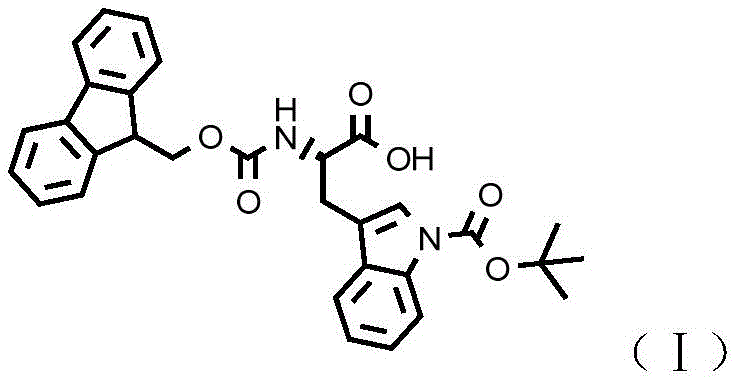
Advanced Synthesis of Fmoc-Trp(Boc)-OH: Enhancing Purity and Commercial Scalability
Advanced Synthesis of Fmoc-Trp(Boc)-OH: Enhancing Purity and Commercial Scalability
The pharmaceutical industry's relentless pursuit of efficient peptide synthesis has brought significant attention to the preparation of high-purity protected amino acids, specifically N-alpha-9-fluorenylmethoxycarbonyl-N-in-tert-butoxycarbonyl-L-tryptophan, commonly known as Fmoc-Trp(Boc)-OH. This critical intermediate, identified by CAS number 143824-78-6, serves as a fundamental building block for the synthesis of complex peptide therapeutics such as semaglutide and teriparatide. Recent technological advancements disclosed in patent CN115286554A offer a transformative approach to manufacturing this compound, addressing long-standing challenges related to cost, equipment requirements, and impurity profiles. By shifting away from expensive fluorinated reagents and high-pressure hydrogenation processes, this new methodology leverages a robust trityl-protection strategy that aligns perfectly with the needs of modern reliable pharmaceutical intermediate supplier networks seeking scalable and compliant production routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fmoc-Trp(Boc)-OH has been plagued by significant operational and economic hurdles that hinder large-scale commercialization. Traditional pathways often relied on trifluoroacetyl groups as temporary protecting agents, necessitating the use of costly trifluoroacetic anhydride. The removal of these groups typically required harsh reaction conditions that posed a severe risk to the stability of other sensitive protecting groups, such as the tert-butoxycarbonyl (Boc) moiety, leading to compromised purity and difficult downstream purification. Alternatively, other established methods employed benzyloxycarbonyl groups, which mandated high-pressure catalytic hydrogenation for deprotection. This requirement not only introduced substantial capital expenditure for specialized high-pressure reactors but also depended on expensive noble metal catalysts, creating a bottleneck for cost reduction in API manufacturing and limiting the flexibility of production facilities.
The Novel Approach
In stark contrast to these legacy techniques, the innovative process detailed in the patent utilizes triphenylmethanol (trityl alcohol) as a temporary amino protecting group, fundamentally altering the economic and technical landscape of production. This strategic substitution allows the reaction to proceed under remarkably mild conditions, typically between 0°C and 60°C, without the need for extreme pressures or hazardous fluorinated reagents. The trityl group can be introduced efficiently and, crucially, removed under controlled acidic conditions that preserve the integrity of the Boc group, thereby significantly enhancing the overall yield and purity of the final product. This streamlined workflow eliminates the dependency on heavy metal catalysts and high-pressure infrastructure, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates in diverse manufacturing environments.
Mechanistic Insights into Trityl-Mediated Protection Strategy
The core of this technological breakthrough lies in the precise mechanistic control of the trityl protection and deprotection cycle. In the initial protection phase, triphenylmethanol reacts with the L-tryptophan ester hydrochloride in the presence of a dehydrating agent, such as acetic anhydride or acetic acid. This reagent plays a dual role: it acts as a solvent or co-solvent while simultaneously consuming the water generated during the condensation reaction. By continuously removing water from the equilibrium, the reaction is driven decisively towards the formation of the N-alpha-trityl-L-tryptophan ester, achieving conversion rates that consistently exceed 86%. This thermodynamic driving force ensures that the reaction proceeds to completion without requiring excessive temperatures that could degrade the sensitive indole ring of the tryptophan side chain.
Furthermore, the deprotection mechanism is engineered to maximize selectivity and minimize impurity generation. The removal of the bulky trityl group is accomplished using mild organic acids like acetic acid at moderate temperatures (30-60°C). This specific condition window is critical because it is sufficiently energetic to cleave the trityl-amino bond yet gentle enough to leave the acid-labile Boc group on the indole nitrogen intact. This orthogonality in protecting group stability is the key to maintaining a clean impurity profile, as it prevents the formation of de-Boc by-products that are notoriously difficult to separate. Consequently, the resulting N-in-tert-butoxycarbonyl-L-tryptophan ester is obtained with high purity (over 98%), providing a superior substrate for the final Fmoc installation step and ensuring the quality of the high-purity pharmaceutical intermediate.
How to Synthesize Fmoc-Trp(Boc)-OH Efficiently
The implementation of this synthesis route involves a logical six-step sequence designed to maximize yield at every stage while minimizing waste and operational complexity. The process begins with the esterification of L-tryptophan, followed by the critical trityl protection and subsequent Boc installation on the indole nitrogen. Once the dual protection is secured, the temporary trityl group is selectively removed, the ester is hydrolyzed to the free acid, and finally, the Fmoc group is attached to the alpha-amino position. This systematic approach ensures that each functional group is manipulated in an order that preserves stereochemical integrity and chemical stability. For laboratory and pilot plant teams looking to adopt this methodology, the detailed standardized synthetic steps are outlined below to ensure reproducibility and safety.
- Esterify L-tryptophan with alcohol and hydrogen chloride to form L-tryptophan ester hydrochloride.
- Protect the amino group using triphenylmethanol and a dehydrating agent like acetic anhydride to obtain N-alpha-trityl-L-tryptophan ester.
- Condense with di-tert-butyl dicarbonate to introduce the Boc protecting group on the indole nitrogen.
- Remove the trityl group under mild acidic conditions to regenerate the free alpha-amino group.
- Hydrolyze the ester under alkaline conditions to obtain N-in-tert-butoxycarbonyl-L-tryptophan.
- React with Fmoc-OSu to install the final Fmoc group, yielding the target double-protected amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this trityl-based synthesis route represents a significant opportunity to optimize the total cost of ownership for peptide raw materials. By eliminating the need for high-pressure hydrogenation equipment and expensive noble metal catalysts, manufacturers can drastically reduce both capital expenditure and ongoing operational costs. The reliance on commodity chemicals like triphenylmethanol and acetic acid, rather than specialized fluorinated reagents, further stabilizes the supply chain against price volatility and sourcing disruptions. This robustness ensures a more predictable delivery schedule for reducing lead time for high-purity pharmaceutical intermediates, allowing downstream peptide synthesizers to plan their production campaigns with greater confidence and efficiency.
- Cost Reduction in Manufacturing: The replacement of trifluoroacetic anhydride and noble metal catalysts with economically accessible reagents like triphenylmethanol results in substantial raw material savings. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the simplified workup procedures lower solvent usage and waste disposal costs. These cumulative effects drive down the unit cost of production without compromising quality, offering a competitive edge in the global market for peptide building blocks.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of supply bottlenecks often associated with specialized reagents. Since the process does not require unique high-pressure reactors, it can be executed in a broader range of standard chemical manufacturing facilities, increasing the available capacity and redundancy within the supply network. This flexibility ensures consistent availability of Fmoc-Trp(Boc)-OH, safeguarding the production timelines for critical peptide drugs like semaglutide.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts simplifies the purification process and reduces the environmental burden of heavy metal waste, facilitating easier compliance with stringent environmental regulations. The mild temperature profile (0-60°C) and atmospheric pressure operation make the process inherently safer and easier to scale from kilogram to multi-ton quantities. This scalability supports the growing demand for peptide therapeutics while adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this synthesis route for their specific applications, we have compiled answers to common inquiries regarding the process parameters and quality attributes. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production lines or for qualifying new suppliers.
Q: Why is triphenylmethanol preferred over trifluoroacetic anhydride for temporary protection?
A: Triphenylmethanol is significantly more economical and easier to handle than trifluoroacetic anhydride. Furthermore, the deprotection of the trityl group occurs under milder conditions, reducing the risk of damaging the acid-sensitive Boc protecting group and minimizing by-product formation.
Q: What are the typical yields for the key protection and deprotection steps?
A: According to the patent data, the trityl protection step achieves a yield of over 86%, while the subsequent trityl deprotection step reaches a yield of approximately 89%, demonstrating high efficiency throughout the synthesis.
Q: Does this method require high-pressure hydrogenation equipment?
A: No, unlike previous methods that utilized benzyloxycarbonyl groups requiring high-pressure catalytic hydrogenation and noble metal catalysts, this novel route operates under standard atmospheric pressure and mild temperatures (0-60°C), eliminating the need for specialized high-pressure reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-Trp(Boc)-OH Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality protected amino acids play in the success of modern peptide drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering Fmoc-Trp(Boc)-OH with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and impurity profiles. Our capability to implement the advanced trityl-protection technology described in patent CN115286554A allows us to offer a product that balances exceptional quality with commercial viability.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your peptide synthesis projects proceed with the highest level of reliability and efficiency.
