Advanced Pd/Cu Co-Catalyzed Synthesis of Imidazophenanthridine Compounds for Commercial Scale-Up
Advanced Pd/Cu Co-Catalyzed Synthesis of Imidazophenanthridine Compounds for Commercial Scale-Up
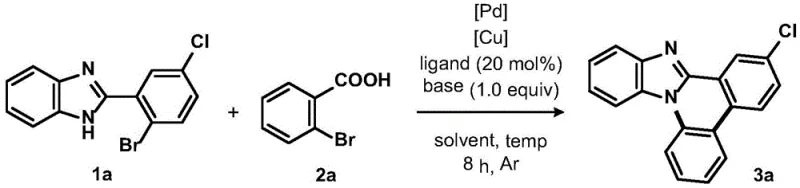
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern pharmaceutical research, particularly for scaffolds exhibiting potent biological activity. Patent CN115385909A introduces a groundbreaking palladium-copper co-catalyzed methodology for the synthesis of imidazophenanthridine compounds, addressing critical limitations in existing literature regarding yield, substrate scope, and reaction severity. This technology leverages a synergistic dual-metal catalytic system to facilitate an intermolecular [4+2] cyclization between 2-(2-bromophenyl)benzimidazoles and o-bromobenzoic acids. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process chemistry, offering a robust pathway to access complex phenanthridine derivatives that serve as key precursors for DNA intercalators and antitumor agents.
The strategic value of this invention lies in its ability to construct the imidazophenanthridine skeleton through a modular one-pot procedure that eliminates the need for harsh reaction conditions often associated with traditional C-H activation strategies. By utilizing o-halobenzoic acids as versatile building blocks, the method not only simplifies the synthetic design but also enhances the atom economy of the transformation. The process operates effectively within a temperature range of 100 to 120°C, utilizing standard organic solvents like DMF, which ensures compatibility with existing manufacturing infrastructure. This technical advancement provides a compelling solution for cost reduction in pharmaceutical intermediates manufacturing by streamlining the production of high-value heterocyclic cores essential for next-generation therapeutic development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of phenanthridine derivatives has historically been plagued by significant operational challenges that hinder industrial adoption. For instance, earlier methodologies reported by the Miura group in 2012 relied on direct cyclization via C-H and N-H bond cleavage, which often suffered from narrow substrate tolerance and required forcing conditions to drive the reaction to completion. Similarly, approaches described by Chen et al. in 2014 necessitated extremely high temperatures of 160°C and prolonged reaction times, leading to increased energy consumption and potential thermal degradation of sensitive functional groups. Furthermore, electrochemical methods introduced by Zhang's group in 2021, while innovative, were limited by restricted substrate scope and the complexity of setting up specialized electrochemical cells, making them less viable for large-scale commercial production. These conventional pathways frequently result in low product yields and generate substantial waste, failing to meet the rigorous standards of green chemistry required by modern regulatory frameworks.
The Novel Approach
In stark contrast, the Pd/Cu co-catalyzed method disclosed in CN115385909A offers a transformative solution by enabling the construction of the target scaffold under significantly milder thermal conditions. The innovation utilizes a dual-catalyst system where palladium acetate and cuprous iodide work in concert to activate both the benzimidazole and the benzoic acid components simultaneously. This cooperative catalysis allows for the formation of the critical C-N bond and subsequent ring closure without the need for extreme heat or exotic reagents. The reaction proceeds efficiently in common polar aprotic solvents, and the use of cesium carbonate as a base ensures smooth deprotonation and neutralization of acidic byproducts. This novel approach not only improves the overall yield but also simplifies the downstream purification process, as the reaction mixture can be worked up using standard extraction and chromatography techniques, thereby enhancing the practical utility of the method for industrial applications.
Mechanistic Insights into Pd/Cu Co-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated dance of organometallic steps that underscores the necessity of the bimetallic system. The cycle initiates with the oxidative addition of the o-bromobenzoic acid to the palladium(0) species, generating a reactive aryl-palladium intermediate. Under basic conditions, this intermediate undergoes a C(sp2)-N bond coupling with the 2-(2-bromophenyl)benzimidazole substrate, followed by a reductive elimination step that forms a key biaryl intermediate. Crucially, the copper catalyst then enters the cycle, facilitating a second oxidative addition to form a copper-ring intermediate. This unique copper-mediated step is followed by a decarboxylation event, which drives the thermodynamic equilibrium towards the final product, and a final reductive elimination releases the imidazophenanthridine compound while regenerating the active catalytic species. This intricate mechanism explains why single-metal systems fail to achieve comparable efficiency, as they lack the specific capability to mediate the decarboxylative cyclization effectively.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity pharmaceutical intermediates. The specificity of the oxidative addition steps minimizes side reactions such as homocoupling of the aryl halides, which are common pitfalls in palladium-catalyzed cross-couplings. Furthermore, the decarboxylation step serves as a driving force that pushes the reaction to completion, reducing the amount of unreacted starting material remaining in the crude mixture. The use of triphenylphosphine as a ligand stabilizes the metal centers and prevents the formation of palladium black, which can act as a sink for the catalyst and lead to inconsistent batch-to-batch performance. By understanding these mechanistic nuances, process chemists can fine-tune parameters such as ligand loading and base equivalents to maximize selectivity, ensuring that the final API intermediate meets stringent quality specifications required for clinical trials and commercial supply.
How to Synthesize Imidazophenanthridine Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves a straightforward one-pot procedure where all reagents are combined in a single vessel, eliminating the need for intermediate isolation steps that often lead to material loss. Detailed standardized synthesis steps for this high-efficiency route are provided in the guide below, outlining the precise stoichiometry and thermal profiles required to achieve optimal results. By adhering to these guidelines, manufacturers can consistently produce high-quality imidazophenanthridine derivatives with minimal variability, ensuring a stable supply chain for downstream drug development projects.
- Combine 2-(2-bromophenyl)benzimidazole, o-bromobenzoic acid, palladium acetate, cuprous iodide, triphenylphosphine, and cesium carbonate in DMF solvent.
- Heat the reaction mixture to 110-120°C under an inert atmosphere and stir for approximately 8 hours until TLC indicates completion.
- Quench the reaction in water, extract with ethyl acetate, wash the organic phase, dry over calcium chloride, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Pd/Cu co-catalyzed technology translates into tangible operational benefits that directly impact the bottom line. The shift from high-temperature, long-duration processes to a milder, faster protocol significantly reduces energy consumption and reactor occupancy time, allowing for greater throughput within existing facilities. Moreover, the use of commercially available and inexpensive starting materials like o-bromobenzoic acid and simple benzimidazoles mitigates the risk of supply chain disruptions associated with exotic or custom-synthesized reagents. This robustness ensures a continuous flow of materials, which is critical for maintaining production schedules in the fast-paced pharmaceutical industry.
- Cost Reduction in Manufacturing: The elimination of extreme reaction conditions and the use of a one-pot strategy drastically simplify the manufacturing process, leading to substantial cost savings. By avoiding the need for specialized high-pressure equipment or extended heating cycles, the operational expenditure is significantly lowered. Additionally, the high efficiency of the catalyst system means that lower loadings of expensive palladium can be utilized without compromising yield, further optimizing the cost structure of the final product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, cesium carbonate, and triphenylphosphine ensures that the supply chain remains resilient against market volatility. These reagents are widely produced and stocked by multiple global suppliers, reducing the dependency on single-source vendors. This diversification of the supply base enhances the reliability of raw material delivery, ensuring that production timelines are met without unexpected delays caused by material shortages.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by mild conditions and straightforward workup procedures, aligns perfectly with modern environmental regulations. The simplified purification process reduces the volume of solvent waste generated, lowering disposal costs and environmental impact. Furthermore, the scalability of the reaction from gram to kilogram scales has been demonstrated, confirming its viability for commercial-scale production of complex pharmaceutical intermediates without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic method. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on critical process parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their current manufacturing workflows.
Q: What are the optimal reaction conditions for this Pd/Cu co-catalyzed synthesis?
A: The optimal conditions involve using palladium acetate (5 mol%) and cuprous iodide (10 mol%) with triphenylphosphine ligand in DMF solvent at 110-120°C for 8 hours.
Q: Why is the Pd/Cu co-catalytic system superior to single metal catalysis?
A: The co-catalytic system enables a unique intermolecular [4+2] cyclization via decarboxylation that single palladium catalysis cannot achieve efficiently, resulting in higher yields and milder conditions.
Q: Is this method suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the method uses readily available raw materials, operates at moderate temperatures, and involves a simple one-pot procedure with straightforward workup, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazophenanthridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Pd/Cu co-catalyzed synthesis described in CN115385909A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of imidazophenanthridine intermediate delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a successful partnership driven by scientific excellence and commercial reliability.
