Scalable Nickel-Catalyzed Trifluoromethylation for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Trifluoromethylation Technology
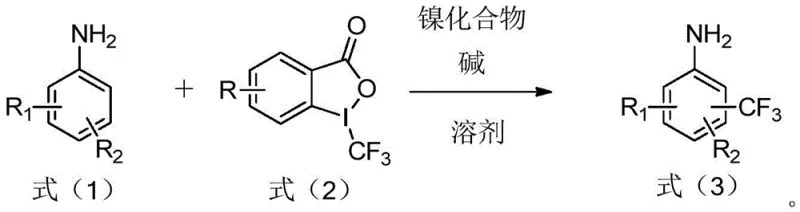
The introduction of trifluoromethyl groups into aromatic systems is a cornerstone strategy in modern medicinal chemistry and agrochemical development, significantly enhancing metabolic stability, lipophilicity, and bioavailability of active pharmaceutical ingredients (APIs). Patent CN108503552B discloses a groundbreaking preparation method for trifluoromethyl aromatic amines that addresses critical bottlenecks in existing synthetic routes. This technology leverages a nickel-catalyzed system utilizing 1-trifluoromethyl-1,2-benziodoxol-3(1H)-one as the trifluoromethylating agent, enabling direct C-H functionalization under remarkably mild conditions. Unlike traditional methods that often require harsh reagents or expensive noble metal catalysts, this approach utilizes abundant nickel compounds and simple inorganic bases, representing a paradigm shift towards more sustainable and economically viable manufacturing processes for high-value fluorinated intermediates.
The strategic importance of this innovation cannot be overstated for global supply chains dependent on fluorinated building blocks. By optimizing reaction parameters such as solvent choice, catalyst loading, and temperature, the disclosed method achieves yields ranging from 60% to 90% across a broad substrate scope. This level of efficiency, combined with the use of commercially available starting materials, positions this technology as a robust solution for the commercial scale-up of complex pharmaceutical intermediates. For R&D teams and procurement specialists alike, understanding the mechanistic nuances and operational advantages of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl aromatic amines has been plagued by significant technical and economic hurdles. Early methodologies, such as those reported by Wakselman involving trifluoromethyl bromide under pressure with zinc and sulfur dioxide, required extreme conditions that posed severe safety risks and engineering challenges for industrial implementation. Subsequent developments introduced electrophilic trifluoromethyl reagents, but these often suffered from limited substrate scope, failing to tolerate diverse functional groups present in complex drug candidates. Furthermore, recent advances utilizing photocatalysis with tris(2-phenylpyridine)iridium, while effective, introduced prohibitive costs due to the reliance on scarce iridium metals and the need for specialized photo-reactors. These conventional pathways often result in high production costs, complex purification procedures to remove heavy metal residues, and inconsistent yields that hinder cost reduction in API manufacturing.
The Novel Approach
The method detailed in CN108503552B offers a transformative alternative by employing a nickel-catalyzed system that operates efficiently without the need for light irradiation or expensive noble metals. The core innovation lies in the synergistic combination of a nickel compound, such as nickel hydroxide, with a base like potassium carbonate in a polar aprotic solvent. This system activates the C-H bond ortho to the amino group through a coordination-assisted mechanism, allowing for the direct installation of the trifluoromethyl group using a stable hypervalent iodine reagent. The process is characterized by its operational simplicity, proceeding at temperatures as low as 35°C to 80°C, which drastically reduces energy consumption compared to high-temperature alternatives. Moreover, the tolerance for various substituents—including halogens, esters, and nitriles—ensures that this method can be applied to late-stage functionalization of advanced intermediates, thereby streamlining synthetic routes and enhancing overall process efficiency.
Mechanistic Insights into Nickel-Catalyzed C-H Trifluoromethylation
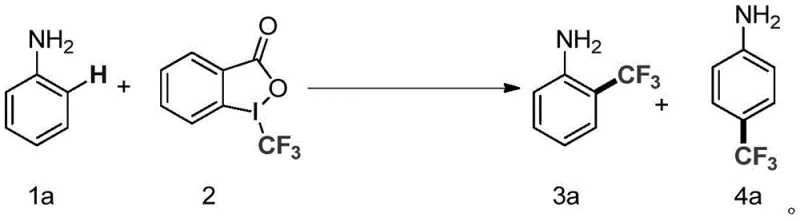
The catalytic cycle proposed for this transformation involves the initial coordination of the nickel catalyst with the amino group of the aromatic substrate, which serves as a directing group to facilitate ortho-C-H activation. This coordination creates a metallacycle intermediate that is susceptible to oxidative addition or radical capture involving the trifluoromethylating reagent, 1-trifluoromethyl-1,2-benziodoxol-3(1H)-one. The presence of the base is critical not only for neutralizing the acidic byproducts generated during the reaction but also for potentially activating the nickel center or the substrate to lower the energy barrier of the C-H cleavage step. Experimental data indicates that nickel hydroxide performs superiorly compared to other nickel salts, likely due to its specific solubility profile and Lewis acidity in solvents like DMSO, which stabilizes the transition states involved in the trifluoromethyl transfer.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this mechanism offers inherent advantages in selectivity. The directing effect of the amino group minimizes random poly-trifluoromethylation, ensuring that the major products are the mono-substituted ortho- and para-isomers. The patent data highlights that optimizing the molar ratio of the aromatic amine to the trifluoromethylating agent (preferably 3:1) significantly suppresses side reactions and improves the isolated yield of the desired isomer. Furthermore, the choice of solvent plays a crucial role in managing the reaction kinetics; polar solvents like DMSO enhance the solubility of the ionic intermediates and facilitate the turnover of the catalytic cycle, leading to cleaner reaction profiles and simplified downstream processing, which is vital for maintaining high-purity pharmaceutical intermediates standards.
How to Synthesize Trifluoromethyl Aromatic Amines Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the careful selection of the nickel catalyst and base, with nickel hydroxide and potassium carbonate identified as the optimal combination for broad substrate applicability. The reaction is typically conducted under an inert atmosphere to prevent oxidation of the catalyst or sensitive intermediates. Following the reaction, a standard aqueous workup followed by silica gel chromatography allows for the isolation of the target compounds with high purity. The detailed standardized synthesis steps for this specific transformation are outlined in the guide below.
- Prepare the reaction mixture by combining the aromatic amine substrate, 1-trifluoromethyl-1,2-benziodoxol-3(1H)-one, a nickel catalyst (e.g., Ni(OH)2), and a base (e.g., K2CO3) in a polar aprotic solvent like DMSO.
- Maintain the reaction temperature between 35°C and 80°C under an inert atmosphere (argon or nitrogen) for 2 to 12 hours to ensure complete conversion while minimizing side reactions.
- Upon completion, quench the reaction with water, extract the organic phase with dichloromethane, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed technology translates into tangible strategic benefits beyond mere technical feasibility. The elimination of expensive iridium photocatalysts and the associated lighting infrastructure represents a significant reduction in raw material and capital costs. Additionally, the ability to run reactions at moderate temperatures (35-80°C) lowers energy requirements and simplifies reactor design, allowing for production in standard stainless steel vessels rather than specialized glass-lined or photo-reactors. This flexibility enhances supply chain resilience by enabling manufacturing across a wider range of facilities, thereby reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The substitution of noble metal catalysts with abundant nickel compounds drastically lowers the cost of goods sold (COGS). Since nickel hydroxide is significantly cheaper than iridium complexes, the overall catalyst cost per kilogram of product is minimized. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps often required for palladium or iridium residues, reduces solvent usage and waste disposal costs, contributing to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including aromatic amines and the benziodoxole-based trifluoromethylating agent, are commercially available from multiple global suppliers. This diversity in sourcing prevents single-point failures in the supply chain. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch reproducibility and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The method's compatibility with green solvents like DMSO and the absence of toxic heavy metals align with increasingly stringent environmental regulations. The high atom economy and reduced waste generation simplify the permitting process for new manufacturing lines. Scalability is further supported by the exothermic nature of the reaction being manageable at lower temperatures, allowing for safe scale-up from laboratory grams to multi-ton commercial production without significant re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is crucial for integrating this chemistry into existing production pipelines.
Q: What are the primary advantages of this nickel-catalyzed method over traditional iridium-based photocatalysis?
A: The nickel-catalyzed method described in CN108503552B eliminates the need for expensive iridium photocatalysts and specialized light equipment. It operates under thermal conditions (35-80°C) rather than requiring specific wavelengths of light, significantly reducing capital expenditure and operational complexity for large-scale manufacturing.
Q: How does the amino group influence the regioselectivity of the trifluoromethylation?
A: The amino group acts as a powerful directing group, facilitating ortho-selective C-H activation. However, depending on the substrate structure and reaction conditions, para-substitution can also occur. The process allows for the synthesis of both ortho- and para-trifluoromethyl aromatic amines, providing flexibility for diverse intermediate synthesis.
Q: Is this process suitable for substrates containing sensitive functional groups like halogens or esters?
A: Yes, the method demonstrates excellent functional group tolerance. The patent data confirms successful trifluoromethylation of substrates containing bromine, chlorine, fluorine, cyano, ester, and ketone groups without significant dehalogenation or hydrolysis, making it highly versatile for complex molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Aromatic Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the nickel-catalyzed pathway described in CN108503552B and possesses the technical expertise to optimize it for your specific project needs. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl aromatic amine delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your next-generation therapeutics. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your supply chain.
