Scalable Production of 5-Oxohexanoate: A Novel Catalytic Michael Addition Strategy for Fine Chemical Manufacturing
The global demand for high-purity ketone esters, specifically 5-oxohexanoate derivatives, has surged due to their critical role as versatile building blocks in the synthesis of complex pharmaceutical active ingredients and agrochemical agents. As the industry seeks more sustainable and economically viable manufacturing routes, recent intellectual property developments have shed light on significant process improvements. Specifically, the methodology disclosed in Chinese Patent CN114516796A presents a groundbreaking approach to synthesizing 5-oxohexanoate through a direct Michael addition reaction. This patent details a novel catalytic system that overcomes the historical limitations of low selectivity and harsh reaction conditions associated with traditional synthesis pathways. By utilizing readily available raw materials such as acetone and various acrylate compounds in the presence of specific dual-function catalysts, this technology offers a robust alternative for reliable pharmaceutical intermediate supplier networks looking to optimize their supply chains. The core innovation lies in the strategic selection of catalysts that possess both acidic and basic centers, a feature that fundamentally alters the reaction landscape to favor the desired product while suppressing deleterious side reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-oxohexanoate has been plagued by significant technical and economic hurdles that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Prior art methods often rely on expensive or hazardous reagents that complicate downstream processing and increase environmental liabilities. For instance, earlier literature describes the use of onium tetrafluoroborate catalysts under solvent-free conditions at elevated temperatures, which, while yielding moderate results, involves catalysts that are prohibitively expensive for large-scale industrial application and prone to deactivation. Furthermore, traditional acid-catalyzed routes using concentrated sulfuric acid generate substantial acidic waste streams, posing severe challenges for environmental compliance and waste treatment infrastructure. Another notable failure in prior art, as highlighted in comparative studies within the patent, involves the use of secondary amines like tetrahydropyrrole. In these scenarios, the formation of an enamine intermediate between acetone and the amine catalyst creates a kinetic trap where the intermediate fails to effectively undergo Michael addition with the acrylate, leading to negligible product formation. This specific failure mode is visually represented in the reaction scheme below, illustrating why simple amine catalysis is insufficient for this transformation.
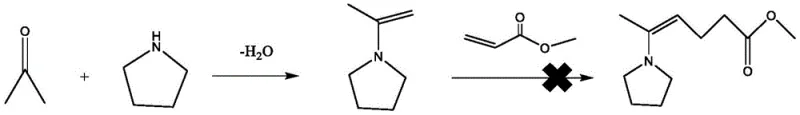
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in CN114516796A leverages a sophisticated understanding of acid-base bifunctional catalysis to drive the reaction forward with remarkable efficiency. By employing catalysts such as dipotassium hydrogen phosphate (K2HPO4), sodium bicarbonate (NaHCO3), or specific amine hydrochloride salts, the process creates a reaction environment that is simultaneously capable of activating the nucleophile and managing the acidity of the system. This method operates under relatively mild thermal conditions, typically ranging from 20°C to 120°C, which drastically reduces energy consumption compared to high-temperature solvent-free protocols. The use of acetone as both a reactant and a solvent further simplifies the process flow, eliminating the need for exotic solvents and reducing the overall material footprint. Most critically, this approach addresses the pervasive issue of acrylate self-polymerization caused by commercial polymerization inhibitors. The unique chemical nature of the selected catalysts allows them to neutralize these acidic inhibitors without being consumed or deactivated, ensuring that the acrylate remains available for the desired carbon-carbon bond formation. This results in a streamlined process that delivers high-purity 5-oxohexanoate with minimal byproduct formation, setting a new standard for commercial scale-up of complex fine chemicals.
Mechanistic Insights into Dual-Function Catalyzed Michael Addition
The success of this synthetic route hinges on the precise mechanistic interplay between the ketone enolate and the activated olefin, mediated by the unique properties of the catalyst. The reaction proceeds via a classic Michael addition mechanism where the alpha-proton of the acetone is abstracted to form a nucleophilic enolate species. However, unlike traditional base-catalyzed reactions where strong bases can lead to uncontrolled polymerization or multiple additions, the catalysts used here modulate the basicity to ensure selective mono-addition. The catalyst acts as a proton shuttle, facilitating the deprotonation of acetone while simultaneously stabilizing the transition state. Crucially, the presence of an acidic center within the catalyst structure (or the conjugate acid formed in situ) plays a pivotal role in managing the reaction medium's pH. This prevents the accumulation of highly reactive basic species that could otherwise attack the ester moiety of the product or initiate radical polymerization of the acrylate monomer. The detailed mechanistic pathway, including the enolization step, the nucleophilic attack on the beta-carbon of the acrylate, and the subsequent protonation to yield the final keto-ester, is illustrated in the following scheme. This visualization underscores how the electronic properties of the catalyst govern the reaction trajectory, ensuring that the thermodynamic product is formed with high fidelity.
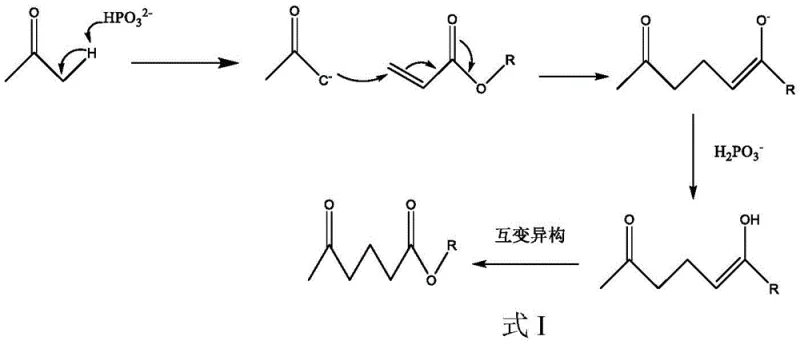
Furthermore, the control of impurities is intrinsically linked to this mechanistic design. In many competing processes, the primary impurity profile includes oligomers of the acrylate or over-alkylated ketone products. The dual-function catalyst system mitigates these risks by maintaining a steady-state concentration of the active enolate that is sufficient for reaction but low enough to prevent runaway side reactions. The patent data indicates a Michael addition selectivity exceeding 92%, which is a testament to the precision of this catalytic system. By avoiding the use of transition metals, the process also eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for downstream API synthesis. This inherent purity advantage reduces the burden on purification units, allowing for simpler workup procedures such as distillation rather than complex chromatographic separations, thereby enhancing the overall process mass intensity (PMI) and sustainability metrics of the manufacturing campaign.
How to Synthesize 5-Oxohexanoate Efficiently
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and selectivity. The protocol involves dissolving acetone in a solvent, introducing the catalyst, and controlling the addition rate of the acrylate to manage exotherms and local concentration gradients. While the general procedure is robust, optimizing the molar ratios and temperature profiles is essential for achieving the reported performance metrics. The following guide outlines the standardized operational steps derived from the patent examples, providing a clear roadmap for laboratory validation and pilot plant trials.
- Dissolve acetone in a suitable solvent and add a dual-function catalyst such as K2HPO4 or NaHCO3.
- Maintain the reaction temperature between 20-120°C and slowly add the acrylate compound solution over 2-6 hours.
- Continue stirring for 1-8 hours post-addition, then remove residual solvents and reactants via rotary evaporation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards this methodology represents a significant opportunity for cost reduction in fine chemical manufacturing by fundamentally altering the input cost structure. By replacing expensive, specialized catalysts with commodity chemicals like potassium phosphate or sodium bicarbonate, the direct material costs are drastically reduced. Moreover, the elimination of harsh acids like sulfuric acid removes the need for specialized corrosion-resistant equipment and expensive neutralization waste streams, leading to substantial operational expenditure savings. The simplicity of the workup, which primarily involves the removal of volatile components via distillation, contrasts sharply with the labor-intensive column chromatography required by some legacy methods, further driving down processing costs and cycle times.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of acetone, one of the most abundant and inexpensive organic solvents and reagents globally. Unlike methods requiring 1,3-cyclohexanedione or other specialized precursors, the reliance on bulk commodities insulates the production cost from volatile niche market fluctuations. Additionally, the high selectivity of the reaction minimizes the loss of valuable acrylate starting materials to polymeric waste, ensuring that a greater proportion of input mass is converted into saleable product. This efficiency translates directly into improved margins and a more competitive pricing structure for the final intermediate, making it an attractive option for long-term supply contracts.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry enhances security of supply. The catalysts employed are stable, non-hazardous solids that are easily sourced from multiple global suppliers, reducing the risk of single-source bottlenecks. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without requiring exotic metallurgy, meaning that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant capital investment. This flexibility ensures reducing lead time for high-purity intermediates by allowing for rapid scale-up and diversified production capacity, safeguarding against disruptions in the global logistics network.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with modern green chemistry principles and increasingly stringent regulatory frameworks. The absence of heavy metals and the use of benign catalysts simplify the environmental permitting process and reduce the liability associated with hazardous waste disposal. The process generates minimal aqueous waste compared to acid-catalyzed routes, and the potential for solvent recycling (particularly if acetone is used in excess) further enhances its sustainability credentials. For multinational corporations with aggressive carbon reduction targets, adopting this lower-energy, lower-waste pathway contributes directly to Scope 3 emissions reductions, adding value to the end product beyond its chemical utility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance expectations and operational constraints.
Q: Why are dual-function catalysts preferred over traditional bases for this synthesis?
A: Traditional strong bases often react with polymerization inhibitors in commercial acrylates, causing self-polymerization. Dual-function catalysts possess both acidic and basic centers, neutralizing inhibitors while catalyzing the Michael addition, thereby significantly improving selectivity.
Q: What are the typical yields and selectivity for this process?
A: According to patent data, the process achieves a Michael addition selectivity of over 92% with isolated yields reaching up to 37%, depending on the specific acrylate and catalyst used.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes inexpensive raw materials like acetone and avoids expensive transition metal catalysts or harsh acids, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Oxohexanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. We have closely analyzed the potential of the catalytic Michael addition route described in CN114516796A and are well-positioned to leverage this technology for our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising selectivity and yield data observed at the gram scale can be faithfully reproduced in multi-ton batches. Our facilities are equipped with rigorous QC labs and stringent purity specifications protocols, guaranteeing that every batch of 5-oxohexanoate meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to evaluate the feasibility of integrating this cost-effective synthesis route into your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us to obtain specific COA data from our pilot runs and to discuss detailed route feasibility assessments. Let us help you secure a sustainable, high-quality supply of this critical intermediate while optimizing your overall production economics.
