Advanced Synthesis of Acetate-Bridged Chiral Binuclear Copper Complexes for Next-Gen Optical Storage
The landscape of molecular-based functional materials is undergoing a significant transformation with the emergence of multifunctional compounds that integrate distinct physical properties into a single molecular architecture. Patent CN109180708B introduces a groundbreaking advancement in this field by disclosing a novel acetate-bridged chiral binuclear copper complex, specifically formulated as Cu2(CH3COO)4(L)2. This innovation addresses a critical gap in the development of materials that simultaneously possess chiral optical activity and magnetic properties, a combination that is exceptionally rare and highly sought after for applications in nonlinear optics and information storage. The patent details a robust synthetic methodology that utilizes a chiral monodentate nitrogen-containing organic ligand, identified as [(–)-2-(4′-pyridyl)-4,5-pinene-pyridine], which coordinates with copper acetate under remarkably mild conditions. Unlike traditional methods that often require harsh thermal treatments or complex catalytic systems, this process operates effectively at room temperature, yielding high-purity blue crystals through a straightforward volatilization technique. For R&D directors and procurement specialists in the electronic materials sector, this represents a pivotal opportunity to access high-performance optical-magnetic materials with a simplified supply chain and reduced manufacturing complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral magnetic materials has been plagued by significant technical hurdles that hinder their commercial viability and widespread adoption in high-tech industries. Conventional approaches often rely on rigid multidentate ligands that are difficult to synthesize and prone to racemization during the crystallization process, leading to products with diminished or non-existent chiral optical activity. Furthermore, many existing protocols necessitate elevated temperatures and prolonged reaction times, which not only increase energy consumption but also elevate the risk of thermal decomposition of sensitive chiral centers. The separation and purification of these complexes frequently involve cumbersome chromatographic techniques or recrystallization from hazardous solvent systems, creating bottlenecks in production throughput and escalating operational costs. Additionally, achieving strong magnetic coupling between metal centers typically requires precise control over bridging distances, which is often difficult to maintain in less controlled synthetic environments, resulting in materials with inconsistent magnetic performance. These cumulative challenges have limited the availability of reliable chiral magnetic bifunctional materials for advanced device fabrication.
The Novel Approach
The methodology outlined in patent CN109180708B offers a transformative solution by leveraging a self-assembly strategy driven by acetate bridging groups to construct the binuclear copper core. This novel approach utilizes a specifically designed chiral ligand derived from myrtenal, a naturally occurring terpene, which imparts inherent stability to the chiral configuration throughout the synthesis. By employing copper acetate as both the metal source and the bridging agent, the process ensures a short and rigid bridging distance between the two copper(II) ions, which is critical for enhancing antiferromagnetic coupling interactions. The reaction proceeds spontaneously at ambient temperature in a mixed solvent system of methanol and acetonitrile, eliminating the need for external heating sources and significantly reducing the energy footprint of the manufacturing process. Post-reaction processing is exceptionally efficient, requiring only simple filtration and slow solvent volatilization to obtain high-quality single crystals, thereby streamlining the workflow from reactor to final product. This simplicity not only accelerates the production cycle but also enhances the reproducibility of the material's physical properties, making it a superior candidate for industrial scale-up.
Mechanistic Insights into Acetate-Bridged Coordination and Magnetic Coupling
The structural integrity and functional performance of the Cu2(CH3COO)4(L)2 complex are fundamentally rooted in its unique coordination geometry and the electronic interactions facilitated by the acetate bridges. In this binuclear architecture, each copper(II) ion adopts a square pyramidal or distorted octahedral geometry, coordinated by a nitrogen atom from the chiral pyridine ligand and oxygen atoms from four acetate groups. The acetate ions act as syn-syn bridging ligands, connecting the two copper centers in a paddle-wheel-like arrangement that enforces a close proximity between the metal ions. This short Cu-Cu distance is the mechanistic key to the material's strong antiferromagnetic coupling, as it allows for effective overlap of magnetic orbitals through the bridging oxygen atoms. The chiral ligand L, depicted in the structural diagram below, plays a dual role by not only stabilizing the metal center through coordination but also transmitting chirality to the entire supramolecular assembly, preventing the formation of racemic mixtures.
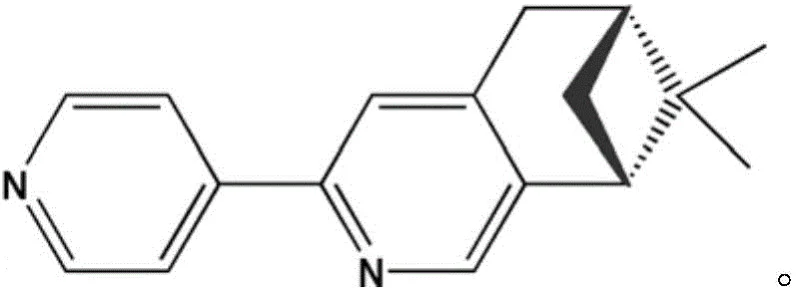
The preservation of chirality is further evidenced by the circular dichroism (CD) spectra, which display distinct Cotton effects at specific wavelengths, confirming the non-centrosymmetric nature of the bulk material. From a mechanistic standpoint, the use of a monodentate ligand rather than a bulky multidentate one reduces steric hindrance during the self-assembly process, allowing the acetate bridges to form more readily and uniformly. This precise control over the primary coordination sphere ensures that the magnetic exchange pathways remain consistent across the crystal lattice, resulting in uniform magnetic susceptibility behavior as observed in variable-temperature measurements. For researchers focusing on spintronics or quantum computing components, understanding this structure-property relationship is vital for tuning the magnetic exchange constants through ligand modification while maintaining the robust acetate-bridged core.
How to Synthesize Acetate-Bridged Chiral Binuclear Copper Complex Efficiently
The practical implementation of this synthesis route is designed for efficiency and ease of operation, making it accessible for both laboratory-scale research and pilot-plant production. The process begins with the preparation of the chiral ligand L via a condensation reaction between a pyridinium salt intermediate and (–)-myrtenal in formamide, followed by the direct coordination step with copper acetate. The simplicity of the protocol minimizes the requirement for specialized equipment or inert atmosphere conditions beyond standard nitrogen protection during the ligand synthesis. Detailed standardized operating procedures regarding stoichiometry, solvent ratios, and crystallization kinetics are essential for maximizing yield and crystal quality. The following guide outlines the critical phases of this synthesis, ensuring that technical teams can replicate the high yields and purity levels reported in the patent documentation.
- Prepare the chiral monodentate ligand L by reacting 4-(pyridylacetyl)pyridinium iodide salt with myrtenal in formamide at 75-85°C.
- Dissolve copper acetate monohydrate in methanol and add it to an acetonitrile solution of the chiral ligand L.
- Stir the mixture at room temperature for 20-25 minutes, filter, and allow slow volatilization to obtain blue crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical performance. The reliance on commercially available starting materials, such as copper acetate and myrtenal derivatives, ensures a stable and resilient supply chain that is less susceptible to the volatility often associated with exotic reagents. The elimination of high-temperature reaction steps translates directly into lower utility costs and reduced wear on reactor vessels, contributing to a more sustainable and cost-effective manufacturing profile. Furthermore, the straightforward isolation method involving filtration and washing avoids the use of expensive chromatography resins or large volumes of extraction solvents, significantly reducing waste disposal costs and environmental compliance burdens. These factors collectively enhance the overall economic feasibility of producing this advanced material, positioning it as a viable option for mass-market electronic applications.
- Cost Reduction in Manufacturing: The ambient temperature reaction conditions eliminate the need for energy-intensive heating systems, leading to significant operational expenditure savings over long production runs. By utilizing copper acetate as a dual-function reagent that provides both the metal center and the bridging ligand, the process reduces the total number of raw materials required, simplifying inventory management and purchasing logistics. The high yield reported in the patent examples indicates minimal material loss during conversion, which maximizes the return on investment for every kilogram of input material. Additionally, the simplified workup procedure reduces labor hours associated with purification, allowing technical staff to focus on value-added activities rather than routine processing tasks.
- Enhanced Supply Chain Reliability: The precursors for this synthesis, including myrtenal and pyridine derivatives, are established commodity chemicals with multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction against minor variations in conditions suggests a high tolerance for scale-up, ensuring consistent output quality even when transitioning from benchtop to industrial reactors. This reliability is crucial for maintaining continuous production schedules in the fast-paced electronics industry, where delays in material delivery can disrupt downstream device assembly lines. The stability of the final crystalline product also facilitates easier storage and transportation, reducing the need for specialized cold-chain logistics.
- Scalability and Environmental Compliance: The use of common organic solvents like methanol and acetonitrile, which are easily recoverable through distillation, aligns with green chemistry principles and facilitates solvent recycling programs. The absence of heavy metal catalysts or toxic reagents simplifies the treatment of process effluents, ensuring compliance with increasingly stringent environmental regulations in major manufacturing hubs. The crystallization-driven purification method is inherently scalable, as slow volatilization can be adapted to larger vessel geometries or replaced by controlled cooling crystallization without compromising product integrity. This scalability ensures that the supply of high-purity optical-magnetic materials can grow in tandem with market demand for advanced data storage and photonic devices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral binuclear copper complex. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the material's capabilities. Understanding these aspects is essential for stakeholders evaluating the integration of this material into their product development pipelines.
Q: What are the key advantages of this acetate-bridged copper complex?
A: The complex exhibits rare bifunctional properties, combining chiral optical activity with strong antiferromagnetic coupling, making it ideal for optical-magnetic devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process operates at room temperature with simple filtration and crystallization steps, eliminating the need for energy-intensive heating or complex purification.
Q: How is chirality maintained during the coordination process?
A: The use of a robust chiral pinene-derived ligand prevents racemization during crystallization, ensuring the final material retains high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Binuclear Copper Complex Supplier
As the demand for multifunctional electronic materials continues to surge, partnering with an experienced chemical manufacturer is essential for securing a stable supply of high-performance compounds. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global technology firms. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the magnetic and optical properties of every batch, guaranteeing that the material meets the exacting standards required for optical-magnetic device fabrication. We understand the critical nature of supply continuity in the electronics sector and have optimized our logistics networks to deliver these specialized intermediates with precision and reliability.
We invite R&D directors and procurement professionals to engage with our technical team to discuss how this acetate-bridged copper complex can enhance your next-generation material portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to support your validation processes and accelerate your time to market.
