Advancing Pharmaceutical Intermediates: Scalable Electrooxidative Synthesis of Fluoroalkyl Dibenzocycloheptanones
Advancing Pharmaceutical Intermediates: Scalable Electrooxidative Synthesis of Fluoroalkyl Dibenzocycloheptanones
The landscape of modern pharmaceutical synthesis is rapidly shifting towards greener, more sustainable methodologies that do not compromise on efficiency or purity. A significant breakthrough in this domain is detailed in Chinese Patent CN115305490A, which discloses a novel method for the electrooxidative synthesis of fluoroalkyl-substituted dibenzocycloheptanone compounds. This technology addresses a critical need in the development of colchicine analogs, a class of compounds renowned for their potent anti-tumor activities yet historically plagued by toxicity issues. By leveraging electrochemical oxidation, this process enables the direct construction of the complex 6-7-6 polycyclic core structure found in bioactive molecules like NSC 51046 and ZD 6126 without the reliance on stoichiometric chemical oxidants.
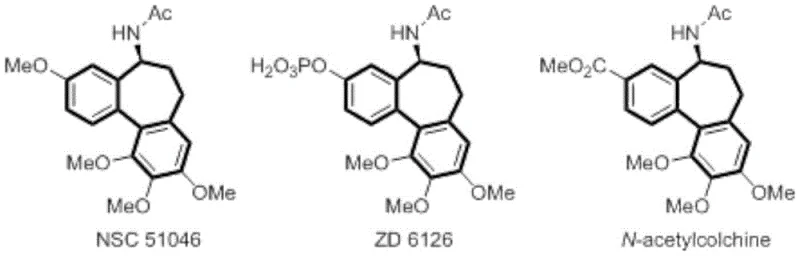
For R&D directors and process chemists, the introduction of fluorine atoms into drug scaffolds is a strategic move to enhance metabolic stability, bioavailability, and binding affinity. The patent highlights that while the dibenzocycloheptanone skeleton has been a target of synthetic interest for decades, efficient methods to introduce fluoroalkyl groups directly during ring construction have remained elusive. This electrochemical approach utilizes readily available o-propargyl propionyl biphenyl compounds and fluoroalkyl sodium sulfinate salts as starting materials. The reaction proceeds under mild constant-current conditions, generating fluoroalkyl radicals in situ that trigger a cascade cyclization. This represents a paradigm shift from traditional multi-step sequences, offering a streamlined pathway to high-purity pharmaceutical intermediates that are essential for next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex polycyclic frameworks like dibenzocycloheptanones often relies on transition-metal catalyzed cross-coupling reactions or the use of harsh stoichiometric oxidants. These conventional routes frequently suffer from significant drawbacks that impact both the economic viability and the environmental footprint of the manufacturing process. For instance, methods employing palladium or copper catalysts necessitate rigorous purification protocols to remove trace metal residues, which is a mandatory requirement for API manufacturing but adds substantial time and cost to the production cycle. Furthermore, the use of strong chemical oxidants can lead to poor atom economy and the generation of hazardous waste streams, complicating waste management and increasing the overall cost of goods sold (COGS).
Another critical limitation of prior art is the difficulty in controlling regioselectivity and functional group tolerance when introducing fluorine-containing moieties. Many traditional fluorination reagents are unstable, expensive, or require extreme reaction conditions such as cryogenic temperatures or high pressures, which pose safety risks and engineering challenges during scale-up. The inability to efficiently construct the seven-membered ring while simultaneously installing the fluoroalkyl group often forces chemists to adopt linear synthetic strategies with low overall yields. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times for clinical trial materials and delaying the commercialization of promising drug candidates.
The Novel Approach
The electrooxidative method described in patent CN115305490A offers a transformative solution by replacing chemical oxidants with electrons, effectively using electricity as a clean reagent. This approach utilizes an undivided cell equipped with a graphite felt (GF) anode and a platinum (Pt) cathode, operating under constant current electrolysis. The core innovation lies in the anodic oxidation of fluoroalkyl sulfinate anions to generate reactive fluoroalkyl radicals, which then attack the alpha-position of the carbonyl group in the alkynone substrate. This initiates a radical cascade that culminates in a 7-endo-trig cycloaddition, seamlessly constructing the seven-membered ring and installing the fluoroalkyl group in a single operational step.
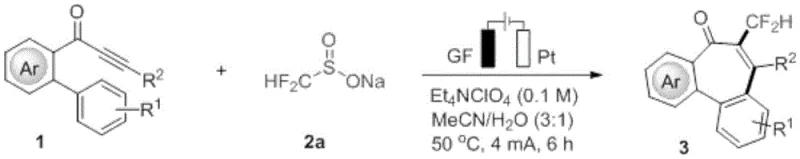
This novel strategy significantly simplifies the synthetic route, reducing the number of unit operations and eliminating the need for external oxidizing agents. The reaction conditions are remarkably mild, typically proceeding at 50°C in a mixture of acetonitrile and water, which enhances safety and reduces energy consumption compared to high-temperature thermal processes. Moreover, the metal-free nature of the electrochemical system ensures that the final product is free from heavy metal contamination, a crucial advantage for regulatory compliance in the pharmaceutical industry. By demonstrating compatibility with various substituents on the aromatic rings, including halogens and alkoxy groups, this method provides a robust and versatile platform for the rapid diversification of the dibenzocycloheptanone library, accelerating the discovery of new therapeutic agents.
Mechanistic Insights into Electrooxidative Radical Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for process optimization and scale-up. The reaction mechanism, as elucidated in the patent, begins with the anodic oxidation of the sodium fluoroalkyl sulfinate (e.g., sodium difluoromethanesulfinate) at the graphite felt electrode. This single-electron transfer process generates a sulfonyl radical intermediate which rapidly extrudes sulfur dioxide (SO2) to yield the highly reactive difluoromethyl radical (·CF2H). This radical species then selectively attacks the electron-deficient alkyne moiety of the o-propargyl biphenyl substrate at the alpha-position relative to the carbonyl group. This addition forms a vinyl radical intermediate, designated as species A in the mechanistic proposal, which is thermodynamically stabilized by the adjacent carbonyl functionality.
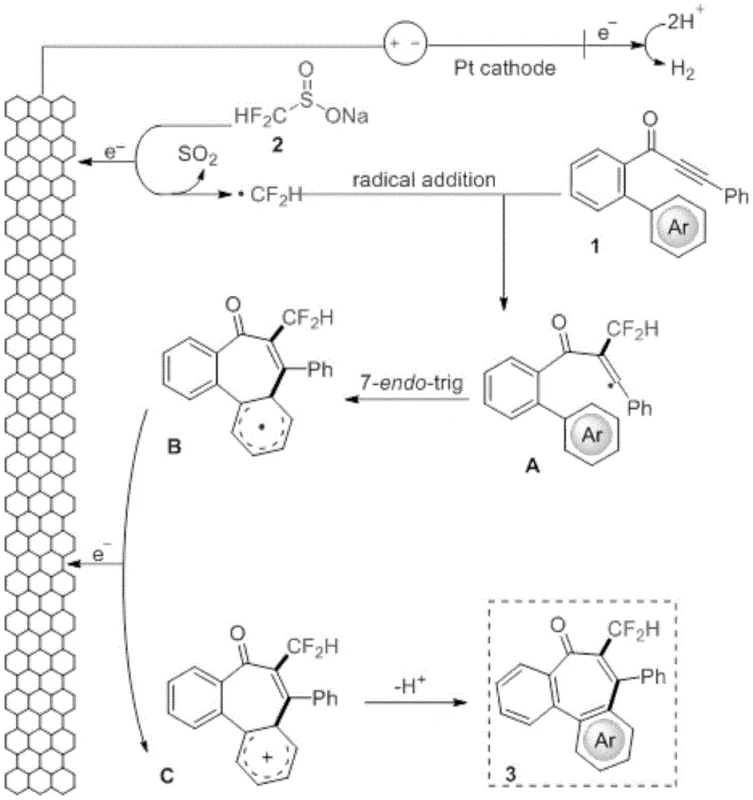
Following the initial radical addition, the vinyl radical undergoes an intramolecular 7-endo-trig cyclization onto the pendant aromatic ring. This step is the key ring-forming event that constructs the central seven-membered ring of the dibenzocycloheptanone scaffold, generating a new cyclohexadienyl radical intermediate (species B). To complete the catalytic cycle and restore aromaticity, this radical intermediate undergoes a second oxidation event at the anode to form a cationic species (species C). Finally, a deprotonation step occurs, likely facilitated by the basicity of the medium or the sulfinate salt, to yield the neutral fluoroalkyl-substituted dibenzocycloheptanone product. This elegant cascade demonstrates precise control over radical reactivity, minimizing side reactions such as polymerization or non-selective hydrogen abstraction, which ensures high selectivity and purity of the final API intermediate.
From an impurity control perspective, the electrochemical nature of the reaction allows for fine-tuning of the oxidation potential by adjusting the current density. This prevents over-oxidation of the sensitive organic substrates, a common issue with chemical oxidants like hypervalent iodine or permanganates. The use of a mixed solvent system of acetonitrile and water (3:1) not only solubilizes both the organic substrate and the inorganic electrolyte but also aids in proton management during the final deprotonation step. The absence of transition metals eliminates the risk of metal-catalyzed decomposition pathways, resulting in a cleaner crude reaction profile that simplifies downstream purification. This mechanistic clarity provides confidence to process engineers that the reaction can be reliably reproduced and scaled with consistent quality attributes.
How to Synthesize Fluoroalkyl Dibenzocycloheptanones Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and reaction parameters to maximize yield and throughput. The patent outlines a standardized procedure where the substrate and reagent are dissolved in the electrolyte solution and subjected to constant current electrolysis. The choice of electrode material is critical; graphite felt serves as a high-surface-area anode that promotes efficient electron transfer, while the platinum cathode facilitates the reduction of protons to hydrogen gas, maintaining charge balance. The reaction is typically conducted at a constant current of 4 mA for a duration of 6 hours at a controlled temperature of 50°C. These parameters have been optimized to balance the rate of radical generation with the rate of the cyclization step, preventing the accumulation of reactive intermediates that could lead to byproduct formation.
- Prepare the electrolytic cell with a graphite felt (GF) anode and a platinum (Pt) cathode, ensuring the system is ready for constant current operation.
- Mix o-propargyl biphenyl compounds and sodium fluoroalkyl sulfinate in a MeCN/H2O (3: 1) solvent system containing 0.1 M tetraethylammonium perchlorate as the electrolyte.
- Apply a constant current of 4 mA at 50°C for 6 hours to facilitate the radical cascade cyclization, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary value driver is the elimination of expensive transition metal catalysts and stoichiometric oxidants, which directly translates to significant cost reduction in pharmaceutical intermediate manufacturing. By removing the need for precious metals like palladium or rhodium, companies can avoid the volatility associated with commodity pricing of these materials. Furthermore, the absence of heavy metals in the reaction mixture obviates the need for specialized scavenging resins or complex extraction protocols designed to meet strict residual metal limits, thereby streamlining the purification workflow and reducing the consumption of silica gel and solvents.
- Cost Reduction in Manufacturing: The operational expenditure (OPEX) is lowered through the use of electricity as the primary oxidant, which is generally cheaper and more consistent in price than chemical reagents. The simplified workup procedure, involving solvent evaporation and standard column chromatography, reduces labor hours and waste disposal costs associated with hazardous chemical byproducts. Additionally, the high atom economy of the radical cascade reaction means that a larger proportion of the starting material mass is incorporated into the final product, minimizing raw material waste and improving the overall process mass intensity (PMI). These factors collectively contribute to a more lean and cost-effective production model suitable for competitive generic or specialty chemical markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium sulfinate salts and simple biphenyl derivatives ensures a stable and diversified supply base, reducing the risk of shortages associated with specialized reagents. The robustness of the electrochemical method, which tolerates a wide range of functional groups including halides and ethers, allows for the use of diverse starting materials without requiring extensive protection-deprotection strategies. This flexibility enables manufacturers to pivot quickly between different analogs based on market demand or clinical trial results. Moreover, the mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned downtime due to maintenance, thus ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry reactors, which offer superior mass and heat transfer compared to batch systems. The inherent safety of operating at ambient pressure and moderate temperatures (50°C) lowers the barrier for scale-up, as it does not require high-pressure rated vessels or extensive cooling infrastructure. From an environmental standpoint, the generation of hydrogen gas as the only byproduct at the cathode aligns with green chemistry principles, significantly reducing the E-factor of the process. This eco-friendly profile supports corporate sustainability goals and simplifies regulatory filings related to environmental impact, making the technology attractive for long-term commercial partnerships focused on responsible manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrooxidative technology. These insights are derived directly from the experimental data and scope defined in patent CN115305490A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production pipelines for high-value pharmaceutical intermediates.
Q: What are the key advantages of this electrochemical method over traditional transition-metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, thereby removing the costly and complex downstream purification steps required to reduce heavy metal residues to ppm levels, which is critical for pharmaceutical compliance.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the use of simple undivided cells, commercially available graphite felt electrodes, and mild reaction conditions (50°C) suggests high scalability potential without the need for specialized high-pressure or cryogenic equipment often found in traditional organic synthesis.
Q: What is the substrate scope regarding the fluoroalkyl group?
A: The patent demonstrates successful incorporation of both difluoromethyl (CF2H) and trifluoromethyl (CF3) groups using their respective sodium sulfinate salts, allowing for versatile modification of the drug candidate's metabolic stability and lipophilicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Dibenzocycloheptanones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electroorganic synthesis in the production of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115305490A can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for global regulatory submissions. We are committed to delivering high-purity fluoroalkyl-substituted dibenzocycloheptanones that serve as critical building blocks for next-generation anti-cancer therapies.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free electrochemical route. We encourage you to contact us to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that will accelerate your drug development timeline. Let us collaborate to bring safer, more effective medicines to patients worldwide through superior chemical manufacturing.
