Scalable Total Synthesis of Rupestine G: A Breakthrough in Guaipyridine Alkaloid Manufacturing
Introduction to Patent CN107129462B and Technological Breakthrough
The pharmaceutical and fine chemical industries are constantly seeking reliable methods to access complex natural product scaffolds that exhibit potent biological activities. Patent CN107129462B represents a significant milestone in this domain by disclosing the first comprehensive total synthesis and enantiomer resolution method for Rupestine G, a novel guaipyridine sesquiterpene alkaloid originally isolated from Artemisia rupestris. Historically, the supply of such complex alkaloids has been severely constrained by their low natural abundance and the ecological variability of plant sources. This patent overcomes these bottlenecks by establishing a convergent synthetic route that begins with the cheap and readily available industrial raw material, 2-methyl-5-bromopyridine. The methodology not only secures a stable supply chain for this high-value intermediate but also provides a versatile platform for synthesizing structural analogs for structure-activity relationship (SAR) studies.
The technical significance of this invention extends beyond mere accessibility; it solves the intricate problem of constructing the fused seven-membered carbocyclic ring onto the pyridine nucleus with precise stereochemical control. By leveraging modern catalytic technologies, including palladium-catalyzed cross-couplings and olefin metathesis, the inventors have created a pathway that is both chemically elegant and commercially viable. For R&D directors and procurement managers in the pharmaceutical sector, this development signals a shift from unpredictable botanical extraction to deterministic, high-purity chemical manufacturing, ensuring consistent quality and batch-to-batch reproducibility essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of guaipyridine sesquiterpene alkaloids like Rupestine G relied heavily on extraction from natural plant sources, a process fraught with inefficiencies and supply chain risks. The concentration of these bioactive compounds in Artemisia rupestris is inherently low, necessitating the processing of massive quantities of biomass to obtain gram-scale amounts, which is neither economically sustainable nor environmentally friendly. Furthermore, natural extraction yields a complex mixture of congeners that are structurally similar, making downstream purification arduous and often resulting in poor overall recovery rates. From a regulatory perspective, relying on botanical sources introduces variability in impurity profiles due to seasonal changes, soil conditions, and harvest times, complicating the validation processes required for pharmaceutical ingredients.
Prior synthetic attempts to access similar guaipyridine skeletons often suffered from lengthy linear sequences, harsh reaction conditions, or the use of prohibitively expensive chiral pool starting materials that limited scalability. Many existing routes struggled to efficiently close the seven-membered ring without forming thermodynamic byproducts or requiring protecting group strategies that added unnecessary steps and reduced atom economy. These conventional limitations resulted in high manufacturing costs and long lead times, rendering such compounds inaccessible for widespread preclinical evaluation or commercial therapeutic application.
The Novel Approach
The novel approach detailed in the patent revolutionizes the synthesis of this class of compounds by employing a modular strategy centered on the functionalization of a simple pyridine derivative. By starting with 2-methyl-5-bromopyridine, the process leverages a commodity chemical that ensures cost stability and supply security. The route ingeniously builds complexity through a Blaise reaction to install the beta-ketoester functionality, followed by a strategic alkylation and Suzuki coupling to introduce the necessary carbon framework for ring closure. This divergent thinking allows for the rapid assembly of the acyclic precursor required for the critical cyclization step.
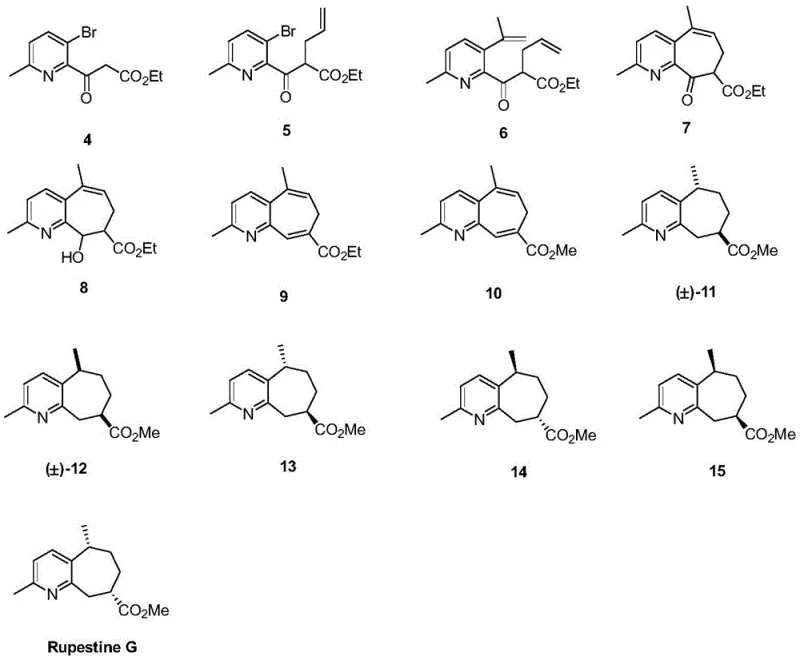
Crucially, the new method utilizes an intramolecular olefin metathesis reaction to forge the seven-membered ring, a transformation that is highly efficient and tolerant of various functional groups. This stands in stark contrast to older cyclization methods that might require high dilution or toxic tin reagents. Following ring closure, the synthesis proceeds through a streamlined sequence of reduction, elimination, and esterification to set the stage for the final stereochemical differentiation. This modern synthetic design drastically reduces the step count compared to traditional approaches and improves the overall yield, making the commercial production of Rupestine G and its isomers a realistic possibility for the first time.
Mechanistic Insights into Ring-Closing Metathesis and Catalytic Hydrogenation
The heart of this synthetic achievement lies in the sophisticated application of transition metal catalysis to construct the core scaffold. The formation of the cyclohepta[b]pyridine system is accomplished via a Ring-Closing Metathesis (RCM) reaction using the second-generation Grubbs catalyst. Mechanistically, this involves the coordination of the ruthenium carbene species to the terminal olefins present in compound 6, followed by a [2+2] cycloaddition to form a metallacyclobutane intermediate. The subsequent retro-[2+2] cycloaddition releases ethylene gas and generates the desired cyclic alkene in compound 7. This step is particularly remarkable because forming medium-sized rings (7-membered) is entropically disfavored; however, the specific positioning of the olefinic handles and the robustness of the Grubbs II catalyst drive the equilibrium toward the cyclic product with high efficiency.
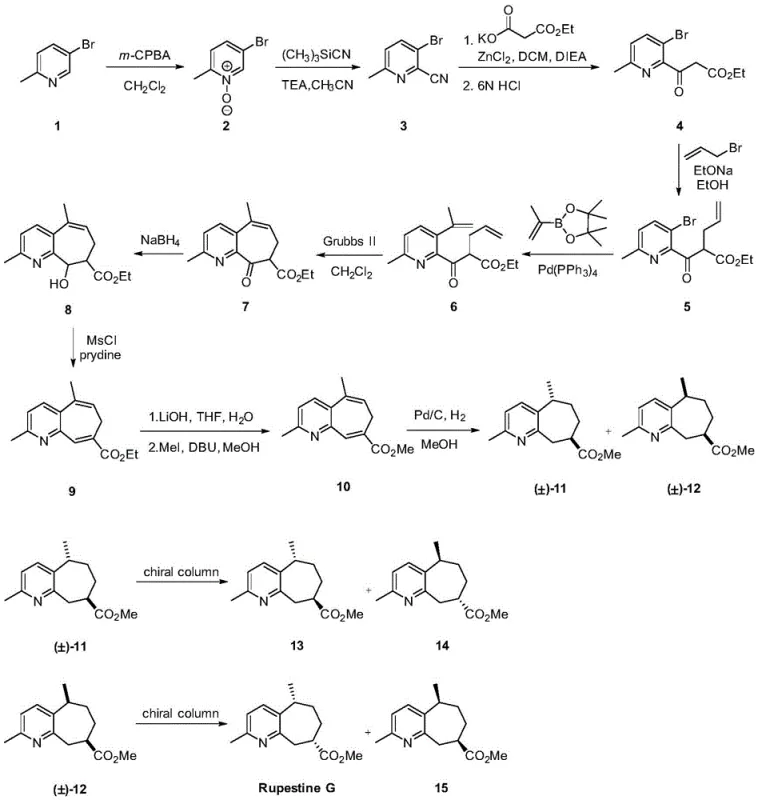
Following the construction of the carbon skeleton, the control of stereochemistry becomes paramount. The patent describes a catalytic hydrogenation step using palladium on carbon (Pd/C) to reduce the olefinic bonds. This step is diastereoselective, generating a mixture of cis and trans isomers (compounds 11 and 12) based on the facial selectivity of hydrogen addition to the fused ring system. The mechanism involves the adsorption of the substrate onto the metal surface, where steric interactions between the methyl substituents and the catalyst surface dictate the approach of hydrogen. While this step produces a mixture, it sets the relative stereochemistry required for the final resolution. The subsequent separation of enantiomers via semi-preparative chiral HPLC ensures that the specific biological activity associated with the natural (5S,8R) configuration can be isolated with high optical purity, meeting the stringent demands of modern drug discovery.
How to Synthesize Rupestine G Efficiently
The synthesis of Rupestine G described in patent CN107129462B offers a clear, step-by-step protocol that transforms simple starting materials into a complex chiral natural product. The process is designed to be robust, utilizing standard laboratory equipment and reagents that are easily sourced from global chemical suppliers. For process chemists looking to replicate or scale this route, the key lies in maintaining strict control over reaction temperatures during the exothermic oxidation steps and ensuring the removal of water during the Blaise reaction to prevent hydrolysis of the organozinc intermediate. The following guide outlines the critical phases of this manufacturing process.
- Oxidation of 2-methyl-5-bromopyridine with m-CPBA followed by Reissert-Henze cyano substitution to form the pyridine nitrile core.
- Construction of the side chain via Blaise reaction and alkylation, followed by Suzuki coupling and intramolecular olefin metathesis (RCM) using Grubbs II catalyst to close the seven-membered ring.
- Functional group manipulation including reduction, elimination, esterification, and catalytic hydrogenation, concluding with chiral HPLC resolution to isolate pure Rupestine G.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to total synthesis offers profound strategic advantages that directly impact the bottom line and operational reliability. The most immediate benefit is the decoupling of supply from agricultural variables. By synthesizing Rupestine G from petrochemical-derived precursors like bromopyridine, manufacturers eliminate the risks associated with crop failures, seasonal fluctuations, and geopolitical instability in sourcing regions. This ensures a continuous, year-round supply of high-purity material, which is critical for maintaining clinical trial timelines and commercial production schedules without interruption.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers the cost of goods sold (COGS) by replacing expensive, low-yield extraction processes with high-efficiency catalytic reactions. The use of commodity chemicals as starting materials avoids the premium pricing of natural isolates. Furthermore, the streamlined nature of the synthesis, which avoids excessive protecting group manipulations and utilizes atom-economical transformations like RCM, reduces waste generation and solvent consumption. This operational efficiency translates into substantial cost savings, allowing for more competitive pricing of the final API or intermediate without compromising on quality margins.
- Enhanced Supply Chain Reliability: Relying on a synthetic pathway diversifies the supply base, reducing dependency on single-source botanical vendors. The reagents used, such as palladium catalysts and boronic esters, are widely available from multiple tier-1 chemical suppliers, mitigating the risk of raw material shortages. Additionally, the ability to produce the compound on demand allows for better inventory management and Just-In-Time (JIT) delivery models, reducing the capital tied up in safety stock and warehousing costs for volatile natural products.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram and metric-ton commercial production. The reactions employed, such as Suzuki coupling and hydrogenation, are well-understood unit operations in the fine chemical industry, facilitating easy technology transfer to manufacturing plants. Moreover, the synthetic route offers better control over waste streams compared to biomass extraction, which generates large volumes of organic sludge. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, reducing the burden of waste treatment and enhancing the overall green profile of the manufacturing process.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the purity, scalability, and regulatory aspects of producing Rupestine G via this patented method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the key strategic advantage of this synthetic route for Rupestine G?
A: The primary advantage lies in the use of inexpensive, industrially available 2-methyl-5-bromopyridine as the starting material, combined with a robust Ring-Closing Metathesis (RCM) strategy that efficiently constructs the challenging seven-membered ring system, avoiding the low yields and supply instability associated with natural extraction.
Q: How is the stereochemistry of Rupestine G controlled in this process?
A: Stereochemical control is achieved through a combination of diastereoselective catalytic hydrogenation using Pd/C, which generates specific cis/trans isomers, followed by rigorous semi-preparative chiral HPLC resolution to separate the enantiomers and isolate the biologically active natural product configuration.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the route utilizes standard reagents and well-established catalytic cycles such as Suzuki coupling and olefin metathesis, which are amenable to scale-up. The avoidance of exotic reagents and the use of common solvents like dichloromethane and ethanol facilitate safe and cost-effective manufacturing at the metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rupestine G Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN107129462B for the pharmaceutical and agrochemical sectors. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, including air-sensitive organometallic reactions and high-pressure hydrogenation, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for global regulatory filings.
We invite you to collaborate with us to leverage this advanced technology for your specific applications. Whether you require custom synthesis of Rupestine G derivatives or bulk manufacturing of the parent compound, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation that demonstrates how our manufacturing expertise can optimize your supply chain and accelerate your project timelines.
