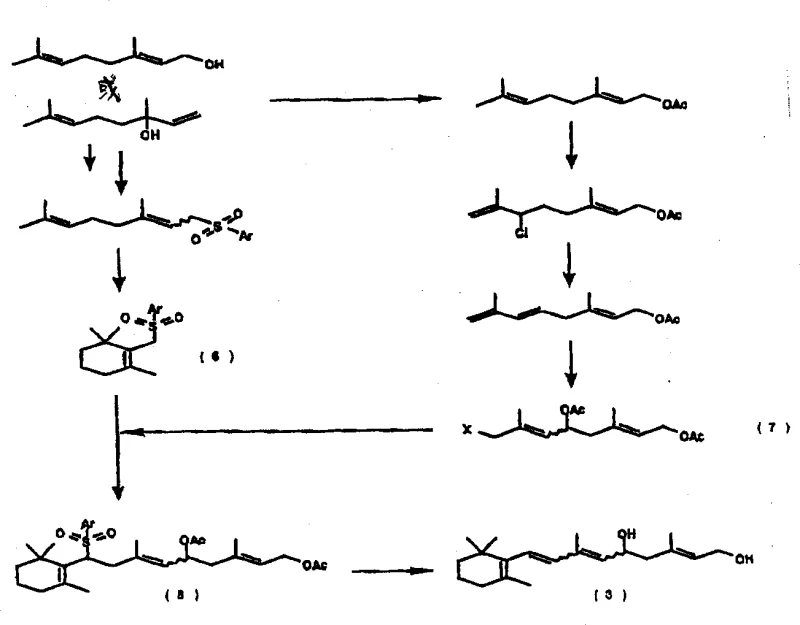
Furthermore, the mechanism includes a critical dehydration or elimination step to finalize the conjugated system. In one embodiment, the hydroxyaldehyde derivative undergoes acid-catalyzed dehydration, where protonation of the hydroxyl group facilitates the loss of water and the formation of the final double bond. Alternatively, the pathway can proceed through a methoxy intermediate, where selective methylation protects one hydroxyl group while the other is oxidized, followed by a base-mediated elimination of the methoxy group. This dual-pathway flexibility allows manufacturers to optimize conditions based on available reagents and desired throughput. The ability to toggle between acid-catalyzed dehydration and base-mediated elimination provides a powerful tool for impurity control, enabling the suppression of unwanted geometric isomers and ensuring that the final product meets the stringent purity specifications required for regulatory approval in the food additive and pharmaceutical sectors.
How to Synthesize Retinal Efficiently
The synthesis of retinal via this patented route involves a sequence of well-defined chemical transformations that prioritize stability and yield. The process begins with the preparation of the key diol intermediate through sulfone coupling, followed by selective oxidation to the aldehyde stage. Detailed operational parameters regarding temperature control, solvent selection, and stoichiometry are critical to maximizing the efficiency of each step. For instance, the oxidation step typically employs manganese dioxide in solvents like dichloromethane or toluene at ambient temperatures to ensure selectivity. The subsequent dehydration requires careful management of acid catalysts to prevent polymerization of the polyene chain. While the general workflow is established, the precise execution requires adherence to standardized protocols to ensure batch-to-batch consistency. The detailed standardized synthesis steps see the guide below.
- Couple cyclic sulfone derivatives with allyl halides to form the key sulfone intermediate, followed by base treatment to generate the stable diol derivative.
- Perform selective oxidation of the diol derivative using manganese dioxide or similar oxidants to obtain the hydroxyaldehyde intermediate.
- Execute acid-catalyzed dehydration or base-mediated elimination of methoxy intermediates to finalize the conjugated polyene system of retinal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this diol-based synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The most significant advantage is the substitution of expensive and volatile raw materials with abundant C10 terpenes like linalool and geraniol, which are produced on a massive scale for the fragrance industry, ensuring a stable and competitive pricing structure. This shift reduces dependency on niche chemical suppliers and mitigates the risk of supply disruptions caused by the limited availability of beta-ionone. Moreover, the enhanced stability of the intermediates translates to reduced waste generation and lower storage costs, as the materials do not require cryogenic preservation or rigorous inert gas blanketing. These factors collectively contribute to substantial cost savings in fine chemical manufacturing, making the production of retinal more resilient against market fluctuations and raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like beta-ionone and the avoidance of complex protection-deprotection sequences associated with retinol handling lead to a streamlined cost structure. By utilizing common oxidants such as manganese dioxide and readily available acid catalysts, the process minimizes the need for exotic reagents that drive up production expenses. Additionally, the higher stability of intermediates reduces the frequency of batch rejections due to degradation, effectively increasing the overall yield of saleable product per unit of raw material input. This efficiency gain is crucial for maintaining competitiveness in the global market for vitamin precursors and carotenoid ingredients.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like linalool and geraniol ensures a robust supply chain that is less susceptible to the bottlenecks often seen with specialized pharmaceutical intermediates. Since these C10 compounds are produced in large volumes for multiple industries, sourcing is straightforward and lead times are predictable. Furthermore, the stability of the diol intermediates allows for the strategic stocking of semi-finished goods, enabling manufacturers to respond rapidly to spikes in demand without the long lead times associated with synthesizing from scratch. This flexibility is a key asset for reducing lead time for high-purity pharmaceutical intermediates and ensuring continuous supply to downstream customers.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The use of standard organic solvents and the absence of highly toxic heavy metal catalysts simplify waste treatment and disposal, aligning with increasingly stringent environmental regulations. The process generates fewer hazardous byproducts compared to traditional oxidation methods, reducing the burden on effluent treatment plants. This environmental compatibility not only lowers compliance costs but also enhances the sustainability profile of the manufacturing operation, a factor that is becoming increasingly important for corporate social responsibility reporting and customer preference.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this retinal synthesis technology. These answers are derived directly from the technical disclosures within the patent documentation, focusing on the practical implications for industrial application. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The responses cover aspects of raw material sourcing, process safety, and product quality control, providing a comprehensive overview of the value proposition offered by this innovative manufacturing route.
Q: Why is the diol derivative route superior to direct retinol oxidation?
A: Direct oxidation of retinol requires handling a compound that is extremely sensitive to heat, light, and oxygen, leading to significant degradation and safety hazards. The diol derivative route utilizes stable intermediates that can be stored and processed under standard industrial conditions, drastically reducing loss and improving operational safety.
Q: What are the primary cost drivers in this synthesis method?
A: The primary cost advantage stems from the use of C10 terpene precursors like linalool or geraniol, which are significantly more abundant and less expensive than traditional C13 beta-ionone or C10 citral starting materials. Additionally, the stability of intermediates reduces waste disposal costs associated with degraded batches.
Q: How is stereochemistry controlled in this process?
A: The process allows for the formation of specific E or Z isomers or their mixtures depending on the reaction conditions during the sulfone coupling and subsequent dehydration steps. The patent details methods to manage these geometric isomers to ensure the final retinal product meets specific biological activity requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Retinal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for essential nutrients and pharmaceutical ingredients. Our technical team has extensively analyzed the potential of the diol-based retinal synthesis route described in CN1277960A and is prepared to implement this technology to meet your specific volume requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of retinal or intermediate delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for carotenoid production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your current procurement model, highlighting exactly where this new synthetic route can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth in the global health and nutrition market.
