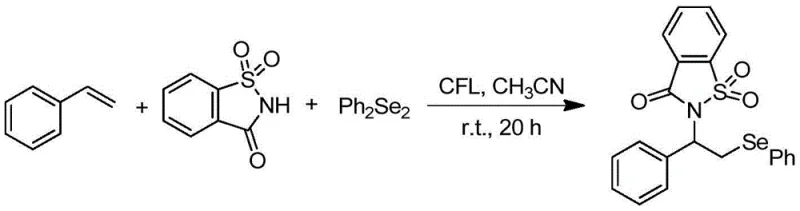
Advanced Visible Light-Promoted Synthesis of Beta-Amino Selenides for Commercial Scale-Up
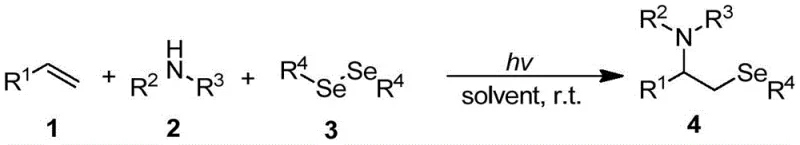
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to synthesize complex organic molecules, particularly those containing selenium which exhibit significant biological activity. Patent CN111217766A introduces a groundbreaking method for the synthesis of visible light-promoted beta-amino selenides, addressing critical limitations in current organic synthetic chemistry. This technology leverages visible light irradiation to drive the coupling of olefins, nitrogen-containing compounds, and diselenides under exceptionally mild conditions. For R&D Directors and Procurement Managers, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity. The ability to generate high-value selenium-containing intermediates without harsh thermal conditions or toxic heavy metal catalysts opens new avenues for drug discovery and material science applications. As a reliable pharmaceutical intermediates supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing carbon-selenium bonds often rely heavily on the use of stoichiometric amounts of oxidants, strong acids, or expensive transition metal catalysts which pose significant environmental and safety challenges. These conventional methods frequently require elevated temperatures and rigorous anhydrous conditions, leading to higher energy consumption and increased operational risks in a manufacturing setting. Furthermore, the use of pre-prepared organic selenium reagents as reaction precursors adds extra synthetic steps, reducing overall atom economy and increasing the cost of goods sold. The presence of transition metals also necessitates stringent purification processes to meet regulatory standards for residual metals in pharmaceutical ingredients, adding complexity and time to the production cycle. These factors collectively hinder the scalability and economic viability of producing selenium-containing compounds on a commercial scale.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes visible light promotion to facilitate the reaction at room temperature, eliminating the need for external heating and reducing energy costs drastically. By employing simple diselenides and olefins directly, the method bypasses the requirement for pre-functionalized selenium reagents, streamlining the synthetic route and improving overall efficiency. The use of a standard 23-watt white compact fluorescent lamp as the light source demonstrates the accessibility and low cost of the required equipment, making it feasible for widespread adoption. This strategy not only enhances functional group compatibility but also ensures high yields, as evidenced by specific examples achieving up to 92% conversion. For those seeking cost reduction in fine chemical manufacturing, this metal-free and energy-efficient protocol offers a compelling alternative to legacy technologies.
Mechanistic Insights into Visible Light-Promoted Selenylation
The core of this innovation lies in the photo-induced generation of selenium radicals which initiate the addition across the olefin double bond without the aid of external photocatalysts in many embodiments. Under the irradiation of visible light, the diselenide bond undergoes homolytic cleavage to generate selenyl radicals that are highly reactive towards electron-rich alkenes. This radical intermediate subsequently interacts with the nitrogen source, such as saccharin or its derivatives, to form the final beta-amino selenide product through a concerted or stepwise mechanism. The mild nature of the radical generation ensures that sensitive functional groups on the substrate remain intact, thereby broadening the scope of applicable starting materials. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for diverse substrate classes.
Impurity control is inherently superior in this system due to the absence of metal catalysts which often lead to side reactions or difficult-to-remove metal complexes. The reaction proceeds with high regioselectivity, minimizing the formation of isomeric byproducts that would otherwise complicate downstream purification. The use of inert gas protection further prevents oxidative degradation of the selenium species, ensuring consistent product quality batch after batch. For Quality Control teams, this translates to a cleaner crude profile and reduced burden on analytical resources. The robustness of the radical pathway under ambient conditions suggests that the process is less susceptible to minor fluctuations in temperature, enhancing reproducibility in large-scale reactors.
How to Synthesize Beta-Amino Selenide Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure that can be adapted for both laboratory screening and pilot plant operations. It involves the sequential addition of reactants into a Schlenk tube followed by irradiation, making it compatible with standard glassware found in most chemical facilities. The detailed standardized synthesis steps below provide a clear roadmap for replicating the high yields reported in the intellectual property documentation. Operators should ensure proper inert atmosphere maintenance to maximize the efficiency of the radical propagation cycle.
- Charge a reactor with olefin, nitrogen-containing compound, and diselenide in a suitable solvent like acetonitrile under inert gas protection.
- Irradiate the reaction mixture with a white fluorescent light source at room temperature while stirring for approximately 20 hours.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to obtain the target beta-amino selenide.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this visible light-promoted technology offers substantial strategic benefits regarding raw material availability and process safety. The elimination of transition metal catalysts removes a significant bottleneck associated with the sourcing and price volatility of precious metals like palladium or platinum. This shift allows procurement teams to secure more stable pricing structures and reduces dependency on single-source suppliers for specialized catalytic systems. Additionally, the operation at room temperature significantly lowers the risk profile of the manufacturing process, potentially reducing insurance costs and simplifying facility safety requirements. These factors contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the reduction in energy consumption due to room temperature operation lead to significant cost savings. By avoiding the need for high-pressure or high-temperature reactors, capital expenditure on specialized equipment is also minimized. The high atom economy of the reaction means less waste generation, which further lowers disposal costs and environmental compliance fees. Overall, the simplified workflow reduces labor hours and increases throughput capacity without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as styrenes and common diselenides ensures a robust supply chain that is less prone to disruptions. The mild reaction conditions allow for flexible manufacturing schedules, as the process does not require extensive warm-up or cool-down periods associated with thermal reactions. This agility enables suppliers to respond more quickly to fluctuating market demands and urgent customer orders. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly optimized.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge. Scaling up this process is facilitated by the use of simple light sources that can be arrayed in flow reactors or large batch vessels. The reduced generation of hazardous waste simplifies the permitting process for new manufacturing lines. This sustainability advantage enhances the corporate social responsibility profile of the production facility and meets the green chemistry goals of major multinational clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and reliability. They serve to clarify the operational feasibility and strategic value of adopting this method for industrial applications.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented visible light-promoted method operates without the need for expensive transition metal catalysts or harsh oxidants, significantly reducing metal residue concerns in pharmaceutical intermediates.
Q: What are the typical reaction conditions for this process?
A: The reaction proceeds efficiently at room temperature under the irradiation of a standard 23-watt white compact fluorescent lamp, utilizing common solvents like acetonitrile or dichloromethane.
Q: Is this method scalable for industrial production?
A: Yes, the mild reaction conditions and high atom economy make this process highly suitable for commercial scale-up, offering improved safety and operational simplicity compared to traditional thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Selenide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions, leveraging technologies like the visible light-promoted synthesis described herein. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale methods are successfully transferred to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-amino selenide meets the highest international standards. Our commitment to technical excellence allows us to offer customized solutions that address the specific needs of complex molecule synthesis.
We invite you to collaborate with us to explore how this cutting-edge technology can enhance your product portfolio and reduce manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Partner with us to secure a sustainable and efficient supply chain for your next generation of selenium-containing compounds.
