Revolutionizing Fluorination Chemistry: Scalable Production of Recyclable Fluoroamines for Global Supply Chains
The landscape of organofluorine chemistry is undergoing a significant transformation driven by the urgent need for environmentally sustainable and industrially scalable processes. Patent CN101454272A introduces a groundbreaking class of fluoroamines bearing perfluoroalkyl groups, specifically designed to address the critical challenges associated with traditional fluorination reagents. These novel compounds, represented by general formula (I), function not merely as fluorinating agents but as integral components of a green chemistry cycle that prioritizes waste reduction and resource efficiency. By incorporating a fluorous tag into the molecular structure, this technology enables a unique separation mechanism that drastically simplifies the isolation of fluorinated products from reaction byproducts. For R&D directors and process chemists, this represents a paradigm shift away from hazardous halogenating agents towards a system where the reagent itself can be recovered and regenerated, thereby aligning synthetic efficiency with rigorous environmental compliance standards required by modern regulatory bodies.
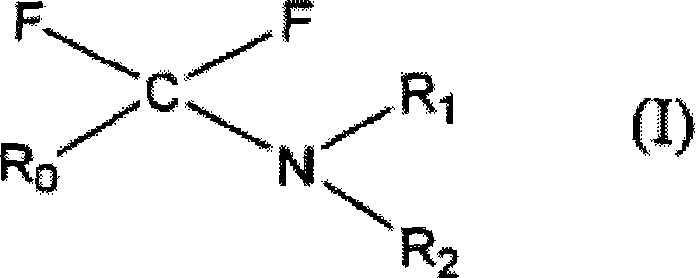
The implementation of this technology offers substantial strategic advantages for procurement and supply chain management within the fine chemical sector. Traditional fluorination methods often rely on reagents that are difficult to handle, highly toxic, or generate substantial amounts of hazardous waste that require costly disposal procedures. In contrast, the fluoroamines described in this patent exhibit high thermal stability and can be distilled, which facilitates their handling even at elevated temperatures exceeding 150°C. This robustness translates directly into operational reliability, reducing the risk of batch failures due to reagent decomposition. Furthermore, the ability to recover the amide precursor means that the effective cost of the fluorinating agent is amortized over multiple cycles, providing a compelling economic argument for adoption in large-scale manufacturing environments where margin compression is a constant concern for procurement managers seeking cost reduction in fluorination manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of fluorine atoms into organic molecules has been fraught with significant technical and environmental hurdles that hinder efficient commercial scale-up of complex fluorinated intermediates. Conventional halogenating agents such as sulfur tetrafluoride (SF4), diethylaminosulfur trifluoride (DAST), or various phosphorus-based halides are notorious for their high toxicity, moisture sensitivity, and the generation of corrosive byproducts like hydrogen fluoride. These characteristics necessitate specialized equipment, stringent safety protocols, and complex waste treatment systems, all of which inflate the capital expenditure and operational costs of a production facility. Moreover, the separation of the desired fluorinated product from the reaction mixture is often challenging, requiring extensive chromatographic purification or energy-intensive distillation steps that lower overall yield and throughput. The environmental burden associated with disposing of halogenated waste streams has also become a major liability, forcing companies to seek alternatives that mitigate ecological impact while maintaining synthetic efficacy.
The Novel Approach
The novel approach detailed in the patent leverages the principles of fluorous chemistry to overcome these entrenched limitations by designing a reagent system that is inherently separable. By attaching a perfluoroalkyl group (Rf) to the nitrogen backbone of the amine, the resulting fluoroamine acquires distinct solubility properties known as fluorophilicity. This means that after the fluorination reaction is complete, the spent reagent (which reverts to an amide form) can be selectively extracted into a fluorous solvent phase, leaving the organic fluorinated product in a separate organic phase. This biphasic separation eliminates the need for complex purification techniques and allows for the direct recovery of the amide precursor. The recovered amide can then be recycled back into the fluoroamine, creating a closed-loop process that significantly reduces raw material consumption. This methodology not only enhances the purity of the final product but also streamlines the workflow, making it an ideal solution for reliable fluoroamine supplier networks aiming to deliver high-value intermediates with minimal environmental footprint.
Mechanistic Insights into Fluorous-Tagged Deoxofluorination
The core mechanistic advantage of this technology lies in the reversible transformation between the fluoroamine and its corresponding amide, driven by the unique electronic and steric properties of the perfluoroalkyl substituent. The synthesis begins with the formation of the amide precursor via a palladium-catalyzed Heck reaction, which couples a halogenated aromatic amide with a perfluoroalkene. This step installs the crucial fluorous tag that dictates the subsequent separation behavior. Following hydrogenation to saturate the alkene linkage, the amide undergoes deoxofluorination using activating agents like oxalyl chloride followed by a fluoride source. The resulting difluoroamine is a potent electrophilic fluorinating agent capable of converting hydroxyl groups into fluorine atoms with high fidelity. The presence of the electron-withdrawing perfluoroalkyl chain stabilizes the nitrogen center while simultaneously rendering the molecule compatible with fluorous biphasic systems. This dual functionality ensures that the reagent is reactive enough for efficient fluorination yet stable enough to withstand the rigors of industrial processing and recovery cycles without degradation.
Impurity control is inherently built into this system through the physical property differences between the fluorous and organic phases. During the workup procedure, the addition of a fluorous solvent and an immiscible organic solvent creates a distinct interface where the fluorous-tagged amide partitions exclusively into the fluorous layer. This physical separation effectively removes the bulk of the reagent-derived impurities from the product stream before any crystallization or distillation occurs. Consequently, the fluorinated product obtained in the organic phase exhibits high purity levels, reducing the need for downstream polishing steps that often contribute to yield loss. For quality assurance teams, this mechanism provides a robust method for controlling the impurity profile, ensuring that heavy metal residues from the catalyst or side products from the fluorination are minimized. The ability to tune the length of the perfluoroalkyl chain (C4 to C15) further allows chemists to optimize the partition coefficient for specific solvent systems, offering a customizable approach to process optimization that is rarely available with conventional small-molecule reagents.
How to Synthesize N,N-Diethyl-alpha,alpha-difluoro-benzylamine Efficiently
The synthesis of these advanced fluoroamines follows a logical sequence of organometallic coupling and functional group transformation that is well-suited for kilogram-to-tonne scale production. The process initiates with the construction of the fluorous-tagged carbon skeleton, followed by the activation of the amide carbonyl to install the fluorine atoms. Detailed procedural parameters, including specific molar ratios, temperature gradients, and solvent choices, are critical for maximizing yield and ensuring the integrity of the perfluoroalkyl chain. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process engineers looking to implement this technology.
- Perform a Palladium-catalyzed Heck reaction between a halogenated N,N-disubstituted amide and a perfluoroalkene to attach the fluorous tag.
- Conduct catalytic hydrogenation using Pd/C to reduce the internal alkene bond, stabilizing the perfluoroalkyl chain.
- Execute deoxofluorination using oxalyl chloride followed by treatment with a fluoride source like triethylamine trihydrofluoride to generate the active fluoroamine.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement strategists, the adoption of this fluoroamine technology presents a compelling value proposition centered on sustainability and long-term cost stability. Traditional fluorination supply chains are often vulnerable to fluctuations in the availability of hazardous reagents and the escalating costs of waste disposal compliance. By shifting to a recyclable reagent system, manufacturers can decouple their production costs from the volatile market prices of single-use fluorinating agents. The ability to recover and reuse the amide precursor means that the effective consumption of raw materials is drastically reduced, leading to substantial cost savings over the lifecycle of the product. Furthermore, the reduced environmental burden simplifies regulatory reporting and lowers the risk of shutdowns due to non-compliance, ensuring a more resilient and continuous supply of critical fluorinated intermediates for downstream pharmaceutical and agrochemical applications.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it transforms a consumable reagent into a recyclable catalyst-like species. By recovering the amide byproduct, the facility avoids the recurring expense of purchasing fresh fluorinating agents for every batch. This circular economy approach significantly lowers the variable cost per kilogram of the final product. Additionally, the simplified workup procedure reduces the consumption of solvents and energy associated with extensive purification, further driving down operational expenditures. The elimination of hazardous waste disposal fees associated with traditional halogenating agents adds another layer of financial benefit, making the overall process economically attractive for high-volume production.
- Enhanced Supply Chain Reliability: Reliability in the supply of fluorinated building blocks is paramount for pharmaceutical companies managing tight development timelines. This technology enhances reliability by reducing dependence on external suppliers of toxic reagents that may face shipping restrictions or production bottlenecks. Since the key amide precursor can be synthesized in-house and recycled, the supply chain becomes more self-sufficient and less susceptible to external disruptions. The thermal stability of the fluoroamine also allows for safer storage and transportation, reducing the logistical complexities and insurance costs associated with handling dangerous goods. This stability ensures that production schedules can be maintained consistently, meeting the demanding delivery windows of global clients.
- Scalability and Environmental Compliance: Scaling fluorination reactions is notoriously difficult due to heat management and safety concerns, but the distillable nature of these fluoroamines mitigates many of these risks. The process is designed to be scalable from laboratory benchtop to multi-tonne commercial reactors without significant re-engineering. From an environmental perspective, the fluorous biphasic system minimizes the release of fluorine-containing waste into the environment, aligning with increasingly strict global environmental regulations. This compliance advantage future-proofs the manufacturing asset, ensuring that the facility remains operational and competitive even as regulatory standards tighten. The green chemistry credentials of this process also enhance the brand reputation of the manufacturer, appealing to end-users who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of fluorous-tagged fluorination technologies. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of adopting this innovative chemical process. Understanding these details is essential for technical teams evaluating the feasibility of integrating this methodology into existing production lines.
Q: How does the fluorous tag facilitate product separation?
A: The perfluoroalkyl group imparts high fluorophilicity to the amide byproduct, making it soluble in fluorous solvents but insoluble in standard organic solvents, allowing for easy phase separation and recovery.
Q: Is this fluorination method safer than traditional reagents?
A: Yes, unlike toxic reagents like SF4 or DAST, these fluoroamines are thermally stable, distillable, and generate less hazardous waste, significantly improving industrial safety profiles.
Q: Can the amide byproduct be reused?
A: Absolutely. The recovered amide serves as the direct starting material for regenerating the fluoroamine, creating a closed-loop system that reduces raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced fluorination technologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101454272A can be realized in a practical industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluoroamine or fluorinated intermediate meets the exacting standards required by the pharmaceutical and agrochemical industries. Our commitment to quality extends beyond mere compliance; we actively collaborate with clients to optimize reaction conditions and maximize the efficiency of the fluorous recycling loop.
We invite you to engage with our technical procurement team to discuss how this recyclable fluoroamine technology can be tailored to your specific synthetic needs. Whether you require a Customized Cost-Saving Analysis for your current fluorination processes or need specific COA data to validate the purity of our intermediates, we are prepared to provide comprehensive route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective but also aligned with the future of sustainable chemical manufacturing. Let us help you engineer a bottleneck-free pathway to your target fluorinated molecules.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
