Advanced Photocatalytic Synthesis of Difluoromethylated 2,3-Dihydrobenzopyran-4-One Derivatives for Industrial Scale
Advanced Photocatalytic Synthesis of Difluoromethylated 2,3-Dihydrobenzopyran-4-One Derivatives for Industrial Scale
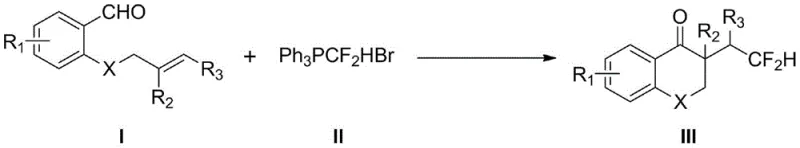
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate fluorine motifs into complex molecular scaffolds, driven by the unique metabolic stability and binding affinity these groups confer to bioactive molecules. Patent CN114853707A introduces a groundbreaking visible-light-induced protocol for the synthesis of difluoromethylated 2,3-dihydrobenzopyran-4-one derivatives, a privileged scaffold found in numerous therapeutic agents. This innovation leverages a tandem difluorohydromethylation-cyclization strategy that constructs both C-CF2H and C-C(O) bonds in a single operational step, utilizing stable difluoromethyltriphenylphosphonium bromide as the fluorinating source. By replacing hazardous liquid reagents with stable solids and employing commercially available photocatalysts, this technology addresses critical safety and scalability bottlenecks inherent in previous synthetic routes, positioning it as a vital asset for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
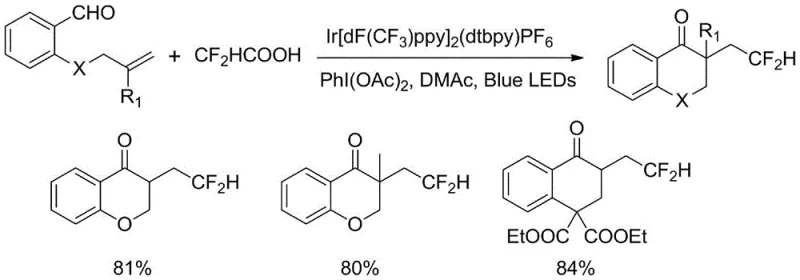
Historically, the construction of the 2,3-dihydrobenzopyran-4-one skeleton has relied on classical transformations such as Aldol condensations, intramolecular Stetter reactions, or transition-metal-catalyzed radical cyclizations. While effective in academic settings, many of these traditional pathways suffer from significant industrial drawbacks, including the requirement for harsh reaction temperatures, poor regioselectivity, and the use of expensive or toxic metal catalysts. Specifically, recent photocatalytic approaches reported prior to this invention often utilized difluoroacetic acid as the difluoromethylating reagent, which poses severe handling challenges due to its strong corrosivity, pungent odor, and high toxicity. Furthermore, the photocatalysts employed in these earlier methods, such as Ir[dF(CF3)ppy]2(dtbpy)PF6, are not commercially available and require tedious, multi-step synthesis under inert atmosphere conditions, creating a barrier to entry for large-scale manufacturing and increasing the overall cost of goods.
The Novel Approach
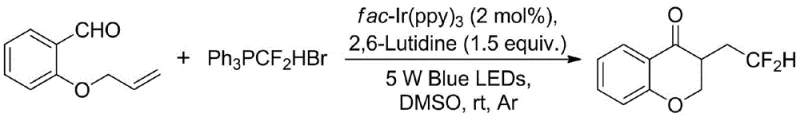
The methodology disclosed in CN114853707A represents a paradigm shift by utilizing difluoromethyltriphenylphosphonium bromide, a stable and easily handled solid reagent, to replace the hazardous difluoroacetic acid. This substitution fundamentally alters the safety profile of the process, eliminating the need for specialized corrosion-resistant equipment and reducing environmental hazards associated with volatile acidic waste. Additionally, the process employs fac-Ir(ppy)3, a robust and commercially sourced photocatalyst, which removes the dependency on custom-synthesized catalytic materials. The reaction proceeds under mild conditions using visible blue LED irradiation in dimethyl sulfoxide (DMSO), a common analytical grade solvent, thereby simplifying the workflow and enabling the efficient one-pot assembly of complex fluorinated heterocycles with high atom economy and operational simplicity suitable for modern green chemistry standards.
Mechanistic Insights into Visible-Light Induced Radical Tandem Cyclization
The core of this synthetic advancement lies in the elegant mechanistic pathway where visible light energy drives the generation of difluoromethyl radicals from the phosphonium salt precursor. Upon irradiation, the excited state of the fac-Ir(ppy)3 photocatalyst facilitates a single-electron transfer (SET) process that activates the difluoromethyltriphenylphosphonium bromide, releasing the reactive •CF2H radical species. This radical subsequently adds to the electron-rich alkene moiety of the alkenal substrate, initiating a cascade that culminates in an intramolecular cyclization onto the aldehyde carbonyl group. This tandem sequence effectively forges two critical bonds simultaneously: the carbon-carbon bond linking the difluoromethyl group and the carbon-oxygen bond closing the pyran ring. The use of 2,6-lutidine as a base is crucial for neutralizing the acidic byproducts and facilitating the final oxidation or proton transfer steps required to aromatize or stabilize the ketone functionality, ensuring high conversion rates without the need for external oxidants.
From an impurity control perspective, this radical mechanism offers superior selectivity compared to ionic pathways that might suffer from competing nucleophilic attacks or polymerization side reactions. The mild nature of the photocatalytic cycle minimizes thermal degradation of sensitive functional groups, which is often a concern when introducing fluorine atoms using aggressive reagents. The patent data indicates that the reaction tolerates a wide array of substituents on the aromatic ring, including electron-donating methyl groups and electron-withdrawing cyano or ester moieties, without significant loss in yield. This robustness suggests that the radical intermediate is sufficiently long-lived to find the correct intramolecular partner but reactive enough to overcome steric barriers, resulting in a clean product profile that simplifies downstream purification and reduces the burden on quality control laboratories tasked with identifying trace impurities in final API intermediates.
How to Synthesize Difluoromethylated Chromanones Efficiently
The practical implementation of this technology involves a straightforward procedure accessible to standard organic synthesis laboratories equipped with basic photochemical apparatus. The process begins with the precise weighing of the alkenal substrate and the difluoromethylating phosphonium salt, followed by their dissolution in DMSO under an inert argon atmosphere to prevent oxygen quenching of the excited photocatalyst. The addition of the base and the photocatalyst completes the reaction mixture, which is then subjected to continuous irradiation with 5W blue LEDs at room temperature. Detailed standardized synthesis steps, including specific molar ratios, workup procedures involving aqueous extraction, and purification protocols via column chromatography, are outlined in the technical guide below to ensure reproducibility and optimal yield for process chemists scaling this route.
- Prepare the reaction mixture by combining the alkenal substrate, difluoromethyltriphenylphosphonium bromide, and fac-Ir(ppy)3 photocatalyst in DMSO under argon.
- Add 2,6-lutidine as the base and irradiate the solution with 5W blue LEDs at room temperature for 24 hours to facilitate the radical cyclization.
- Quench the reaction with water, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel photocatalytic method offers tangible strategic benefits that extend beyond mere chemical efficiency. The replacement of custom-synthesized, exotic iridium complexes with off-the-shelf fac-Ir(ppy)3 drastically simplifies the sourcing landscape, reducing lead times and mitigating the risk of supply disruptions associated with niche reagents. Furthermore, the use of stable solid reagents instead of corrosive liquids lowers transportation and storage costs, as special hazmat handling is no longer required for the fluorinating agent. The ability to run the reaction at room temperature using low-energy LED lights also contributes to significant operational expenditure savings by reducing the thermal load on manufacturing facilities and minimizing the need for energy-intensive heating or cooling systems, aligning perfectly with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of complex catalyst synthesis and the use of commodity-grade solvents like DMSO directly lower the raw material costs per kilogram of product. By avoiding the need for glovebox operations and specialized corrosion-resistant reactors required for difluoroacetic acid, capital expenditure for plant retrofitting is minimized, allowing for immediate integration into existing multipurpose manufacturing suites without significant infrastructure investment.
- Enhanced Supply Chain Reliability: Sourcing stable phosphonium salts and commercial photocatalysts ensures a consistent supply of critical inputs, unlike custom reagents that may have long lead times or limited vendor availability. The robustness of the reaction conditions means that production schedules are less likely to be impacted by minor fluctuations in environmental parameters, guaranteeing reliable delivery timelines for downstream pharmaceutical clients who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods involving strong acids or heavy metal oxidants, simplifying effluent treatment and reducing disposal costs. The mild reaction conditions and high selectivity facilitate straightforward scale-up from gram to kilogram scales, enabling rapid technology transfer from R&D to pilot and commercial production while maintaining strict adherence to increasingly stringent environmental regulations regarding fluorine-containing waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology, derived from the specific advantages and operational parameters detailed in the patent literature. These insights are intended to clarify the feasibility of adopting this route for large-scale manufacturing and to highlight the specific value propositions for stakeholders involved in process development and strategic sourcing of fluorinated intermediates.
Q: What are the safety advantages of using difluoromethyltriphenylphosphonium bromide over difluoroacetic acid?
A: Difluoromethyltriphenylphosphonium bromide is a stable, solid reagent that eliminates the strong corrosivity, toxicity, and irritating odor associated with liquid difluoroacetic acid, significantly improving operational safety and environmental compliance in manufacturing.
Q: Why is fac-Ir(ppy)3 preferred over specialized iridium complexes for this transformation?
A: fac-Ir(ppy)3 is a commercially available, off-the-shelf photocatalyst that avoids the complex, glovebox-dependent synthesis required for specialized catalysts like Ir[dF(CF3)ppy]2(dtbpy)PF6, thereby reducing material costs and supply chain complexity.
Q: Does this photocatalytic method support diverse substrate scopes for drug discovery?
A: Yes, the method demonstrates excellent tolerance for various functional groups including methyl, tert-butyl, ester, and cyano substituents on the aromatic ring, as well as different alkene linkers, making it highly versatile for synthesizing diverse pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylated Chromanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of patent CN114853707A in streamlining the production of high-value fluorinated pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of difluoromethylated 2,3-dihydrobenzopyran-4-one derivative meets the exacting standards required for global drug development pipelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis technology for your next-generation therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in photocatalytic manufacturing can accelerate your project timelines and optimize your overall cost structure.
