Revolutionizing Alkyl Thiosulfonate Production via Direct C-H Bond Activation for Commercial Scale
Revolutionizing Alkyl Thiosulfonate Production via Direct C-H Bond Activation for Commercial Scale
The chemical industry is constantly seeking more efficient pathways to synthesize sulfur-containing compounds, which are critical building blocks in pharmaceuticals and advanced materials. Patent CN113061106B introduces a groundbreaking methodology for the preparation of alkyl thiosulfonate compounds through a direct coupling mechanism. This innovation leverages the power of photochemical activation to transform simple alkanes into valuable thiosulfonates without the need for complex pre-functionalization. By utilizing a direct thionation mode of alkane C-H bonds under acidic and illuminated conditions, this process represents a significant leap forward in green chemistry. The ability to complete the conversion in a single step not only streamlines the synthetic route but also drastically reduces the environmental footprint associated with traditional multi-step syntheses. For R&D directors and procurement managers alike, this technology offers a compelling value proposition centered on simplicity, cost-efficiency, and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for thiosulfonates often suffer from inherent inefficiencies that hinder large-scale commercial adoption. Conventional methods typically require the pre-configuration of specific reaction functional groups, which necessitates additional synthetic steps and increases the overall complexity of the process. These multi-step sequences often involve expensive reagents that are difficult to store and handle safely, posing logistical challenges for supply chain managers. Furthermore, many existing methods are limited in their substrate scope, failing to accommodate a diverse range of alkane structures required for specialized applications. The reliance on harsh reaction conditions, such as high temperatures or strong oxidizing agents, can lead to safety hazards and increased energy consumption. These factors collectively contribute to higher production costs and longer lead times, making conventional methods less attractive for modern manufacturing environments that prioritize sustainability and economic viability.
The Novel Approach
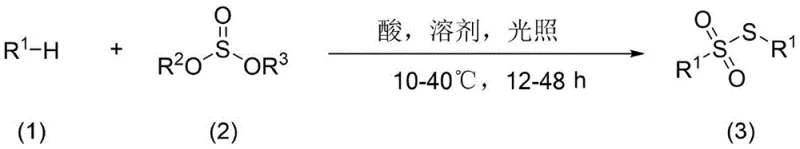
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a green and environment-friendly photoreaction mode to activate alkane C-H bonds directly. This method bypasses the need for pre-functionalization entirely, allowing for the direct construction of C-S bonds in a single operational step. The reaction proceeds under mild conditions, typically between 10-40°C, eliminating the need for energy-intensive heating systems. By employing cheap and easily obtained raw materials such as cyclohexane and sodium sulfite, the process significantly lowers the barrier to entry for production. The simplicity of the operation, combined with the safety of avoiding hazardous reagents, makes this approach highly beneficial for industrial production. This shift from complex, multi-step syntheses to a streamlined, one-step photochemical process represents a paradigm shift in how sulfur-containing intermediates are manufactured, offering clear advantages in both operational efficiency and cost structure.
Mechanistic Insights into Photochemical C-H Activation
The core of this innovation lies in the mechanistic ability to activate inert alkane C-H bonds using light energy. In the presence of an acid catalyst and illumination, the alkane substrate undergoes homolytic cleavage to generate alkyl free radicals. These highly reactive intermediates are then coupled with a sulfur-containing module, specifically a sulfurous acid derivative, to form the target alkyl thiosulfonate compound. This radical thionation reaction is facilitated by the specific wavelength of light, which can range from ultraviolet to visible light (200nm-800nm), providing flexibility in reactor design. The acid catalyst, which can be a protonic acid like hydrochloric acid or a Lewis acid like aluminum trichloride, plays a crucial role in stabilizing the transition state and promoting the coupling efficiency. Understanding this mechanism is vital for R&D teams looking to optimize reaction parameters for specific substrates.
Controlling the impurity profile is another critical aspect of this mechanistic pathway. The direct nature of the reaction minimizes the formation of side products that are commonly associated with multi-step functional group interconversions. By avoiding the use of transition metal catalysts, the process eliminates the risk of heavy metal contamination, which is a significant concern in pharmaceutical manufacturing. The selectivity of the radical reaction is governed by the bond dissociation energies of the C-H bonds and the specific reaction conditions, such as solvent choice and molar ratios. The patent specifies the use of aprotic solvents like ethyl acetate or acetonitrile, which do not interfere with the radical mechanism. This high level of control over the reaction environment ensures that the final product meets stringent purity specifications, reducing the need for extensive downstream purification and thereby enhancing the overall yield and economic efficiency of the process.
How to Synthesize Alkyl Thiosulfonate Efficiently
Implementing this synthesis route requires careful attention to the specific reaction parameters outlined in the patent to ensure optimal yields and reproducibility. The process involves dissolving the alkane and sulfurous acid derivative in a suitable aprotic solvent along with an acid catalyst. The mixture is then subjected to illumination under an inert atmosphere to prevent unwanted oxidation side reactions. While the general principle is straightforward, the specific stoichiometry and reaction time can vary depending on the substrate. For instance, the molar ratio of alkane to sulfite to acid is typically maintained within a specific range to maximize conversion. The following guide outlines the standardized steps derived from the patent examples to assist technical teams in replicating this efficient synthesis.
- Mix alkane substrate and sulfurous acid derivative in an aprotic solvent with an acid catalyst.
- Irradiate the reaction mixture with UV or visible light (200nm-800nm) at mild temperatures (10-40°C).
- Purify the crude product via column chromatography to obtain high-purity alkyl thiosulfonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical synthesis method offers substantial strategic advantages. The elimination of expensive pre-functionalized starting materials directly translates to a reduction in raw material costs. Since the process utilizes commodity chemicals like cyclohexane and sodium sulfite, the supply chain is less vulnerable to fluctuations in the prices of specialized reagents. Furthermore, the simplified workflow reduces the operational complexity, allowing for faster turnaround times from raw material intake to finished product. This efficiency is crucial for maintaining supply continuity in a volatile market. The ability to produce high-value sulfur intermediates from low-cost alkanes represents a significant opportunity for margin improvement and cost leadership in the fine chemical sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the synthetic route. By removing the need for multiple reaction steps and expensive catalysts, the overall cost of goods sold is significantly lowered. The absence of transition metals means there is no need for costly metal scavenging steps, which further reduces processing expenses. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility costs. These cumulative savings allow manufacturers to offer more competitive pricing while maintaining healthy profit margins, making it an attractive option for cost-sensitive applications in the pharmaceutical and agrochemical industries.
- Enhanced Supply Chain Reliability: Reliability is a cornerstone of effective supply chain management, and this method enhances it through the use of readily available raw materials. Alkanes and sulfites are produced in large volumes globally, ensuring a stable and consistent supply base. This reduces the risk of production delays caused by raw material shortages, which are common with specialized intermediates. The robustness of the reaction conditions also means that the process is less prone to failures due to minor variations in input quality. For supply chain heads, this translates to greater predictability in production schedules and the ability to meet customer demand consistently without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this photochemical method is inherently scalable due to its simplicity. The one-step nature of the reaction reduces the equipment footprint required for production, allowing for higher throughput in existing facilities. From an environmental perspective, the green chemistry principles embedded in this method, such as waste reduction and energy efficiency, facilitate compliance with increasingly stringent environmental regulations. The absence of heavy metals and hazardous byproducts simplifies waste treatment and disposal, reducing the environmental liability associated with manufacturing. This alignment with sustainability goals is increasingly important for corporate social responsibility and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation. They are intended to provide clarity on the feasibility, scope, and operational requirements of the process. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into their existing manufacturing portfolios. The responses highlight the versatility of the method and its alignment with modern industrial standards for safety and efficiency.
Q: What are the primary advantages of this photochemical synthesis method?
A: This method eliminates the need for pre-functionalized groups, allowing direct C-H bond activation. It operates under mild conditions (10-40°C) without heating, uses cheap raw materials like cyclohexane, and achieves high yields in a single step, significantly simplifying the process compared to traditional multi-step routes.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is highly amenable to scale-up. It utilizes inexpensive and readily available alkanes and sulfites, avoids expensive transition metal catalysts, and operates at ambient pressure and low temperatures. The simple workup procedure involving solvent removal and chromatography supports efficient large-scale production.
Q: What types of light sources are compatible with this reaction?
A: The patent specifies compatibility with a broad range of light sources, including ultraviolet light and visible light such as blue LEDs (405nm, 380nm). This flexibility allows manufacturers to optimize energy consumption and reactor design based on available industrial lighting infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Thiosulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical synthesis route described in patent CN113061106B for the production of high-purity alkyl thiosulfonates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory method into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this technology are realized at an industrial scale. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and fine chemical industries.
We invite you to collaborate with us to optimize your supply chain for sulfur-containing intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging this advanced synthesis method, we can help you achieve significant reductions in manufacturing costs while improving supply reliability. We encourage you to contact us to request specific COA data and route feasibility assessments for your target compounds. Together, we can drive innovation and efficiency in the production of critical chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
