Advanced One-Pot Synthesis of Diphenhydramine Citrate for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust methodologies to enhance the efficiency of active pharmaceutical ingredient (API) production, and patent CN102229537B presents a transformative approach to the synthesis of Diphenhydramine Citrate. This specific intellectual property details a novel 'one-pot' synthetic strategy that fundamentally alters the traditional manufacturing landscape by integrating multiple reaction stages into a single reactor vessel. By reacting dibenzyl alcohol-β-chloroethyl ether directly with dimethylamine under controlled high-pressure conditions, followed by in-situ neutralization and salt formation, the process circumvents the cumbersome isolation of intermediate species. This technological leap is particularly relevant for a reliable pharmaceutical intermediate supplier aiming to optimize throughput while maintaining stringent quality standards required for antihistamine medications. The elimination of intermediate purification steps not only accelerates the timeline from raw materials to final product but also inherently reduces the risk of contamination and mechanical loss, addressing critical pain points for both R&D directors and supply chain managers focused on operational excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Diphenhydramine Citrate has been plagued by inefficient multi-step protocols that rely heavily on the isolation of unstable or difficult-to-handle intermediates. Prior art, such as U.S. Patents US4401665 and US4505862, describes methods starting from diphenhydramine hydrochloride, which itself is an expensive starting material that necessitates prior synthesis and purification, thereby inflating the overall cost of goods. Furthermore, older methodologies like those disclosed in U.S. Patent US2421714 involve complex sequences including ether extractions, acid-base toggling, and rigorous vacuum distillation at high temperatures (150~165°C/2mm), which are energy-intensive and pose significant safety risks due to the handling of volatile organic solvents and high thermal loads. These conventional routes often suffer from low overall yields due to cumulative losses at each isolation stage, and the extensive use of solvents creates a substantial burden on waste management systems, making them less attractive for modern, environmentally conscious cost reduction in API manufacturing initiatives.
The Novel Approach
In stark contrast, the methodology outlined in CN102229537B introduces a streamlined 'one-pot' technique that consolidates the amination, liberation, and salt formation steps into a continuous sequence within the same reactor. This approach begins with the direct reaction of the chloro-ether precursor with aqueous dimethylamine, generating the hydrochloride salt in situ, which is immediately treated with sodium hydroxide to release the free base without ever leaving the reaction vessel. The subsequent addition of citric acid allows for the direct precipitation of the final citrate salt, effectively bypassing the need for solvent extraction, drying, and redissolution of intermediates. This integration drastically simplifies the operational workflow, reduces the requirement for extensive equipment footprint, and minimizes the exposure of reactive intermediates to atmospheric conditions, thereby enhancing the stability and purity profile of the final high-purity antihistamine intermediates. The ability to recover unreacted dimethylamine gas during the cooling phases further underscores the economic and environmental superiority of this closed-loop system.
Mechanistic Insights into Nucleophilic Substitution and Salt Formation
The core chemical transformation driving this synthesis is a nucleophilic substitution reaction where the lone pair electrons of the dimethylamine nitrogen attack the electrophilic carbon adjacent to the chlorine atom in the dibenzyl alcohol-β-chloroethyl ether. This SN2-type mechanism is facilitated by the elevated temperature (110~150°C) and pressure (0.5~2.5MPa), which provide the necessary activation energy to overcome the steric hindrance presented by the bulky diphenylmethyl group. Following the formation of the quaternary ammonium chloride intermediate, the addition of sodium hydroxide serves a dual purpose: it neutralizes the hydrochloride salt to liberate the lipophilic free base of diphenhydramine and simultaneously reduces the solubility of residual dimethylamine in the aqueous phase, facilitating its recovery. The final step involves a straightforward acid-base reaction where the tertiary amine functionality of the free base accepts a proton from citric acid, forming a stable crystalline citrate salt that precipitates out of the solution, driven by the low solubility of the ion-pair complex in the reaction medium.
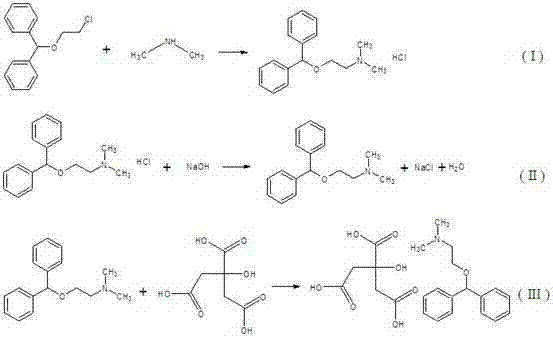
Impurity control in this process is inherently managed by the avoidance of intermediate isolation, which is often a source of degradation and foreign particulate introduction. By maintaining the reaction mixture in a controlled environment throughout the transition from chloro-ether to final citrate salt, the formation of side products such as elimination byproducts (vinyl ethers) or oxidation products is minimized. The use of aqueous dimethylamine and subsequent washing steps with inorganic salt solutions and hot water during the precursor synthesis ensures that acidic residues and unreacted alcohols are effectively removed before the main amination step. This rigorous control over the reaction environment ensures that the final product meets the stringent spectral requirements (UV, IR, NMR, MS) consistent with pharmacopoeial standards, providing R&D teams with confidence in the commercial scale-up of complex pharmaceutical salts without compromising on chemical integrity or safety profiles.
How to Synthesize Diphenhydramine Citrate Efficiently
The implementation of this synthesis route requires precise control over thermodynamic parameters and stoichiometric ratios to maximize yield and minimize byproduct formation. The process initiates with the preparation of the key precursor, dibenzyl alcohol-β-chloroethyl ether, via an etherification reaction between benzhydrol and 2-chloroethanol catalyzed by concentrated sulfuric acid, followed by the main one-pot sequence involving high-pressure amination and salt formation. Detailed operational parameters, including specific temperature ramps, pressure maintenance strategies, and workup procedures, are critical for reproducibility and are essential reading for process engineers looking to adopt this technology. For a comprehensive breakdown of the standardized operating procedures and critical process parameters (CPPs) required to execute this synthesis successfully, please refer to the technical guide below.
- React dibenzyl alcohol-β-chloroethyl ether with excess aqueous dimethylamine at 110-150°C and 0.5-2.5 MPa to form diphenhydramine hydrochloride.
- Cool the reaction mixture, add NaOH solution to liberate the free base diphenhydramine, and recover unreacted amine gas.
- Directly add citric acid solution to the reactor at room temperature to precipitate pure diphenhydramine citrate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this one-pot synthesis methodology offers profound strategic advantages that extend beyond simple chemical efficiency. By eliminating the need to purchase or synthesize isolated diphenhydramine hydrochloride as a starting material, manufacturers can significantly reduce their raw material inventory costs and simplify their supplier base. The consolidation of multiple reaction steps into a single vessel reduces the total batch cycle time, allowing for increased production capacity within existing infrastructure and enabling faster response times to market demand fluctuations. Furthermore, the reduction in solvent usage and the elimination of multiple filtration and drying steps translate directly into lower utility consumption and waste disposal costs, aligning with global sustainability goals and regulatory pressures for greener manufacturing practices.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of unit operations associated with intermediate isolation. Traditional methods require separate reactors, filtration equipment, and drying ovens for the hydrochloride salt and the free base, each adding capital expenditure and operational labor costs. By performing the reaction sequence in one pot, the process drastically cuts down on energy consumption for heating and cooling cycles, as well as the manpower required for material transfer and equipment cleaning. Additionally, the recovery of unreacted dimethylamine gas allows for the recycling of this reagent, further lowering the variable cost per kilogram of the final product and enhancing the overall economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as benzhydrol, 2-chloroethanol, and aqueous dimethylamine ensures a robust and resilient supply chain that is less susceptible to bottlenecks compared to routes requiring specialized or imported intermediates. The simplified process flow reduces the number of potential failure points in the manufacturing line, thereby increasing the reliability of delivery schedules and ensuring consistent availability of the final API intermediate. This stability is crucial for downstream pharmaceutical formulators who depend on a steady supply of high-quality excipients and active ingredients to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard high-pressure reactor technology that is widely available in the fine chemical industry, facilitating a smooth transition from pilot plant to full commercial production. The absence of toxic organic solvents in the precursor synthesis and the minimized solvent load in the final step significantly reduce the environmental footprint of the manufacturing process, easing the burden on effluent treatment plants and ensuring compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible producer of high-purity active ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction conditions, safety considerations, and quality outcomes. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of the one-pot synthesis method for Diphenhydramine Citrate?
A: The one-pot method eliminates the need for isolating and purifying intermediate diphenhydramine hydrochloride and free base, significantly simplifying operation steps, reducing material loss, and shortening the production cycle compared to conventional multi-step processes.
Q: How does this process improve safety and environmental compliance?
A: The synthesis of the key precursor, dibenzyl alcohol-β-chloroethyl ether, utilizes concentrated sulfuric acid without toxic organic solvents, enhancing personnel safety. Furthermore, the integrated process reduces solvent usage and waste generation associated with multiple isolation steps.
Q: What are the critical reaction conditions for the amination step?
A: The amination reaction requires elevated temperatures between 110°C and 150°C and pressures ranging from 0.5 MPa to 2.5 MPa, with a molar ratio of ether to dimethylamine between 1:3 and 1:8 to ensure high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenhydramine Citrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of pharmaceutical intermediates. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one-pot synthesis of Diphenhydramine Citrate can be seamlessly transferred from the laboratory to the factory floor. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized process can drive value for your organization. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable and cost-effective supply of this vital pharmaceutical intermediate.
