Advanced Synthesis of 2-Nitro-4-Mesyltoluene for High-Purity Mesotrione Manufacturing
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN103121961A introduces a robust and economically viable synthesis method for 2-nitro-4-mesyltoluene, a pivotal precursor in the manufacturing of mesotrione. This novel approach addresses long-standing challenges in the industry, specifically targeting the limitations of yield, purity, and environmental safety that have plagued traditional production methods. By leveraging a sequential sulfonation and chlorosulfonation strategy, the patent outlines a pathway that not only enhances reaction efficiency but also aligns with modern green chemistry principles. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the technical nuances of this patent is essential for optimizing supply chains and reducing manufacturing costs in the competitive herbicide market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitro-4-mesyltoluene has been fraught with significant technical and economic hurdles that hinder large-scale industrial adoption. One prominent conventional route, documented by Olah et al., utilizes 2-nitrotoluene reacted with methylsulfonic acid anhydride and boron trifluoride. While conceptually straightforward, this method suffers from a critically low yield of approximately 47%, rendering it economically unfeasible for mass production. Furthermore, the generation of substantial quantities of spent acid creates severe environmental disposal challenges, increasing the overall cost burden and ecological footprint of the process.
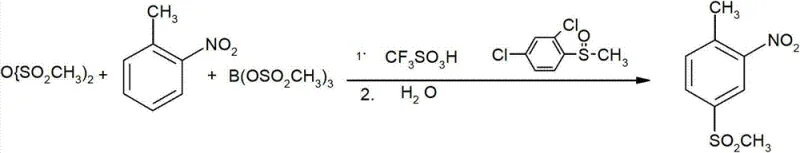
Alternative pathways have attempted to circumvent these issues but introduced new complexities. For instance, direct sulfonation methods often result in complex mixtures of isomers, including 2-nitro-5-methylsulfonyl toluene and 2-nitro-6-methylsulfonyl toluene, which are extremely difficult to separate to the required specification quality. Other routes relying on p-toluenesulfonyl chloride as a starting material face volatility in raw material pricing and hydrolysis stability issues. Perhaps most concerning are methods involving mercaptans, which possess strong, noxious odors and high toxicity, alongside reaction conditions requiring extreme temperatures up to 600°C. These factors collectively compromise operational safety and make consistent commercial scale-up of complex agrochemical intermediates nearly impossible using legacy technologies.
The Novel Approach
The methodology disclosed in patent CN103121961A represents a paradigm shift in the manufacturing of this key intermediate by establishing a streamlined, high-yield synthetic route. The core innovation lies in the strategic conversion of 2-nitrotoluene into 3-nitro-4-methylbenzenesulfonyl chloride via a controlled sulfonation followed by an acyl-chlorination step. This intermediate is then seamlessly transformed into the final 2-nitro-4-mesyltoluene product through a reaction with sodium sulfite, sodium carbonate, and chloroacetic acid. This sequence effectively bypasses the formation of difficult-to-separate isomers and eliminates the need for hazardous mercaptan reagents.
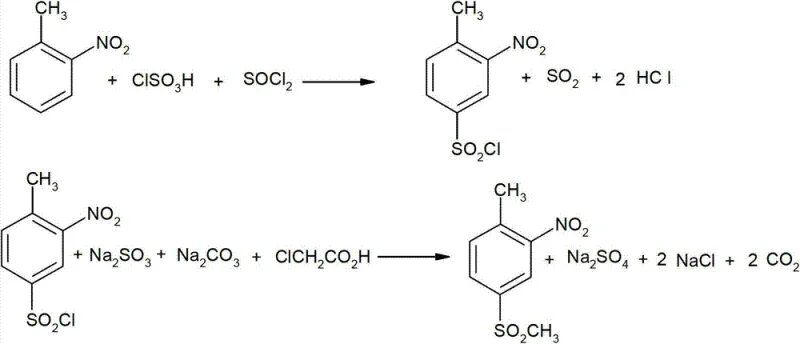
By optimizing the molar ratios of chlorosulfonic acid to 2-nitrotoluene between 1.05:1 and 1.20:1, and maintaining precise temperature controls during the acyl-chlorination phase, the process achieves exceptional conversion rates. The elimination of tedious purification steps associated with isomer separation significantly reduces processing time and solvent consumption. This novel approach not only ensures a product purity exceeding 99% but also establishes a foundation for cost reduction in agrochemical manufacturing by utilizing widely available and stable raw materials, thereby securing a more resilient supply chain for downstream herbicide producers.
Mechanistic Insights into Sulfonation and Acyl-Chlorination
The chemical efficacy of this synthesis relies heavily on the precise control of electrophilic aromatic substitution and subsequent nucleophilic displacement mechanisms. In the initial sulfonation stage, chlorosulfonic acid acts as both the sulfonating agent and the solvent, facilitating the introduction of the sulfonyl group onto the aromatic ring of 2-nitrotoluene. The presence of the nitro group directs the incoming electrophile primarily to the para-position relative to the methyl group, although ortho-substitution is sterically hindered. Maintaining the reaction temperature between 110°C and 120°C is critical to driving the equilibrium towards the desired sulfonic acid intermediate while minimizing side reactions such as polysulfonation or oxidative degradation of the organic substrate.
Following sulfonation, the addition of a catalytic amount of DMF and thionyl chloride initiates the acyl-chlorination mechanism. The DMF activates the thionyl chloride, generating a highly reactive Vilsmeier-Haack type intermediate that efficiently converts the sulfonic acid group into the corresponding sulfonyl chloride. This transformation is conducted at a moderate temperature range of 60°C to 65°C to prevent thermal decomposition of the sensitive nitro-aromatic structure. The resulting 3-nitro-4-methylbenzenesulfonyl chloride is a versatile electrophile that undergoes nucleophilic substitution with sodium sulfite in the final stage. The subsequent addition of chloroacetic acid serves to methylate the sulfonate group, finalizing the mesyl functionality. This multi-step cascade is designed to maximize atom economy and minimize the formation of inorganic salt byproducts, ensuring a cleaner reaction profile.
Impurity control is intrinsically built into this mechanistic pathway through the regioselectivity of the initial sulfonation and the specificity of the chlorination step. Unlike nitration-first routes that often lead to polynitrated byproducts which are hazardous and difficult to remove, this method introduces the nitro group prior to the sulfonation sequence (using commercially available 2-nitrotoluene). This pre-existing substitution pattern dictates the orientation of subsequent reactions, effectively suppressing the formation of unwanted isomers like the 5- or 6-sulfonyl variants. Furthermore, the aqueous workup in the final step allows for the efficient removal of inorganic salts such as sodium sulfate and sodium chloride, resulting in a crude product that requires minimal recrystallization to meet stringent purity specifications required for pharmaceutical and agrochemical applications.
How to Synthesize 2-Nitro-4-Mesyltoluene Efficiently
The practical implementation of this synthesis requires careful attention to reaction kinetics and thermal management to ensure reproducibility and safety. The process begins with the slow addition of 2-nitrotoluene to chlorosulfonic acid under reflux conditions, followed by the controlled introduction of thionyl chloride in the presence of a catalyst. The intermediate sulfonyl chloride is typically used directly in the next step without isolation, which streamlines the workflow and reduces material loss. The final conversion involves a biphasic reaction system where temperature gradients are managed to optimize the nucleophilic attack of the sulfite ion. For detailed operational parameters and safety protocols, the standardized synthesis steps are outlined below.
- Perform sulfonation by reacting 2-nitrotoluene with chlorosulfonic acid at 110-120°C to form the sulfonic acid intermediate.
- Conduct acyl-chlorination by adding a catalyst (DMF) and thionyl chloride to the mixture at 60-65°C to generate 3-nitro-4-methylbenzenesulfonyl chloride.
- React the sulfonyl chloride with sodium sulfite, sodium carbonate, and chloroacetic acid in an aqueous system to finalize the synthesis of 2-nitro-4-mesyltoluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the substantial cost savings achieved by replacing expensive and unstable starting materials like p-toluenesulfonyl chloride with commodity chemicals such as 2-nitrotoluene and chlorosulfonic acid. This shift in raw material sourcing mitigates exposure to volatile market prices and ensures a more predictable cost structure for long-term production contracts. Additionally, the simplified process flow reduces the requirement for specialized equipment capable of handling extreme temperatures or highly toxic gases, thereby lowering capital expenditure requirements for facility upgrades.
- Cost Reduction in Manufacturing: The elimination of costly purification steps associated with isomer separation directly translates to lower operational expenditures. By avoiding the use of precious metal catalysts or exotic reagents, the process minimizes the financial burden of raw material procurement. Furthermore, the high conversion efficiency reduces the volume of waste generated per kilogram of product, leading to significant savings in waste treatment and disposal costs. This lean manufacturing approach allows suppliers to offer more competitive pricing without compromising on margin, providing a distinct advantage in price-sensitive agrochemical markets.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by the scarcity of niche precursors. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in utility supplies or ambient conditions, guaranteeing consistent output quality. This reliability is crucial for maintaining just-in-time inventory levels for downstream herbicide manufacturers, reducing the risk of stockouts and ensuring continuity of supply for critical agricultural seasons.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-kilogram batches with consistent results. The integration of tail gas absorption systems for hydrogen chloride aligns with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. By minimizing the use of toxic mercaptans and avoiding high-energy consumption steps, the technology supports corporate sustainability goals and facilitates easier permitting for capacity expansion, making it an ideal candidate for commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-nitro-4-mesyltoluene based on the patented methodology. These insights are derived from a detailed analysis of the reaction parameters and comparative performance data against legacy methods. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This route avoids the use of expensive p-toluenesulfonyl chloride and toxic mercaptans found in older methods. It achieves higher yields (over 80%) and superior purity (99.5%) while utilizing readily available raw materials like 2-nitrotoluene.
Q: How does this process address environmental and safety concerns in agrochemical manufacturing?
A: Unlike routes requiring high temperatures (600°C) or generating large amounts of spent acid, this method operates at moderate temperatures (40-120°C) and includes efficient tail gas absorption systems for hydrogen chloride, significantly reducing environmental impact.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It eliminates difficult separation steps associated with isomer mixtures and uses standard reaction conditions that are easily adaptable to commercial reactors, ensuring consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Mesyltoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch against the highest industry standards. Our capability to implement the advanced synthesis methods described in patent CN103121961A allows us to offer a superior product profile that supports the efficiency and safety of your final herbicide formulations.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your production economics. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
