Optimizing Rivaroxaban Production: A Seven-Step High-Efficiency Synthetic Route for Commercial Scale
Optimizing Rivaroxaban Production: A Seven-Step High-Efficiency Synthetic Route for Commercial Scale
The pharmaceutical industry continuously seeks robust manufacturing processes for critical anticoagulants, and the preparation method disclosed in patent CN112159402B represents a significant advancement in the synthesis of rivaroxaban. This intellectual property details a novel seven-step efficient synthesis method that utilizes p-nitroaniline and (S)-N-glycidylphthalimide as primary raw materials, fundamentally restructuring the production pathway to enhance yield and purity. By integrating addition, cyclization, hydrolysis, amidation, reduction, and final cyclization reactions, this technology addresses the longstanding challenges of impurity control and operational safety associated with earlier generations of synthetic routes. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is crucial for securing a reliable rivaroxaban intermediate supplier capable of meeting stringent regulatory standards. The following analysis dissects the technical superiority of this approach, highlighting its potential for cost reduction in API manufacturing and improved supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of rivaroxaban has relied on several distinct synthetic pathways, each plagued by specific technical bottlenecks that hinder scalability and cost-efficiency. For instance, Route A, often cited in early literature, involves a nitration reaction in its second step which presents severe selectivity issues and potential safety hazards due to the exothermic nature of nitration chemistry. Furthermore, this route generates significant amounts of hazardous waste and often results in products containing genotoxic impurities that are notoriously difficult to remove to acceptable ppm levels. As illustrated in the reaction scheme below, the complexity of side reactions in traditional nitration-based routes compromises the overall yield and increases the burden on downstream purification processes.
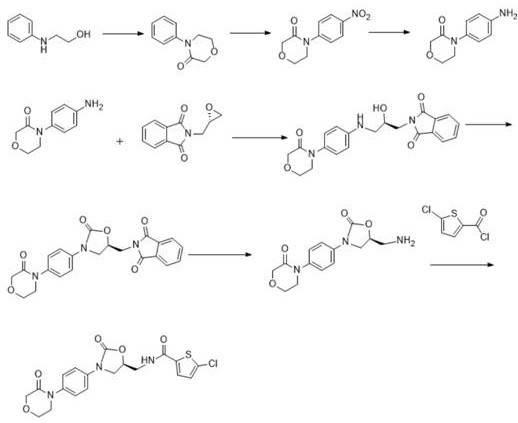
Similarly, Route B and Route C exhibit their own distinct disadvantages that make them less attractive for modern GMP manufacturing environments. Route B typically yields a product with darker coloration and higher impurity profiles, complicating the development of a robust quality control strategy and requiring extensive decolorization steps. Meanwhile, Route C suffers from low overall yields and poor chiral purity, frequently necessitating multiple rounds of column chromatography for purification. The reliance on chromatography is a critical bottleneck for commercial scale-up, as it is labor-intensive, solvent-heavy, and difficult to automate for multi-ton production. These legacy methods collectively underscore the urgent need for a streamlined process that eliminates hazardous steps and simplifies purification.
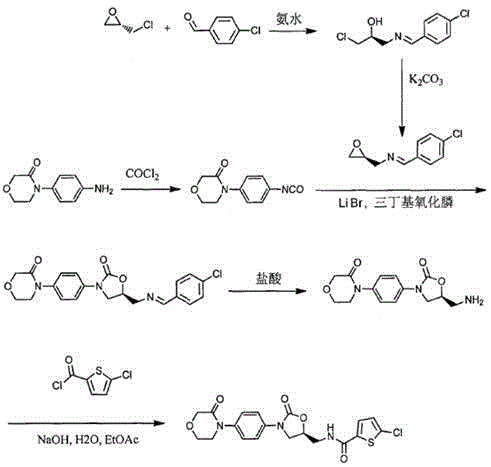
The Novel Approach
In stark contrast to the limitations of legacy methods, the novel seven-step synthesis described in CN112159402B offers a transformative solution by leveraging a condensation-cyclization strategy that bypasses hazardous nitration entirely. This approach initiates with the reaction of p-nitroaniline and (S)-N-glycidylphthalimide, establishing the chiral center early in the sequence with high fidelity. The subsequent steps involve carefully controlled temperature regimes, such as maintaining 0-10°C during condensing agent addition, to minimize side reactions and maximize the formation of the desired oxazolidinone core. By replacing chromatographic purification with crystallization and filtration unit operations, this method drastically simplifies the workflow, making it inherently more suitable for commercial scale-up of complex pharmaceutical intermediates. The result is a process that not only improves the environmental footprint by reducing three wastes but also enhances the economic viability of producing high-purity rivaroxaban.
Mechanistic Insights into the Seven-Step Condensation-Cyclization Strategy
The core innovation of this patented methodology lies in its precise orchestration of reaction conditions to control stereochemistry and impurity profiles throughout the seven-step sequence. The initial addition reaction between p-nitroaniline and (S)-N-glycidylphthalimide is conducted in solvents like ethanol or isopropanol at temperatures ranging from 30-90°C, ensuring complete conversion while preserving the optical purity of the chiral epoxide. Following this, the cyclization step utilizes condensing agents such as N,N'-carbonyldiimidazole (CDI) or bis(trichloromethyl) carbonate at low temperatures (0-20°C) to facilitate ring closure without racemization. This meticulous control over reaction parameters is essential for preventing the formation of diastereomers, which are critical quality attributes for the final anticoagulant activity. The subsequent hydrolysis with methylamine and amidation with 5-chlorothiophene-2-carbonyl chloride further refine the molecular structure, setting the stage for the final ring closure.
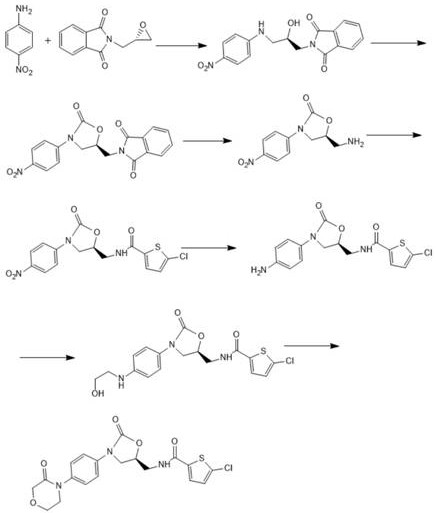
Impurity control is rigorously maintained through the strategic selection of reagents and purification techniques at each stage. For example, the reduction step employs catalysts like Raney nickel or palladium carbon under hydrogen pressure, followed by filtration to remove metal residues effectively, thereby addressing heavy metal concerns common in catalytic processes. The final cyclization involves the reaction with chloroacetyl chloride under alkaline conditions (pH 10-12), where the pH is tightly monitored to prevent hydrolysis of the sensitive amide bonds. The use of sodium iodide as an additive in the final step further enhances the reaction kinetics and yield. By avoiding column chromatography and relying on crystallization from solvent systems like ethanol-water mixtures, the process ensures that the final product meets stringent purity specifications without the risk of solvent entrapment or silica gel contamination, providing a robust framework for reducing lead time for high-purity anticoagulant intermediates.
How to Synthesize Rivaroxaban Efficiently
The implementation of this seven-step synthesis requires strict adherence to the optimized reaction conditions and stoichiometry defined in the patent to achieve the reported high yields. The process begins with the preparation of Intermediate I through condensation, followed by sequential transformations involving cyclization, hydrolysis, and amidation, each requiring specific solvent systems and temperature controls to ensure reproducibility. Detailed standard operating procedures for each step, including exact molar ratios of reagents like ethylene oxide and chloroacetyl chloride, are critical for maintaining batch-to-batch consistency. For a comprehensive guide on executing these reactions in a pilot or production plant setting, please refer to the standardized synthesis steps outlined below.
- Condensation of p-nitroaniline with (S)-N-glycidylphthalimide to form Intermediate I.
- Cyclization using CDI followed by methylamine hydrolysis to generate the oxazolidinone core.
- Amidation with 5-chlorothiophene-2-carbonyl chloride followed by catalytic hydrogenation and final morpholine ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial strategic benefits for procurement managers and supply chain heads seeking to optimize their sourcing strategies for anticoagulant ingredients. By eliminating the need for hazardous nitration reactions and expensive chromatographic purification, the process inherently reduces the operational costs associated with waste disposal, solvent recovery, and specialized equipment maintenance. This structural efficiency translates into a more competitive cost structure, allowing suppliers to offer better pricing stability in a volatile raw material market. Furthermore, the reliance on readily available starting materials like p-nitroaniline mitigates the risk of supply disruptions, ensuring a continuous flow of intermediates necessary for uninterrupted API production.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a primary driver for cost optimization, as this unit operation is notoriously resource-intensive and difficult to scale economically. By substituting chromatography with crystallization and filtration, the process significantly lowers solvent consumption and labor hours, leading to substantial cost savings in the overall manufacturing budget. Additionally, the high yield achieved in each step minimizes the loss of valuable chiral intermediates, further enhancing the material efficiency of the production line. These factors combined create a lean manufacturing model that maximizes output while minimizing variable costs.
- Enhanced Supply Chain Reliability: The use of common industrial solvents such as ethanol, methanol, and toluene, along with widely available reagents like triethylamine and sodium hydroxide, ensures that the supply chain is not dependent on exotic or single-source chemicals. This diversity in sourcing options provides a buffer against market fluctuations and logistical bottlenecks, guaranteeing that production schedules can be met consistently. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, reducing the frequency of batch failures and reworks.
- Scalability and Environmental Compliance: The process design prioritizes safety and environmental sustainability by avoiding genotoxic reagents and minimizing the generation of hazardous waste streams. The simplified workup procedures, which primarily involve phase separation and crystallization, are easily adaptable to large-scale reactors, facilitating a smooth transition from laboratory bench to multi-ton commercial production. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory compliance, accelerating the approval process for new drug master files.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rivaroxaban synthesis method. These answers are derived directly from the patent data and practical experience in scaling similar organic synthesis pathways. They are intended to provide clarity on the feasibility, quality, and logistical aspects of adopting this technology for your manufacturing needs.
Q: How does this new route improve impurity control compared to traditional methods?
A: Unlike conventional routes that require column chromatography or generate genotoxic impurities during nitration, this seven-step method utilizes crystallization for purification, significantly reducing residual solvents and heavy metals while ensuring high chiral purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process avoids hazardous nitration reactions and expensive chromatographic purification, relying instead on robust unit operations like filtration and crystallization, which are ideal for scaling from kilograms to metric tons.
Q: What are the key raw materials required for this synthesis?
A: The primary starting materials are p-nitroaniline and (S)-N-glycidylphthalimide, which are commercially available and cost-effective, ensuring a stable supply chain for continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent methodologies into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN112159402B route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that every kilogram of rivaroxaban intermediate we supply meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to evaluate how this optimized synthesis route can enhance your supply chain efficiency and reduce your overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to support your long-term production goals. Let us help you secure a stable, high-quality supply of this critical anticoagulant intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
