Advanced Cobalt-Catalyzed Asymmetric Hydrogenation for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric catalysis is undergoing a significant transformation, driven by the urgent need to replace expensive and toxic noble metals with earth-abundant alternatives. Patent CN111690009A introduces a groundbreaking class of phosphine pyridine oxazoline compounds and their corresponding metal complexes, specifically designed to address the limitations of traditional catalytic systems. This technology leverages the unique electronic and steric properties of cobalt and iron centers coordinated with chiral P-N-O ligands to achieve exceptional stereocontrol. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable synthetic routes for chiral organosilicon compounds, which are critical building blocks in modern medicinal chemistry and materials science.
Historically, the asymmetric hydrogenation of non-activated olefins, particularly alpha-alkenyl silanes, has relied heavily on precious metal catalysts such as Ruthenium, Rhodium, and Iridium. While effective, these systems suffer from inherent drawbacks including high raw material costs, supply chain volatility, and stringent regulatory limits on residual metal content in final pharmaceutical products. Early attempts to utilize base metals like cobalt, such as the work by Ohgo et al., often resulted in poor enantioselectivity, typically ranging from 7% to 49% ee, rendering them commercially unviable for high-value applications. Furthermore, many existing base metal systems require harsh reaction conditions or exhibit narrow substrate scope, limiting their utility in complex molecule synthesis.
The novel approach disclosed in CN111690009A overcomes these historical barriers through the rational design of a tridentate phosphine pyridine oxazoline ligand framework. By integrating a chiral oxazoline moiety with a phosphine arm on a pyridine scaffold, the invention creates a rigid chiral environment around the cobalt or iron center. This architecture enables the catalyst to effectively differentiate between prochiral faces of the substrate, delivering outstanding enantioselectivity even for sterically demanding substrates. The general reaction scheme illustrates the efficient conversion of alpha-alkenyl silanes to chiral silanes with high fidelity.
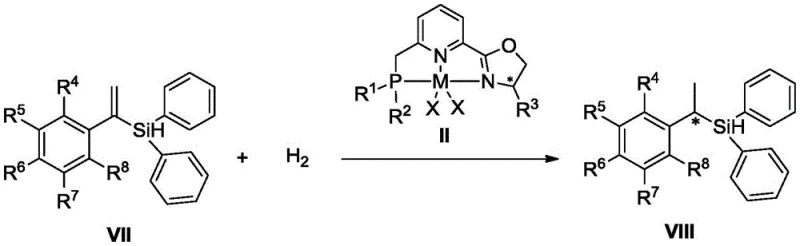
Mechanistically, the success of this system lies in the synergistic cooperation between the soft phosphine donor and the hard nitrogen/oxygen donors within the ligand structure. The synthesis of the ligand itself is robust and scalable, involving a Lewis acid-catalyzed condensation followed by a lithiation-phosphination sequence. This modular synthesis allows for easy tuning of the steric bulk at the phosphorus and oxazoline positions, enabling fine-tuning of the catalyst for specific substrate classes. The subsequent complexation with simple metal salts like CoCl2 proceeds smoothly under mild conditions, generating the active catalytic species in situ or as an isolable solid.
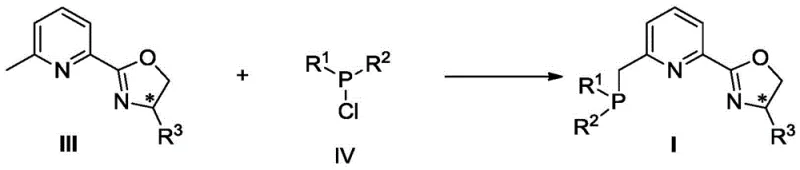
The formation of the active metal complex is a critical step that defines the catalytic performance. The patent details a straightforward protocol where the pre-formed ligand reacts with anhydrous metal halides in ether solvents. This process ensures the formation of a well-defined coordination geometry that is essential for high stereoinduction. The resulting complexes, characterized by their distinct spectroscopic signatures, exhibit remarkable stability and activity. This mechanistic robustness translates directly to process reliability, minimizing batch-to-batch variability—a key concern for quality assurance teams in regulated industries.
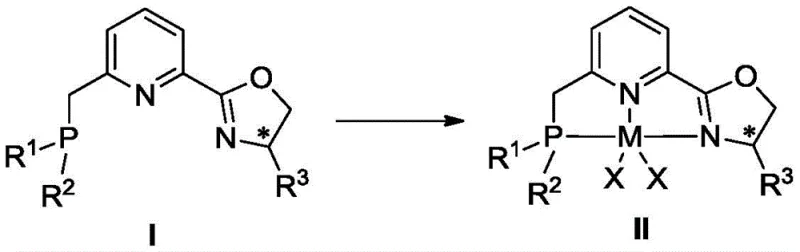
For process development teams looking to implement this technology, the synthetic route is designed for operational simplicity and safety. The preparation of the catalyst precursors avoids the use of highly hazardous reagents where possible, and the hydrogenation step itself operates at near-ambient temperatures and low pressures. Detailed standardized synthesis steps for the ligand and complex preparation are provided in the technical documentation below, ensuring reproducibility across different manufacturing sites.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from noble metal to base metal catalysis offers profound economic and logistical benefits for the supply chain. By substituting expensive ruthenium or rhodium with abundant cobalt or iron, manufacturers can achieve substantial cost reduction in fine chemical manufacturing. This is not merely a saving on the catalyst price itself but extends to the entire downstream purification process. The elimination of rigorous heavy metal scavenging steps, which are mandatory for noble metal residues, significantly reduces the consumption of specialized resins and solvents, thereby lowering the overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The utilization of earth-abundant metals drastically lowers the raw material expenditure compared to traditional precious metal systems. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and high-pressure compression. The simplified workup procedure, which often requires only basic quenching and filtration rather than complex chromatographic purification to remove metal traces, leads to significant operational savings and higher overall throughput.
- Enhanced Supply Chain Reliability: Cobalt and iron are globally abundant and not subject to the same geopolitical supply constraints as platinum group metals. This ensures a stable and continuous supply of catalytic materials, mitigating the risk of production delays due to raw material shortages. The robustness of the catalyst also means lower loading levels may be achievable, further insulating the supply chain from price volatility in the metals market.
- Scalability and Environmental Compliance: The process operates under mild conditions with low hydrogen pressure, reducing the safety infrastructure requirements for large-scale reactors. Additionally, the use of less toxic metals aligns with green chemistry principles and increasingly strict environmental regulations regarding heavy metal discharge. This facilitates easier permitting and waste management, supporting sustainable manufacturing goals without compromising on yield or selectivity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed hydrogenation technology. These answers are derived directly from the experimental data and specifications outlined in the patent literature, providing a reliable foundation for feasibility assessments.
Q: What are the advantages of using Cobalt over Ruthenium for asymmetric hydrogenation?
A: Cobalt is significantly more abundant and less expensive than noble metals like Ruthenium or Rhodium. Furthermore, cobalt catalysts often eliminate the need for rigorous heavy metal scavenging steps required to meet strict pharmaceutical residual metal limits, thereby simplifying downstream processing.
Q: What level of enantioselectivity can be achieved with this catalyst system?
A: According to patent CN111690009A, the phosphine pyridine oxazoline cobalt complexes demonstrate excellent enantioselectivity, achieving up to 99% ee in the asymmetric hydrogenation of sterically hindered alpha-silyl styrene substrates.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process operates under mild conditions, typically requiring low hydrogen pressures (2-10 bar) and temperatures ranging from 10°C to 35°C. These mild parameters reduce energy consumption and safety risks associated with high-pressure hydrogenation, facilitating easier commercial scale-up.
- Condense 2-cyano-6-methylpyridine with chiral amino alcohols using a Lewis acid catalyst like Zn(OTf)2 in toluene under reflux to form the chiral oxazoline intermediate.
- Perform a substitution reaction by treating the oxazoline intermediate with LDA at low temperature (-78°C), followed by the addition of chlorophosphines to install the phosphine arm.
- Complex the resulting ligand with anhydrous cobalt chloride (CoCl2) or iron salts in tetrahydrofuran under inert atmosphere to generate the active catalytic species.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Silane Supplier
The technological potential of phosphine pyridine oxazoline cobalt complexes represents a major leap forward in the synthesis of chiral organosilicon intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess values, ensuring that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative catalytic technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current process. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for efficiency and sustainability.
