Advanced Synthesis of Chiral Alpha-Methyl Aromatic Ethylamine for Scalable API Manufacturing
Advanced Synthesis of Chiral Alpha-Methyl Aromatic Ethylamine for Scalable API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for chiral building blocks that serve as the backbone for life-saving medications. Patent CN112920053A introduces a groundbreaking preparation method for chiral alpha-methyl aromatic ethylamine, a critical structural motif found in potent asthma treatments like Formoterol and Carmoterol, as well as emerging oncology agents such as AZD9496. This technology addresses the longstanding challenges of safety, cost, and environmental impact associated with traditional chiral amine synthesis. By leveraging a unique sulfonamide intermediate strategy, the process bypasses the need for hazardous high-pressure hydrogenation and wasteful chiral resolution techniques. For R&D directors and procurement leaders, this represents a paradigm shift towards greener, more economical manufacturing of high-value pharmaceutical intermediates.
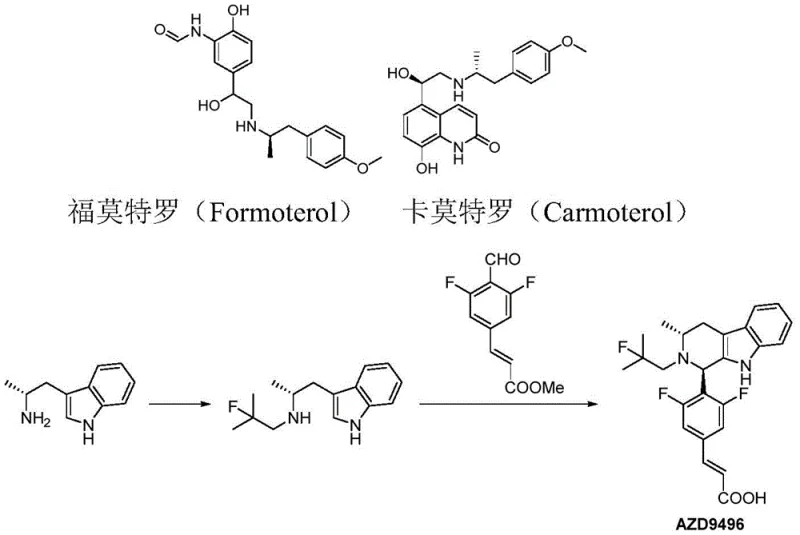
The significance of this technology is best understood by examining the target molecules it enables. As illustrated in the chemical structures above, the chiral alpha-methyl aromatic ethylamine fragment is indispensable for the biological activity of these drugs. Formoterol and Carmoterol rely on specific stereochemistry to act as long-acting beta-agonists for respiratory diseases, while AZD9496 utilizes a similar chiral indole fragment for estrogen receptor modulation in breast cancer therapy. The ability to produce these fragments with high optical purity and yield directly impacts the quality and availability of the final Active Pharmaceutical Ingredients (APIs). This patent provides a versatile platform applicable to various aryl groups, including methoxy-phenyl and indolyl derivatives, making it a highly valuable asset for diverse drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alpha-methyl aromatic ethylamines has been plagued by significant technical and economic hurdles. Prior art methods, such as those described in WO2017172957, often rely on complex multi-step sequences culminating in high-pressure hydrogenation reactions catalyzed by palladium. These processes not only require specialized, expensive equipment to handle high-pressure hydrogen gas safely but also introduce substantial operational risks in a manufacturing environment. Furthermore, alternative approaches involving the resolution of racemic mixtures or diastereomers, as seen in literature from the Journal of Medicinal Chemistry, suffer from poor atom economy. These resolution methods inherently discard up to 50% of the material as unwanted isomers, generating massive amounts of solid waste from silica gel chromatography and driving up production costs unnecessarily.
The Novel Approach
In stark contrast, the method disclosed in CN112920053A offers a streamlined, chiral-pool-based solution that elegantly circumvents these issues. The novel route begins with readily available Boc-protected amino acid methyl esters, which already possess the desired stereochemistry. Through a sequence of reduction, chlorination, and selective oxidation, the process generates a stable sulfonamide intermediate. This intermediate is then subjected to a mild Lewis acid-promoted reduction, effectively removing the oxygen functionality while retaining the chiral center intact. Finally, simple acidic deprotection yields the target amine. This approach eliminates the need for dangerous high-pressure hydrogenation and avoids the generation of isomeric waste entirely, resulting in a process that is inherently safer, cleaner, and more cost-effective for large-scale industrial application.
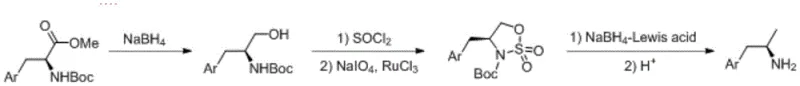
Mechanistic Insights into Sulfonamide-Mediated Stereospecific Reduction
The core innovation of this patent lies in the transformation of the Boc-amino alcohol into a sulfonamide compound, which serves as a superior leaving group precursor for the subsequent reduction. In the second step of the synthesis, the Boc-amino alcohol is first converted to a chloro-intermediate using thionyl chloride. This is followed by a sophisticated oxidation step utilizing sodium periodate (NaIO4) catalyzed by ruthenium trichloride trihydrate (RuCl3·3H2O). This specific oxidative condition converts the sulfur moiety into a sulfonamide group. The presence of the sulfonamide group significantly activates the adjacent carbon center for nucleophilic attack, allowing for a smooth reduction in the subsequent step without the need for harsh conditions that might racemize the chiral center. This mechanistic pathway ensures that the stereochemical integrity established by the starting chiral amino acid is preserved throughout the synthesis.
Furthermore, the final reduction step employs a Lewis acid-promoted system using sodium borohydride (NaBH4) in solvents like diethylene glycol dimethyl ether. The addition of Lewis acids such as anhydrous lithium chloride or zinc chloride is critical; it coordinates with the sulfonamide oxygen atoms, enhancing the electrophilicity of the sulfur-nitrogen bond and facilitating the hydride transfer. This allows the reaction to proceed efficiently at moderate temperatures (50-120°C) rather than requiring extreme conditions. The subsequent acidic deprotection removes the Boc group cleanly. This mechanism effectively replaces the traditional catalytic hydrogenation, avoiding the use of pyrophoric catalysts like Raney Nickel or Palladium on Carbon, thereby simplifying the workup procedure and minimizing the risk of metal contamination in the final pharmaceutical intermediate.
How to Synthesize (R)-Alpha-Methyl-4-Methoxyphenethylamine Efficiently
The practical implementation of this synthesis involves precise control over reaction parameters to maximize yield and purity. The process begins with the reduction of the ester starting material at low temperatures (0-10°C) to prevent side reactions, followed by a carefully monitored oxidation phase where temperature control below 5°C is vital for the stability of the ruthenium catalyst system. The final reduction requires specific solvent choices, such as diglyme or dioxane, to ensure solubility and reactivity of the sulfonamide intermediate. While the general principles are straightforward, the exact stoichiometry and thermal profiles are critical for success. For laboratory and pilot plant teams looking to implement this technology, adhering to the standardized operational procedures is essential to replicate the high purity (>99%) and excellent yields reported in the patent examples.
- Reduce Boc-amino acid methyl ester using sodium borohydride to obtain the corresponding Boc-amino alcohol intermediate.
- React the amino alcohol with thionyl chloride followed by sodium periodate oxidation catalyzed by ruthenium trichloride to form a stable sulfonamide compound.
- Perform Lewis acid-promoted reduction of the sulfonamide using sodium borohydride, followed by acidic deprotection to isolate the target chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling strategic advantages beyond mere technical feasibility. The shift away from high-pressure hydrogenation fundamentally alters the capital expenditure requirements for manufacturing facilities, allowing production to occur in standard glass-lined reactors rather than specialized high-pressure autoclaves. This flexibility significantly enhances supply chain resilience by enabling production across a wider range of contract manufacturing organizations (CMOs) without the bottleneck of limited high-pressure capacity. Additionally, the use of commodity-grade starting materials like Boc-amino acids ensures a stable and predictable raw material supply, insulating the project from the volatility often associated with specialized chiral catalysts or auxiliaries.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of wasteful resolution steps and expensive catalysts. Traditional resolution methods discard half of the synthesized material, effectively doubling the raw material cost for the active isomer. By utilizing a chiral pool approach, this method achieves near-theoretical atom economy, ensuring that every gram of starting material contributes to the final product. Furthermore, the removal of noble metal catalysts like palladium eliminates the costly recovery and purification steps required to meet stringent residual metal specifications in pharmaceuticals, leading to substantial overall cost savings in the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes. This simplified three-step sequence reduces the number of unit operations and intermediate isolations, thereby decreasing the probability of batch failures and delays. The avoidance of column chromatography, which is difficult to scale and prone to variability, in favor of crystallization-based purifications ensures a more robust and reproducible manufacturing process. This reliability translates directly to shorter lead times and a more dependable supply of high-purity pharmaceutical intermediates for downstream API synthesis, securing the production schedule for finished dosage forms.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with modern green chemistry principles. The avoidance of silica gel waste and the reduction of solvent usage through efficient crystallization steps significantly lower the environmental footprint of the manufacturing process. This not only reduces waste disposal costs but also simplifies regulatory filings related to impurity profiles and solvent residues. The mild reaction conditions and lack of high-pressure operations make the process inherently safer to scale from kilogram to multi-ton quantities, facilitating a smoother technology transfer from R&D to commercial production without the need for extensive safety re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral amine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes in terms of safety, efficiency, and product quality. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis method improve safety compared to traditional hydrogenation?
A: Traditional methods often require high-pressure hydrogenation with palladium catalysts, posing significant explosion risks. This patent utilizes a mild Lewis acid-promoted reduction system at atmospheric pressure, drastically reducing operational hazards and equipment costs.
Q: Does this process eliminate the need for chiral resolution?
A: Yes. By starting with commercially available chiral Boc-amino acids, the stereochemistry is inherited directly from the chiral pool. This avoids the generation of unwanted diastereomers and eliminates the wasteful column chromatography or recrystallization steps typically required for resolution.
Q: What represent the key cost-saving drivers in this manufacturing route?
A: The primary cost drivers are the use of inexpensive, commodity-grade starting materials and the elimination of expensive chiral auxiliaries or noble metal catalysts. Furthermore, the high atom economy and avoidance of solid waste from silica gel purification significantly lower disposal and material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Methyl Aromatic Ethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. Whether you require custom synthesis of complex intermediates or reliable supply of established building blocks, our infrastructure is designed to meet the demanding needs of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific drug development projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce manufacturing costs while maintaining the highest standards of quality and compliance.
