Scalable Manufacturing of 2-Fluoro-5-Formyl Chloropyridine via Optimized Oxidation-Chlorination
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for higher purity and more scalable manufacturing processes. Patent CN112358442B introduces a significant advancement in the preparation of 2-fluoro-5-formyl chloropyridine, a critical building block known chemically as 6-fluoro-3-formylchloropyridine (CAS: 65352-94-5). This compound serves as a pivotal precursor for synthesizing hydrazides, acylhydrazones, and amides, which are essential structures in modern pesticide and pharmaceutical development. The patent outlines a robust two-step synthetic route that transforms 2-fluoro-5-methylpyridine into the target acid chloride with exceptional efficiency. By leveraging a specific oxidation protocol followed by a carefully controlled acyl chlorination, the methodology addresses long-standing challenges regarding conversion rates and impurity profiles. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this technology represents a shift towards more predictable and high-yielding production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridine acid chlorides has been fraught with difficulties related to reaction selectivity and downstream purification. Conventional oxidation methods often suffer from incomplete conversion of the methyl group to the carboxylic acid, leading to complex mixtures that are difficult to separate. Furthermore, traditional acyl chlorination processes frequently encounter issues when scaling up, particularly the formation of insoluble hydrochloride salts that can clog reactors and reduce overall throughput. Many existing market offerings of 2-fluoro-5-formyl chloropyridine are limited to small reagent quantities with purities hovering around 98%, which is insufficient for the rigorous demands of active pharmaceutical ingredient (API) manufacturing. These limitations result in increased waste generation, higher solvent consumption for recrystallization, and ultimately, a less cost-effective supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in the patent offers a streamlined solution by optimizing both the oxidation and chlorination steps to ensure industrial viability. The process begins with the oxidation of 2-fluoro-5-methylpyridine using potassium permanganate or sodium permanganate in the presence of a base, which dramatically enhances conversion efficiency. Following the isolation of the carboxylic acid intermediate, the subsequent reaction with thionyl chloride or oxalyl chloride is conducted under specific conditions that prevent the precipitation of unwanted salts. By incorporating mechanical adjustments such as high-speed stirring and inert gas bubbling, the process maintains a homogeneous reaction mixture even at larger scales. This novel approach not only simplifies the workflow but also ensures that the final product meets stringent quality standards, positioning it as a superior choice for cost reduction in electronic chemical manufacturing and fine chemical synthesis.
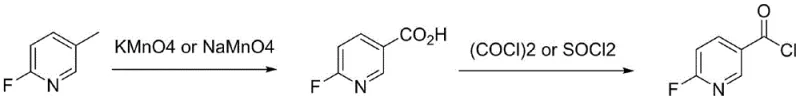
Mechanistic Insights into Permanganate Oxidation and Acyl Chlorination
The core of this synthetic strategy lies in the mechanistic optimization of the oxidation step, where the methyl group on the pyridine ring is converted to a carboxylic acid. The use of permanganate salts (KMnO4 or NaMnO4) provides a strong oxidizing environment, but the patent highlights a critical modification: the addition of sodium hydroxide or potassium hydroxide. Mechanistically, the base likely facilitates the deprotonation of intermediate species or stabilizes the permanganate ion, thereby driving the reaction to completion more effectively than the oxidant alone. The molar ratio of oxidant to substrate is maintained between 2:1 and 4:1, ensuring that there is sufficient oxidative power to fully convert the starting material without excessive waste. This careful balancing of reagents minimizes the formation of over-oxidized byproducts or ring-degraded impurities, which is crucial for maintaining the integrity of the fluorinated pyridine scaffold.
In the second stage, the conversion of the carboxylic acid to the acid chloride involves nucleophilic acyl substitution using thionyl chloride or oxalyl chloride. A key mechanistic challenge identified in this step is the potential formation of 2-fluoro-5-formylchloropyridine hydrochloride, a solid byproduct that can trap the desired product and lower yields. The patent elucidates that this issue is exacerbated by poor mixing or lack of gas exchange during the reflux period. By introducing nitrogen or argon directly into the liquid phase, the system effectively removes generated hydrogen chloride gas, shifting the equilibrium towards the product and preventing salt precipitation. Furthermore, the use of chlorinated solvents like dichloromethane followed by precipitation with alkanes allows for a highly effective purification strategy, ensuring that the final distillation yields a product with single impurities below 0.5%.
How to Synthesize 2-Fluoro-5-Formyl Chloropyridine Efficiently
The synthesis of this valuable intermediate requires precise control over reaction parameters to maximize yield and purity. The process is divided into two distinct phases: the aqueous oxidation of the methyl precursor and the anhydrous chlorination of the resulting acid. Operators must pay close attention to temperature control during the oxidation phase, maintaining the reaction at approximately 95°C to ensure complete conversion within a 5-hour window. Following the isolation of the acid, the chlorination step demands rigorous exclusion of moisture and efficient gas management to handle the evolution of corrosive byproducts. The detailed standardized synthesis steps, including specific stoichiometric ratios and workup procedures, are outlined in the guide below to assist technical teams in replicating these results.
- Oxidize 2-fluoro-5-methylpyridine using KMnO4 or NaMnO4 with a base additive at 95°C to form 2-fluoro-5-pyridinecarboxylic acid.
- React the isolated acid with thionyl chloride or oxalyl chloride in a chlorinated solvent under reflux conditions.
- Remove solvents, precipitate impurities using an alkane solvent, and purify via reduced pressure distillation to obtain the final acid chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits that extend beyond simple chemical yield. The reliance on commodity chemicals such as potassium permanganate and thionyl chloride means that the raw material supply chain is robust and less susceptible to the volatility often seen with exotic catalysts. The elimination of transition metal catalysts, which are common in alternative coupling strategies, removes the need for expensive and time-consuming heavy metal scavenging steps. This simplification of the downstream processing directly translates to substantial cost savings in manufacturing, as it reduces both the consumption of specialized reagents and the volume of hazardous waste requiring disposal. Consequently, this route supports a more sustainable and economically viable production model for high-volume intermediates.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available oxidants and chlorinating agents, avoiding the need for precious metal catalysts that drive up production costs. By optimizing the stoichiometry and reaction conditions, the method achieves high conversion rates, which minimizes the loss of valuable starting materials and reduces the overall cost per kilogram of the final product. The simplified workup procedure, which avoids complex chromatographic separations, further lowers operational expenses by reducing solvent usage and labor time associated with purification.
- Enhanced Supply Chain Reliability: Since the raw materials required for this synthesis are bulk commodities produced by multiple global suppliers, the risk of supply disruption is significantly mitigated. The robustness of the reaction conditions allows for flexible manufacturing schedules, enabling producers to respond quickly to fluctuations in market demand without lengthy lead times for specialized reagent procurement. This reliability is critical for maintaining continuous production lines in the pharmaceutical and agrochemical sectors, where interruptions can have cascading effects on downstream drug formulation.
- Scalability and Environmental Compliance: The patent explicitly addresses scale-up challenges, such as solid formation and gas evolution, providing clear engineering solutions like high-speed stirring and inert gas sparging. These features ensure that the process can be safely transferred from laboratory benchtop to multi-ton industrial reactors without losing efficiency. Additionally, the ability to recover and recycle solvents, combined with the absence of toxic heavy metals, aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details is essential for process chemists and engineers looking to integrate this technology into their existing manufacturing frameworks. The answers are derived directly from the experimental data and technical disclosures found within the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What is the primary advantage of the oxidation step in this patent?
A: The addition of a base (NaOH or KOH) during the permanganate oxidation significantly improves reaction conversion compared to using the oxidant alone, ensuring high yields of the carboxylic acid intermediate.
Q: How does the process address solid formation during acyl chlorination?
A: The patent identifies that solid hydrochloride salts can form during scale-up. This is mitigated by increasing stirring speeds to 200-400 rpm, introducing nitrogen gas below the liquid level, and optimizing the cooling and filtration sequence.
Q: What purity levels can be achieved with this method?
A: The described method consistently achieves product purity greater than 99.0%, with single impurities controlled to less than 0.5%, making it suitable for high-specification pharmaceutical and agrochemical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Fluoro-5-Formyl Chloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics and crop protection agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to implement the optimized oxidation-chlorination route described in recent patents allows us to offer a superior product profile that meets the exacting standards of international regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can enhance the efficiency and reliability of your production processes.
