Advanced Palladium-Catalyzed Synthesis of 3-Indolyl Rhamnosides for Commercial API Manufacturing
Introduction to Next-Generation C-Glycoside Technology
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more stable and bioactive molecular scaffolds. Patent CN110684018A introduces a significant breakthrough in the synthesis of 3-indolyl rhamnosides, a class of C-glycosides known for their superior enzymatic stability compared to their O-glycoside counterparts. These compounds are not merely academic curiosities; they hold immense potential as critical intermediates in the development of SGLT2 inhibitors for type 2 diabetes treatment, mirroring the efficacy seen in related structures like 3-indolyl glycoside B. The core innovation lies in the structural versatility offered by Formula I, where diverse substituents can be introduced to fine-tune biological activity. 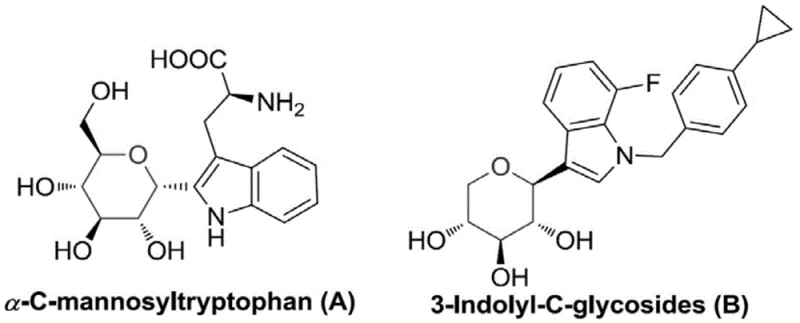 This patent provides a robust foundation for producing high-purity pharmaceutical intermediates, addressing the growing demand for reliable suppliers who can deliver complex sugar-amino acid hybrids with consistent quality.
This patent provides a robust foundation for producing high-purity pharmaceutical intermediates, addressing the growing demand for reliable suppliers who can deliver complex sugar-amino acid hybrids with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C-indole glycosides has been plagued by significant technical hurdles that hinder commercial viability. Traditional routes often involve multi-step sequences with harsh reaction conditions that can degrade sensitive carbohydrate moieties, leading to poor overall yields and difficult purification processes. Furthermore, existing methodologies have predominantly focused on the synthesis of 2-indolyl glycosides, leaving a substantial gap in efficient access to the 3-substituted isomers which are crucial for specific biological targets. The lack of regioselectivity in older methods often results in complex mixtures of isomers, requiring extensive chromatographic separation that drives up costs and extends lead times. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable supply chains, making the search for a more direct synthetic route a top priority for cost reduction in API manufacturing.
The Novel Approach
The methodology disclosed in CN110684018A represents a paradigm shift by utilizing a palladium-catalyzed cyclization of o-alkynylanilines with 1-iodorhamnosene. This approach elegantly constructs the indole ring directly onto the sugar scaffold in a single pot, dramatically improving step economy. 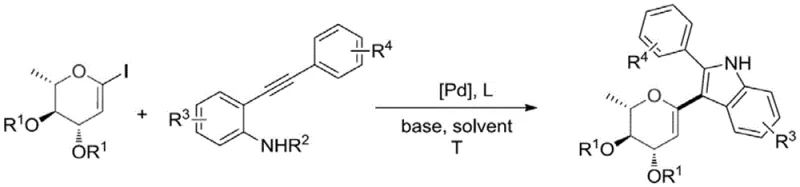 By leveraging transition metal catalysis, the process operates under relatively mild conditions, typically around 100°C in tetrahydrofuran, avoiding the extreme temperatures or strong acids that often compromise product integrity. The substrate universality is exceptional, accommodating various electronic and steric environments on both the aniline and the sugar components. This flexibility allows for the rapid generation of diverse libraries of 3-indolyl rhamnosides, enabling R&D teams to explore structure-activity relationships more efficiently while providing supply chain heads with a scalable, robust process for commercial production.
By leveraging transition metal catalysis, the process operates under relatively mild conditions, typically around 100°C in tetrahydrofuran, avoiding the extreme temperatures or strong acids that often compromise product integrity. The substrate universality is exceptional, accommodating various electronic and steric environments on both the aniline and the sugar components. This flexibility allows for the rapid generation of diverse libraries of 3-indolyl rhamnosides, enabling R&D teams to explore structure-activity relationships more efficiently while providing supply chain heads with a scalable, robust process for commercial production.
Mechanistic Insights into Palladium-Catalyzed Cyclization
At the heart of this synthesis is a sophisticated catalytic cycle involving palladium species that ensures high regioselectivity for the 3-position of the indole ring. The reaction initiates with the oxidative addition of the palladium(0) catalyst to the carbon-iodine bond of the 1-iodorhamnosene, forming a reactive organopalladium intermediate. This species then undergoes coordination and insertion with the alkyne moiety of the o-alkynylaniline, followed by an intramolecular nucleophilic attack by the nitrogen atom to close the indole ring. The presence of specific phosphine ligands, such as tris(4-methoxyphenyl)phosphine, plays a critical role in stabilizing the active catalytic species and facilitating the reductive elimination step that releases the final product. Understanding this mechanism is vital for R&D directors, as it explains the high purity profiles observed; the directed nature of the cyclization minimizes the formation of regioisomeric byproducts that are common in non-catalyzed thermal cyclizations.
Impurity control is further enhanced by the choice of base and solvent system, which suppresses side reactions such as homocoupling of the alkyne or dehalogenation of the sugar iodide. The use of potassium carbonate in THF creates an optimal environment for the transmetallation and cyclization steps without promoting hydrolysis of the sensitive glycosidic bond. 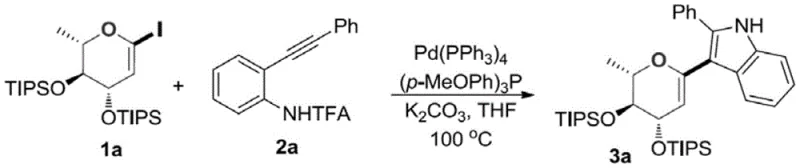 As demonstrated in Example 1, where compound 3a is synthesized with an impressive 81% yield, the process delivers a clean product profile that simplifies downstream processing. The ability to tune the electronic properties of the ligands and the palladium source allows for fine-tuning the reaction kinetics, ensuring that even sterically hindered substrates can be converted efficiently. This level of mechanistic control translates directly into a more predictable and manageable manufacturing process, reducing the risk of batch failures and ensuring consistent quality for high-purity pharmaceutical intermediates.
As demonstrated in Example 1, where compound 3a is synthesized with an impressive 81% yield, the process delivers a clean product profile that simplifies downstream processing. The ability to tune the electronic properties of the ligands and the palladium source allows for fine-tuning the reaction kinetics, ensuring that even sterically hindered substrates can be converted efficiently. This level of mechanistic control translates directly into a more predictable and manageable manufacturing process, reducing the risk of batch failures and ensuring consistent quality for high-purity pharmaceutical intermediates.
How to Synthesize 3-Indolyl Rhamnoside Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters defined in the patent to maximize yield and safety. The process begins with the precise weighing of 1-iodorhamnosene and the specific o-alkynylaniline derivative, maintaining a molar ratio between 1:1 and 1.5:1 to ensure complete conversion of the limiting reagent. The reaction is conducted under an inert atmosphere, preferably nitrogen, to prevent oxidation of the palladium catalyst and the sensitive organic intermediates. Detailed standardized operating procedures regarding temperature ramping, stirring rates, and quenching protocols are essential for reproducibility. For a comprehensive guide on the exact stoichiometry and workup procedures validated by experimental data, please refer to the technical steps outlined below.
- Mix 1-iodorhamnosene, o-alkynylaniline, Pd catalyst (e.g., Pd(PPh3)4), phosphine ligand, base (K2CO3), and solvent (THF) in a reaction vessel.
- Heat the mixture to 90-110°C under nitrogen protection and stir for 4-15 hours until TLC indicates complete consumption of the starting aniline.
- Filter the reaction mixture, concentrate under reduced pressure, and purify the crude product via column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line and operational reliability of pharmaceutical manufacturing. The shift towards a catalytic cyclization strategy eliminates the need for multiple protection and deprotection steps often required in classical glycosylation, significantly streamlining the production timeline. This reduction in synthetic complexity means fewer unit operations, less solvent consumption, and lower energy requirements per kilogram of product, all of which contribute to a more sustainable and cost-effective manufacturing footprint. For procurement managers, this translates into a more competitive pricing structure for the final intermediate, allowing for better margin management in the final drug product. The robustness of the reaction conditions also implies a lower risk of supply disruption due to process sensitivity.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the use of catalytic amounts of palladium significantly lowers the raw material cost per batch. Furthermore, the high yields reported (ranging from 75% to 86%) mean that less starting material is wasted, maximizing the value derived from every kilogram of input. The simplified purification process, often achievable through standard column chromatography or crystallization, reduces the labor and equipment time associated with isolation. These factors combine to create a highly efficient process that drives down the overall cost of goods sold (COGS) for complex C-glycoside intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials, including 1-iodorhamnosene and various substituted o-alkynylanilines, are either commercially available or can be synthesized via well-established routes, ensuring a stable supply of inputs. The reaction's tolerance to a wide range of functional groups means that supply chain disruptions for specific niche reagents can be mitigated by switching to alternative analogs without re-optimizing the entire process. This flexibility is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. The scalability of the process from gram to kilogram scale has been demonstrated, providing confidence in the ability to ramp up production volumes as market demand increases.
- Scalability and Environmental Compliance: Operating at moderate temperatures (90-110°C) and using common solvents like THF makes this process easily adaptable to large-scale reactors without requiring specialized high-pressure or cryogenic equipment. The use of inorganic bases like potassium carbonate generates benign salt byproducts that are easier to handle and dispose of compared to the hazardous waste streams generated by stronger bases or Lewis acids. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate sustainability profile of the manufacturing site. The high atom economy of the cyclization reaction further minimizes waste generation, supporting global initiatives for greener pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances helps in making informed decisions about process integration and resource allocation. For more specific technical data or custom feasibility studies, our team is ready to assist with detailed discussions.
Q: What are the key advantages of this Pd-catalyzed method over traditional glycosylation?
A: Unlike traditional methods that often suffer from harsh conditions and poor regioselectivity for 3-substituted indoles, this patent describes a mild, high-yielding cyclization that specifically targets the 3-position with excellent substrate universality.
Q: Is this synthesis scalable for commercial production of diabetes drug intermediates?
A: Yes, the process utilizes standard solvents like THF and common bases like potassium carbonate at moderate temperatures (100°C), making it highly amenable to scale-up from laboratory to multi-ton commercial manufacturing.
Q: What is the typical yield range for these 3-indolyl rhamnoside derivatives?
A: According to the experimental data in patent CN110684018A, the reaction consistently achieves high yields ranging from 75% to 86% across various substituted substrates, ensuring efficient material utilization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Indolyl Rhamnoside Supplier
As the pharmaceutical industry continues to demand more complex and potent therapeutic agents, the ability to synthesize advanced intermediates like 3-indolyl rhamnosides efficiently becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to process excellence, we provide the reliability and quality assurance that global pharma companies require for their critical supply chains.
We invite you to explore how this innovative synthesis route can optimize your project timelines and budget. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for high-purity pharmaceutical intermediates. Let us be your partner in turning complex chemical challenges into commercial successes.
