Scalable Biocatalytic Synthesis of Chiral Azetidine Amides and Carboxylic Acids for Pharmaceutical Intermediates
Scalable Biocatalytic Synthesis of Chiral Azetidine Amides and Carboxylic Acids for Pharmaceutical Intermediates
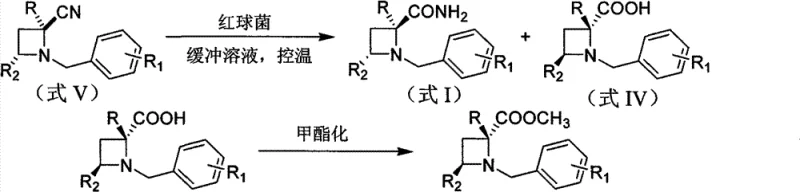
The pharmaceutical industry continuously seeks robust, sustainable, and highly selective methods for constructing chiral building blocks, particularly strained heterocycles like azetidines which are prevalent in modern drug discovery. Patent CN101898990B introduces a groundbreaking microbial system capable of catalyzing the synthesis of chiral azetidine amides and carboxylic acid compounds with exceptional stereocontrol. This technology leverages the whole-cell biocatalyst Rhodococcus erythropolis AJ270 to perform kinetic resolution on racemic N-benzylazetidine nitriles or amides. Unlike traditional chemical methods that often require harsh conditions or expensive chiral ligands, this biotransformation operates under mild physiological conditions, specifically within a pH range of 6.0 to 8.0 and temperatures between 20°C and 37°C. The result is a streamlined pathway to high-purity intermediates with absolute configurations of S and R, respectively, addressing critical needs for reliable pharmaceutical intermediate supplier capabilities in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing enantiopure azetidine derivatives frequently rely on classical resolution techniques involving diastereomeric salt formation or the use of stoichiometric chiral auxiliaries, which inherently limit the maximum theoretical yield to 50% and generate substantial chemical waste. Furthermore, chemical hydrolysis of nitriles to amides or acids often necessitates strong acidic or basic conditions at elevated temperatures, posing significant risks to the integrity of sensitive functional groups and the strained four-membered azetidine ring. These harsh environments can lead to ring-opening side reactions, polymerization, or racemization, thereby compromising the optical purity of the final product. Additionally, the removal of heavy metal catalysts or chiral reagents from the final API intermediate adds complex purification steps, increasing both the cost reduction in API manufacturing challenges and the environmental footprint of the process.
The Novel Approach
In stark contrast, the novel biocatalytic approach detailed in the patent utilizes the inherent stereoselectivity of the nitrile hydratase and amidase enzyme systems within Rhodococcus erythropolis AJ270 to differentiate between enantiomers of the racemic substrate. This method allows for the simultaneous production of a chiral amide and a chiral carboxylic acid from a single racemic starting material, effectively doubling the utility of the feedstock compared to simple destructive resolution. The reaction proceeds in aqueous buffer solutions, eliminating the need for volatile organic solvents during the transformation step and significantly simplifying the downstream workup. By operating at ambient pressures and moderate temperatures, this green chemistry approach not only preserves the structural integrity of the azetidine core but also ensures high enantiomeric excess values, often exceeding 99.5% ee, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Rhodococcus-Mediated Kinetic Resolution
The core of this technology lies in the dual-enzyme activity of the Rhodococcus erythropolis AJ270 strain, which possesses both nitrile hydratase and amidase activities that act sequentially yet with distinct stereochemical preferences. When a racemic nitrile substrate is introduced to the buffered cell suspension, the nitrile hydratase selectively hydrates one enantiomer to form the corresponding amide, while leaving the other enantiomer largely untouched or converting it at a significantly slower rate. Subsequently, the amidase component may further hydrolyze the formed amide into the carboxylic acid, depending on the specific reaction conditions and substrate structure. This cascade allows for precise tuning of the product distribution; by controlling reaction time and biomass loading, manufacturers can isolate either the chiral amide or the chiral acid as the primary product. The microorganism's cell wall structure facilitates substrate transport while protecting the intracellular enzymes, creating a stable microenvironment for catalysis that is far more resilient than isolated enzyme preparations.
Understanding the impurity profile is critical for R&D directors focusing on purity specifications. The biocatalytic nature of this reaction minimizes the formation of regioisomers or over-hydrolyzed byproducts that are common in chemical catalysis. The high chemoselectivity of the bacterial system ensures that substituents on the benzyl ring, such as bromo, methyl, or methoxy groups, remain intact during the transformation. For instance, the patent data demonstrates that substrates with electron-donating methoxy groups or electron-withdrawing bromo atoms are well-tolerated, yielding products with consistent stereochemistry. This robustness against electronic variations in the substrate structure suggests a flexible active site within the enzyme system, reducing the risk of batch-to-batch variability. Consequently, the resulting crude product requires less rigorous purification, directly contributing to cost reduction in pharmaceutical intermediate manufacturing by lowering solvent consumption and chromatography loads.
How to Synthesize Chiral Azetidine Amides Efficiently
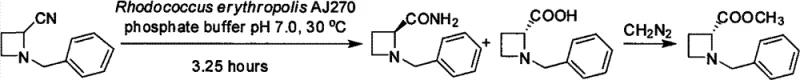
The practical implementation of this biocatalytic route is straightforward and amenable to standard fermentation and processing equipment found in most pilot and production facilities. The process begins with the preparation of a phosphate buffer system, typically potassium phosphate at pH 7.0, which maintains the optimal activity of the biocatalyst throughout the reaction duration. Wet cell mass of Rhodococcus erythropolis AJ270 is suspended in this buffer, and the racemic substrate is added, often dissolved in a water-miscible co-solvent like acetone if solubility is a concern. The mixture is then agitated at controlled temperatures, generally around 30°C, to ensure adequate oxygen transfer and substrate mixing without denaturing the biocatalyst. Reaction monitoring is easily achieved via thin-layer chromatography (TLC), allowing operators to quench the reaction at the precise point of maximum conversion and selectivity.
- Prepare a phosphate buffer solution (pH 6.0-8.0, e.g., 0.1M K2HPO4-KH2PO4) and suspend wet Rhodococcus erythropolis AJ270 cells to a concentration of 0.2-5%.
- Add the racemic N-benzylazetidine nitrile or amide substrate to the buffered cell suspension and incubate at 20-37°C with shaking (e.g., 200 rpm).
- Monitor reaction progress via TLC, then separate cells by filtration, extract the chiral amide with ethyl acetate, and recover the chiral acid from the aqueous phase for subsequent esterification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic platform offers transformative benefits regarding cost stability and supply continuity. The reliance on a fermentable microbial strain eliminates the dependency on scarce precious metal catalysts or complex chiral ligands, the prices of which are subject to volatile geopolitical and market fluctuations. Instead, the biocatalyst can be produced in-house or sourced reliably through established fermentation protocols, ensuring a consistent and renewable supply of the active catalytic agent. This shift fundamentally alters the cost structure of the synthesis, moving expenditure from high-cost reagents to scalable biological processes, which typically offer better economies of scale as production volumes increase.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and chiral resolving agents drastically reduces the raw material costs associated with producing these chiral intermediates. Furthermore, the mild reaction conditions negate the need for specialized high-pressure or cryogenic equipment, lowering capital expenditure and energy consumption. The aqueous nature of the reaction medium simplifies waste treatment, as there are no toxic heavy metal residues to dispose of, leading to substantial cost savings in environmental compliance and waste management operations.
- Enhanced Supply Chain Reliability: Biological catalysts are inherently more stable and easier to store than many sensitive chemical reagents, reducing the risk of supply disruptions due to reagent degradation. The broad substrate tolerance of the Rhodococcus system means that a single catalytic platform can be adapted for multiple analogues within a drug series, streamlining the supply chain for diverse pipeline candidates. This flexibility allows for rapid response to changing demand without the need for requalifying entirely new chemical processes for each derivative.
- Scalability and Environmental Compliance: The process has been validated at gram scales in the patent literature with consistent results, indicating a clear path to kilogram and tonne-scale production without fundamental changes to the chemistry. The use of water as the primary solvent aligns perfectly with green chemistry principles and increasingly stringent environmental regulations, future-proofing the manufacturing process against tighter emissions standards. This sustainability profile enhances the marketability of the final API to environmentally conscious partners and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring that the answers reflect the proven capabilities of the system. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What is the enantioselectivity of the Rhodococcus erythropolis AJ270 catalyzed hydrolysis?
A: The biocatalytic system demonstrates exceptional enantioselectivity, consistently achieving ee values greater than 99.5% for various substrates including unsubstituted and substituted phenyl derivatives.
Q: Can this biocatalytic process handle diverse substrate substituents?
A: Yes, the Rhodococcus erythropolis AJ270 strain exhibits broad substrate specificity, successfully catalyzing the hydrolysis of nitriles and amides bearing hydrogen, methyl, methoxy, and bromo groups at ortho, meta, or para positions.
Q: Is this method suitable for large-scale industrial production?
A: Absolutely. The patent validates gram-scale preparations with consistent yields and purity, and the use of robust whole-cell catalysts in aqueous buffers facilitates straightforward scale-up to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Azetidine Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative biocatalytic platforms in modern drug development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm the >99.5% ee values achievable with this Rhodococcus-mediated process. We are committed to delivering high-purity chiral azetidine intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical procurement team to explore how this biocatalytic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your project benefits from the highest levels of quality and efficiency available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
