Scalable Total Synthesis of Methyl Cajanonic Acid A for Commercial Pharmaceutical Applications
Introduction to Methyl Cajanonic Acid A Synthesis
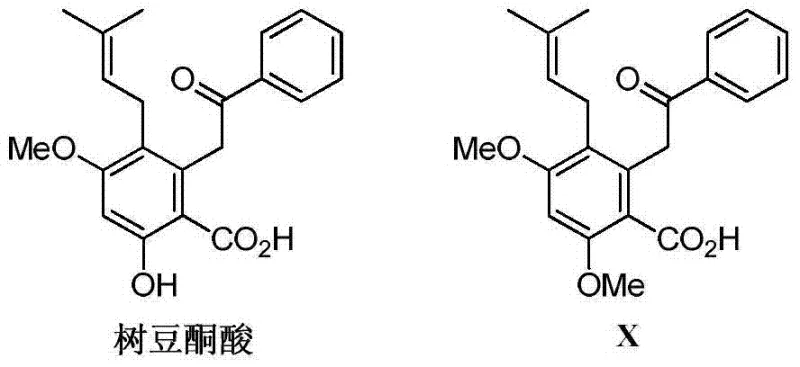
The pharmaceutical industry is constantly seeking robust supply chains for bioactive intermediates, particularly those with metabolic benefits such as hypoglycemic and lipid-lowering properties. Patent CN103772189A introduces a groundbreaking total synthesis method for Methyl Cajanonic Acid A, a structurally modified derivative of the natural product Cajanonic Acid. Historically, obtaining this compound relied heavily on extraction from natural plant sources, a process plagued by low yields, seasonal variability, and complex purification challenges. This patent delineates a precise 13-step chemical synthesis pathway that transforms readily available aromatic starting materials into the high-value target molecule. By shifting from extraction to total synthesis, manufacturers can secure a stable supply of this critical intermediate, ensuring consistent quality and purity essential for downstream drug development. The structural relationship between the natural precursor and the synthesized target highlights the specific pharmacophore modifications aimed at enhancing therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of Cajanonic Acid and its derivatives has been bottlenecked by the constraints of natural product isolation. Extracting bioactive compounds from plant matrices often results in extremely low recovery rates, necessitating the processing of massive quantities of biomass to obtain gram-scale amounts of the pure compound. This approach is not only economically inefficient but also environmentally unsustainable, placing pressure on natural resources. Furthermore, natural extracts inherently contain a complex matrix of impurities, including structurally similar analogs and plant pigments, which require extensive and costly chromatographic purification to meet pharmaceutical grade standards. The variability in plant growth conditions leads to batch-to-batch inconsistency in the content of the active ingredient, posing significant risks for clinical trial reproducibility and regulatory approval. Consequently, relying on natural extraction severely limits the commercial viability of drugs based on these scaffolds.
The Novel Approach
The synthetic methodology disclosed in CN103772189A offers a definitive solution to these supply chain vulnerabilities by establishing a fully synthetic route independent of biological sources. This approach utilizes commodity chemicals such as dimethoxybenzoic acid derivatives as starting points, which are abundant and cost-effective. The 13-step sequence is designed with scalability in mind, employing standard organic transformations like esterification, reduction, and alkylation that are well-understood in industrial settings. By constructing the carbon skeleton chemically, the process allows for precise control over stereochemistry and substitution patterns, ensuring the final product matches the desired structural specifications exactly. This transition from an extraction-based model to a manufacturing-based model fundamentally de-risks the supply chain, enabling the production of Methyl Cajanonic Acid A in kilogram to ton quantities required for commercial drug formulation.
Mechanistic Insights into the 13-Step Synthetic Route
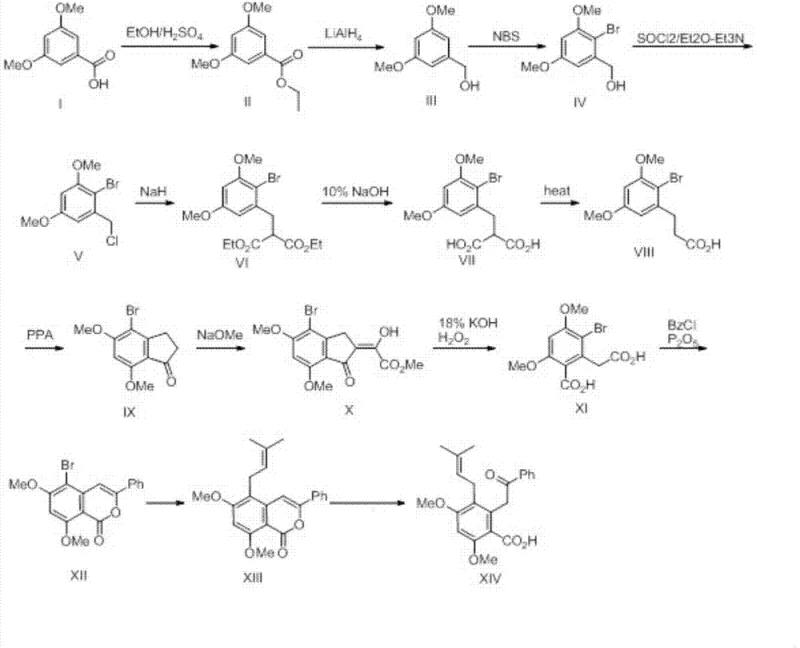
The core of this synthesis lies in the strategic construction of the substituted aromatic core and the subsequent installation of the aliphatic side chains. The pathway begins with the functionalization of the aromatic ring, where esterification and reduction steps convert the carboxylic acid group into a reactive benzyl alcohol. This intermediate is then subjected to halogenation and alkylation sequences to extend the carbon chain, setting the stage for ring closure. A pivotal moment in the synthesis is the polyphosphoric acid (PPA) mediated dehydration cyclization, which efficiently constructs the indanone ring system found in the target molecule. This cyclization step is critical for establishing the rigid core structure that defines the molecule's biological activity. Following ring formation, the synthesis employs an oxidative cleavage strategy using alkaline hydrogen peroxide to open the five-membered ring, generating the necessary dicarbonyl or acid functionalities required for the final assembly.
The final stages of the synthesis demonstrate sophisticated organometallic chemistry to install the specific prenyl side chain. A palladium-catalyzed cross-coupling reaction is utilized to attach the tributyl(3-methylbut-2-enyl)tin moiety to the aromatic core. This step requires careful optimization of catalyst loading and reaction temperature to maximize yield while minimizing the formation of homocoupling byproducts. The use of a palladium catalyst, specifically dichloro[1,1'-bis(diphenylphosphino)ferrocene]palladium(II), ensures high selectivity for the carbon-carbon bond formation. The entire route is meticulously planned to minimize protecting group manipulations, thereby improving the overall atom economy. The final hydrolysis step liberates the free carboxylic acid, yielding Methyl Cajanonic Acid A with high purity, ready for salt formation or further derivatization.
How to Synthesize Methyl Cajanonic Acid A Efficiently
The synthesis of Methyl Cajanonic Acid A described in the patent provides a detailed roadmap for laboratory and pilot-scale production. The process is divided into distinct phases: core construction, ring manipulation, and side-chain installation. Each phase utilizes standard reagents and solvents, facilitating easy technology transfer to manufacturing facilities. The protocol emphasizes strict control over reaction temperatures and stoichiometry, particularly during the exothermic reduction and cyclization steps, to ensure safety and reproducibility. For R&D teams looking to replicate or optimize this pathway, attention should be paid to the purification methods between steps, as the patent suggests techniques like crystallization and column chromatography to maintain high purity throughout the sequence. The detailed标准化 synthesis steps见下方的指南。
- Esterification and Reduction: Convert the starting benzoic acid derivative to the corresponding alcohol via ethyl ester formation and LiAlH4 reduction.
- Chain Extension and Cyclization: Perform alkylation with diethyl malonate, followed by hydrolysis, decarboxylation, and PPA-mediated dehydration cyclization to form the indanone core.
- Functionalization and Coupling: Execute oxidative ring opening, condensation with benzoyl chloride, Pd-catalyzed prenylation, and final ester hydrolysis to yield the target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to a synthetic route for Methyl Cajanonic Acid A represents a significant strategic advantage in terms of cost stability and supply security. Unlike natural extraction, which is subject to agricultural failures and price volatility of raw plant materials, this chemical synthesis relies on petrochemical-derived feedstocks that have established global markets and stable pricing. The ability to produce the compound on demand reduces the need for large inventory buffers, freeing up working capital. Furthermore, the synthetic route avoids the use of exotic or highly regulated reagents, simplifying the logistics of raw material sourcing and storage. The process is designed to be scalable, meaning that production volumes can be increased linearly with market demand without the need for fundamental process re-engineering.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes inexpensive starting materials such as dimethoxybenzoic acid derivatives and common reagents like thionyl chloride and sodium hydride. By eliminating the expensive and inefficient extraction processes associated with natural products, the overall cost of goods sold (COGS) is drastically reduced. The high yields reported in key steps, such as the initial esterification and alkylation reactions, contribute to a favorable mass balance, minimizing waste disposal costs. Additionally, the avoidance of complex chiral resolution steps, which are often cost-prohibitive, further enhances the economic viability of this manufacturing route.
- Enhanced Supply Chain Reliability: Dependence on a single geographic source for plant extraction creates a single point of failure in the supply chain. In contrast, the chemical synthesis described allows for multi-vendor sourcing of raw materials, as the precursors are commodity chemicals available from numerous global suppliers. This diversification mitigates the risk of supply disruptions caused by geopolitical issues or natural disasters affecting crop yields. The robust nature of the chemical steps ensures consistent lead times, allowing pharmaceutical companies to plan their production schedules with greater confidence and accuracy.
- Scalability and Environmental Compliance: The process is amenable to large-scale batch processing, utilizing standard reactor types found in most fine chemical manufacturing plants. The waste streams generated are primarily aqueous and organic solvents which can be managed through established recovery and treatment protocols, ensuring compliance with environmental regulations. The elimination of agricultural land use for raw material production also aligns with sustainability goals by reducing the ecological footprint associated with the manufacturing of this pharmaceutical intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Methyl Cajanonic Acid A. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their drug development pipelines. The information covers aspects of purity, scalability, and regulatory considerations relevant to the pharmaceutical industry.
Q: What are the advantages of synthesizing Methyl Cajanonic Acid A over natural extraction?
A: Synthetic production eliminates dependence on agricultural variables, ensures consistent purity profiles, and allows for industrial-scale manufacturing that natural extraction cannot support due to low natural abundance.
Q: What is the critical step in the synthesis of Methyl Cajanonic Acid A?
A: The palladium-catalyzed coupling reaction (Step 12) to install the prenyl side chain and the oxidative cleavage of the five-membered ring (Step 10) are critical for establishing the correct pharmacophore structure.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the patent explicitly states the method uses simple, readily available raw materials and standard unit operations like esterification, reduction, and crystallization, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Cajanonic Acid A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation antidiabetic therapies. Our team of expert chemists has thoroughly analyzed the synthetic route disclosed in CN103772189A and possesses the technical capability to execute this complex 13-step sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met regardless of the project phase. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Methyl Cajanonic Acid A meets the highest international standards.
We invite you to collaborate with us to secure a stable and cost-effective supply of this valuable intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us support your journey from clinical trials to commercial launch with our reliable manufacturing expertise.
