Advanced 2-Morpholinone Manufacturing: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic scaffolds, and the recent disclosure in patent CN108640884B offers a compelling solution for the production of 2-morpholinone. This specific chemical entity serves as a pivotal building block in the synthesis of quinazoline derivatives, which are extensively investigated for their potent anticancer properties. The disclosed methodology represents a significant departure from conventional oxidative strategies, introducing a reductive amination approach followed by a protected cyclization sequence. By leveraging cheap and readily available starting materials such as glyoxylic acid and ethanolamine, this innovation addresses long-standing challenges regarding cost efficiency and operational simplicity. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The technical depth of this process ensures that manufacturers can achieve stringent quality specifications while maintaining economic viability in a competitive market landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-morpholinone has been plagued by inefficient methodologies that rely heavily on metal-catalyzed oxidation processes. These traditional routes often necessitate the use of expensive oxidants and complex transition metal catalysts, which introduce significant risks regarding heavy metal contamination in the final active pharmaceutical ingredient. Furthermore, the reaction conditions in older protocols are frequently harsh, leading to the formation of various structural isomers that are notoriously difficult to separate without resorting to resource-intensive purification techniques like column chromatography. Such limitations not only drive up the cost of goods sold but also create bottlenecks in the supply chain, as the purification steps are time-consuming and difficult to scale. The low overall yields associated with these oxidative methods further exacerbate the economic burden, making it challenging for manufacturers to meet the growing global demand for quinazoline-based therapeutics without compromising on margin or delivery timelines.
The Novel Approach
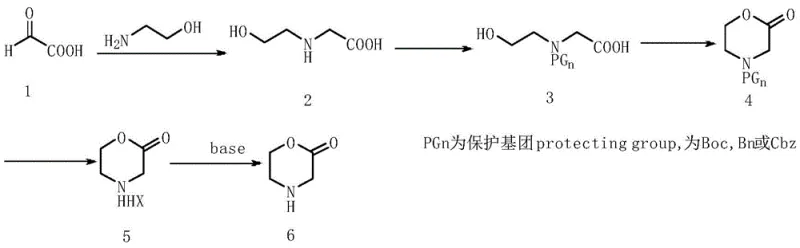
In stark contrast, the novel approach detailed in the patent utilizes a strategic sequence beginning with the reductive amination of glyoxylic acid and ethanolamine to form N-(2-hydroxyethyl)glycine. This intermediate is subsequently protected using standard agents such as di-tert-butyl dicarbonate or benzyl chloroformate, which effectively masks the reactive amino group and prevents unwanted side reactions during the subsequent cyclization step. The core transformation involves an acid-catalyzed lactonization in a refluxing solvent like toluene, driven by catalysts such as p-toluenesulfonic acid or concentrated sulfuric acid. This dehydration cyclization is highly efficient and avoids the generation of complex byproduct mixtures. The final deprotection step yields the 2-morpholinone salt directly, which can be easily converted to the free base. This streamlined pathway eliminates the need for chromatographic purification entirely, relying instead on simple crystallization and extraction, thereby drastically simplifying the manufacturing workflow and enhancing the overall process robustness.
Mechanistic Insights into Acid-Catalyzed Lactonization and Deprotection
The mechanistic elegance of this synthesis lies in the precise control of reactivity through protecting group chemistry and acid catalysis. The initial formation of N-(2-hydroxyethyl)glycine via hydrogenation establishes the carbon-nitrogen backbone required for the morpholine ring. Upon protection, the molecule becomes stable enough to withstand the thermal conditions required for ring closure. The cyclization step is driven by the protonation of the carboxylic acid moiety by strong acids like p-toluenesulfonic acid, facilitating a nucleophilic attack by the hydroxyl group or vice versa depending on the specific activation mode, ultimately leading to the loss of water and the formation of the six-membered lactam ring. This intramolecular condensation is thermodynamically favored under reflux conditions with water removal. Following cyclization, the removal of the protecting group—whether via acidolysis for Boc groups or hydrogenolysis for Cbz groups—unmasks the secondary amine, yielding the target 2-morpholinone structure with high fidelity.
Impurity control is inherently built into this design by avoiding the radical mechanisms often seen in oxidation reactions. In traditional oxidative routes, over-oxidation or incomplete reaction can lead to a spectrum of oxygenated byproducts and regioisomers that share similar physical properties with the target molecule, complicating purification. By contrast, the reductive and acid-catalyzed steps in this new protocol proceed through well-defined ionic intermediates. The use of protecting groups ensures that the amine functionality does not interfere with the esterification or lactonization events, thereby suppressing the formation of polymeric side products or open-chain oligomers. Consequently, the crude product obtained after the cyclization step is of sufficient purity that it often requires only recrystallization to meet pharmaceutical standards. This inherent selectivity reduces the burden on downstream processing and ensures a cleaner impurity profile, which is a critical parameter for regulatory approval in drug substance manufacturing.
How to Synthesize 2-Morpholinone Efficiently
The practical execution of this synthesis involves a series of standardized unit operations that are familiar to any process chemist, ensuring easy technology transfer from the lab to the pilot plant. The process begins with the hydrogenation of glyoxylic acid and ethanolamine in an aqueous medium using a palladium-carbon catalyst, followed by the protection of the resulting amino acid derivative. The key cyclization step requires careful temperature control in a refluxing aromatic solvent to drive the equilibrium towards the lactam. Finally, the deprotection is achieved under mild acidic conditions or hydrogenation, followed by neutralization to isolate the free base. Each step is designed to maximize yield while minimizing waste, reflecting a green chemistry philosophy. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the standardized synthesis guide provided below.
- Perform catalytic hydrogenation of glyoxylic acid and ethanolamine to form N-(2-hydroxyethyl)glycine.
- Protect the amino group using agents like Boc-anhydride or Cbz-chloride to prevent side reactions.
- Execute acid-catalyzed cyclization in refluxing toluene to form the morpholinone ring structure.
- Remove the protecting group via acidification or hydrogenolysis to obtain the final 2-morpholinone salt or free base.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for pharmaceutical intermediates. The shift away from expensive oxidants and precious metal catalysts towards commodity chemicals like glyoxylic acid and simple mineral acids results in a substantial reduction in raw material costs. Furthermore, the elimination of column chromatography, which is a major bottleneck in fine chemical manufacturing, significantly lowers the operational expenditure related to silica gel, solvents, and labor. This streamlined process not only accelerates the production cycle but also enhances the reliability of supply by reducing the complexity of the manufacturing workflow. Companies adopting this route can expect a more resilient supply chain capable of meeting large-volume demands without the volatility associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the substitution of high-cost reagents with inexpensive, bulk-available starting materials. By utilizing glyoxylic acid and ethanolamine, the baseline material cost is kept minimal compared to specialized cyclic amine precursors used in other methods. Additionally, the avoidance of heavy metal catalysts removes the necessity for expensive scavenging resins and rigorous metal testing protocols, further driving down the cost of quality assurance. The high atom economy of the cyclization step ensures that a greater proportion of the input mass is converted into the valuable product, minimizing waste disposal costs. Overall, the simplified purification train translates directly into lower manufacturing overheads, allowing for more competitive pricing in the global marketplace for API intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of non-proprietary, commercially abundant raw materials. Unlike specialized reagents that may be subject to geopolitical supply constraints or single-source dependencies, the inputs for this synthesis are produced by multiple vendors globally. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic high-pressure or cryogenic setups, means that production can be easily distributed across multiple manufacturing sites. This flexibility mitigates the risk of production stoppages due to equipment failure or localized disruptions. Moreover, the stability of the intermediates allows for potential stockpiling at various stages of the synthesis, providing a buffer against sudden spikes in demand and ensuring consistent delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling this process from kilogram to tonnage levels is straightforward due to the reliance on conventional reaction types such as hydrogenation, reflux, and liquid-liquid extraction. The absence of chromatographic purification is a critical factor for scalability, as column chromatography is notoriously difficult to implement efficiently on a multi-ton scale. From an environmental standpoint, the reduction in solvent usage and the elimination of silica waste contribute to a lower environmental footprint, aligning with increasingly stringent global regulations on chemical manufacturing emissions. The process generates fewer hazardous byproducts, simplifying wastewater treatment and waste management protocols. This alignment with green chemistry principles not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturer, a key consideration for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-morpholinone based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own drug development pipelines. The answers reflect the consensus on the method's efficiency, purity profile, and adaptability to industrial settings.
Q: What are the primary advantages of this 2-morpholinone synthesis route over traditional oxidation methods?
A: This method avoids the use of expensive oxidants and complex metal catalysts found in traditional routes, significantly reducing heavy metal residues and simplifying the purification process by eliminating the need for column chromatography.
Q: How does the new process impact the purity and isolation of 2-morpholinone?
A: By utilizing specific protecting groups and acid-catalyzed cyclization, the process minimizes the formation of difficult-to-separate isomers, allowing for high-purity isolation through simple crystallization or extraction techniques.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the protocol relies on common unit operations such as reflux, filtration, and solvent extraction, using cheap and readily available raw materials like glyoxylic acid, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Morpholinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We are committed to delivering 2-morpholinone that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced synthetic strategies like the one detailed in CN108640884B, we offer a product that is not only chemically superior but also economically optimized for your supply chain needs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new developments, we are ready to provide the data-driven insights you need. Partner with us to secure a stable, high-quality supply of 2-morpholinone and accelerate your path to market with confidence.
