Scalable Manufacturing of 6-Methoxycarbonyl Indolone for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously demands more efficient and safer pathways for synthesizing critical intermediates, particularly those serving Phase III clinical candidates. A pivotal advancement in this domain is detailed in patent CN102267934A, which outlines a novel method for preparing 6-methoxycarbonyl indolone, a key structural motif in various bioactive molecules. This technology represents a significant departure from legacy synthetic strategies, addressing long-standing issues regarding selectivity, toxicity, and scalability. By leveraging a unique sequence involving esterification, condensation, and oxidative decarbonylation, the process achieves a robust yield of over 70% with exceptional purity profiles. For R&D directors and supply chain leaders, understanding this methodology is crucial for securing a reliable supply of high-purity pharmaceutical intermediates while mitigating regulatory and operational risks associated with older, more hazardous chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-methoxycarbonyl indolone has been plagued by significant chemical and operational challenges that hinder efficient commercial production. One prevalent conventional route involves the alkylation of methyl m-nitrobenzoate with methyl chloroacetate, followed by catalytic reduction. As illustrated in the reaction scheme below, this approach suffers from inherently poor regioselectivity during the alkylation step on the phenyl ring. This lack of control inevitably leads to the formation of unwanted isomers alongside the target compound, drastically lowering the total recovery rate and complicating the purification process. The presence of these structural impurities often necessitates multiple recrystallization steps or chromatographic separations, which are cost-prohibitive at an industrial scale.
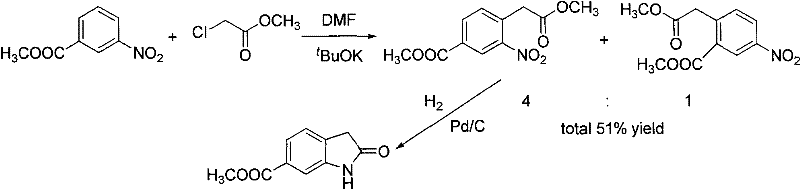
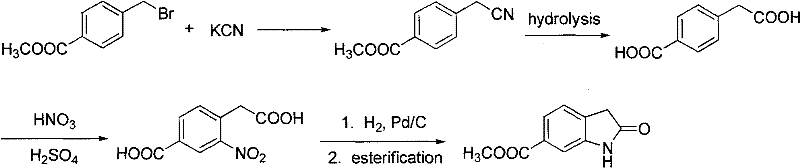
Furthermore, alternative historical pathways have relied on the use of highly toxic reagents such as sodium cyanide to extend the carbon chain. In these methods, methoxycarbonyl benzyl bromide is reacted with potassium cyanide, followed by hydrolysis, nitration, and reduction. While chemically feasible, the reliance on cyanide introduces severe safety hazards for operators and creates substantial environmental liabilities due to the generation of toxic waste streams. These factors make such routes increasingly untenable in modern manufacturing environments where strict adherence to green chemistry principles and operator safety is paramount. The combination of low yields, difficult purification, and high toxicity renders these conventional methods unsuitable for the reliable, large-scale supply required by the global pharmaceutical market.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a strategically designed four-step sequence that circumvents both selectivity issues and toxic reagent usage. Starting from readily available 3-nitro-4-methyl benzoic acid, the process employs a Claisen-type condensation with an oxalic ester derivative to build the necessary carbon framework. This is followed by a sophisticated oxidative decarbonylation step that precisely trims the side chain to the desired length without generating the isomeric by-products seen in alkylation routes. The final stage involves a tandem reduction and cyclization that efficiently closes the indolone ring. This pathway not only eliminates the need for hazardous cyanide salts but also ensures that the reaction proceeds with high specificity, resulting in a crude product that requires minimal purification to reach pharmaceutical-grade standards.
Mechanistic Insights into Oxidative Decarbonylation and Cyclization
The core innovation of this synthesis lies in the mechanistic elegance of the oxidative decarbonylation and subsequent cyclization steps, which dictate the overall efficiency and purity of the final API intermediate. The process begins with the formation of a 3-(2-nitro-4-methoxycarbonyl phenyl)-2-oxo-propionic ether compound via base-catalyzed condensation. Unlike direct alkylation, this condensation occurs at the benzylic position activated by the nitro group, ensuring that substitution happens exclusively at the desired site. Following hydrolysis, the resulting alpha-keto acid undergoes oxidative decarbonylation using hydrogen peroxide. This transformation is critical as it removes the extra carbonyl carbon introduced by the oxalate, converting the side chain into a simple acetic acid derivative while maintaining the integrity of the nitro and ester functional groups. This step effectively bypasses the regioselectivity problems inherent in electrophilic aromatic substitution.
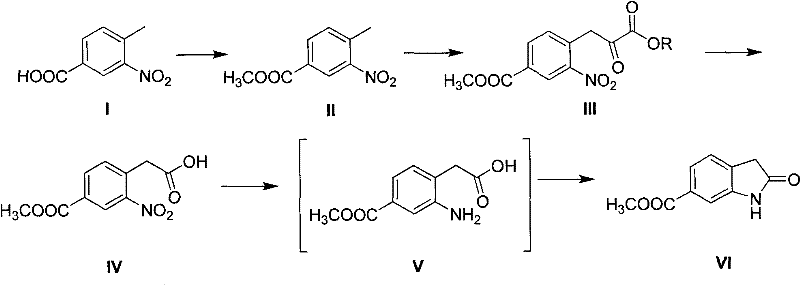
Following the formation of the nitro-acetic acid intermediate, the mechanism shifts to a reductive cyclization. Under hydrogenation conditions using catalysts such as Pd/C or Raney Nickel, the nitro group is selectively reduced to an amine. This newly formed amino group is positioned ortho to the acetic acid side chain, creating an ideal geometry for spontaneous or acid-catalyzed intramolecular amidation. The nucleophilic attack of the amine on the carboxylic acid carbonyl leads to the closure of the five-membered lactam ring, forming the indolone core. Because the precursor is structurally pre-organized by the previous steps, this cyclization proceeds with high fidelity, avoiding the formation of polymeric by-products or alternative ring structures. This mechanistic control is what allows the process to achieve purities exceeding 99% directly after recrystallization, a feat difficult to accomplish with less controlled synthetic routes.
How to Synthesize 6-Methoxycarbonyl Indolone Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and safety, particularly during the exothermic condensation and oxidation phases. The process is designed to be operationally simple, utilizing common solvents like methanol and water, which facilitates easy solvent recovery and waste management. The initial esterification is conducted under mild acidic conditions, followed by a low-temperature condensation to prevent side reactions. The oxidative step utilizes hydrogen peroxide, a green oxidant that produces water as the only by-product, further enhancing the environmental profile of the manufacture. For detailed operational parameters, including specific temperature ramps, stoichiometric ratios, and workup procedures, please refer to the standardized protocol outlined below.
- Esterify 3-nitro-4-methyl benzoic acid with methanol using an acid catalyst to form 3-nitro-4-methyl methyl benzoate.
- React the ester with dialkyl oxalate under alkaline conditions to generate the 2-oxo-propionic ether intermediate.
- Perform hydrolysis and oxidative decarbonylation using hydrogen peroxide to obtain 2-nitro-4-methoxycarbonyl phenylacetic acid.
- Reduce the nitro group to an amino group using hydrogen and Pd/C, followed by acid-catalyzed cyclization to yield the final indolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative advantages in terms of cost structure and supply reliability. By eliminating the use of highly regulated and toxic reagents like sodium cyanide, manufacturers can significantly reduce the costs associated with hazardous waste disposal, specialized containment equipment, and regulatory compliance reporting. This reduction in operational overhead translates directly into a more competitive pricing structure for the final intermediate. Furthermore, the high selectivity of the reaction means that raw material consumption is optimized, as less starting material is lost to the formation of inseparable isomers. This efficiency ensures a more stable cost of goods sold (COGS), protecting margins even when raw material prices fluctuate.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps required to remove isomers drastically lowers processing costs. Traditional methods often require multiple chromatographic separations or extensive recrystallizations to meet purity specs, which consume significant amounts of solvent and energy. By achieving high crude purity through selective chemistry, this new route minimizes downstream processing requirements. Additionally, the use of commodity chemicals like hydrogen peroxide and methanol instead of specialized alkylating agents reduces the direct material cost, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 3-nitro-4-methyl benzoic acid ensures a robust and resilient supply chain. Unlike routes dependent on custom-synthesized or hazardous precursors that may face shipping restrictions or supply interruptions, the inputs for this process are commercially abundant. This accessibility reduces the lead time for high-purity pharmaceutical intermediates and mitigates the risk of production stoppages due to raw material shortages. Manufacturers can maintain consistent inventory levels and meet tight delivery schedules, which is critical for supporting clinical trial timelines and commercial launches.
- Scalability and Environmental Compliance: The mild reaction conditions, operating generally between -20°C and 60°C, make this process highly scalable from pilot plants to multi-ton commercial reactors. The absence of extreme pressures or temperatures reduces the engineering complexity and capital expenditure required for plant infrastructure. Moreover, the generation of benign by-products like water and the avoidance of heavy metal contamination or cyanide waste align perfectly with modern environmental, social, and governance (ESG) goals. This compliance facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain, a key consideration for top-tier pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-methoxycarbonyl indolone. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear perspective on why this method is superior for industrial applications. Understanding these details helps stakeholders make informed decisions about sourcing and process integration.
Q: What are the primary safety advantages of this new synthesis route compared to conventional methods?
A: Unlike conventional routes that utilize highly toxic sodium cyanide or suffer from poor selectivity leading to difficult-to-remove isomers, this patented method avoids hazardous cyanide reagents entirely. It utilizes readily available starting materials and mild reaction conditions, significantly reducing operator risk and environmental hazards associated with toxic waste disposal.
Q: How does the oxidative decarbonylation step impact the overall yield and purity?
A: The oxidative decarbonylation step is critical for removing the extra carbon introduced during the Claisen condensation without generating complex by-products. This specific transformation allows for a streamlined pathway that achieves a total yield of over 70% with a final product purity exceeding 99%, eliminating the need for extensive and costly purification processes typically required for isomer removal.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrialization. It features mild reaction temperatures ranging from -20°C to 60°C and uses common solvents like methanol and water. The avoidance of exotic reagents and the high selectivity of the cyclization step make it robust for scaling from pilot batches to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methoxycarbonyl Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our technical team has extensively evaluated the synthetic pathway described in CN102267934A and possesses the expertise to implement this advanced chemistry at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6-methoxycarbonyl indolone meets the exacting standards required for Phase III clinical trials and commercial manufacturing.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. By leveraging our technical capabilities, you can achieve significant efficiencies and secure a stable source of material. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your project goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
